Journal of Infectious Diseases and Epidemiology
Building Surveillance Capacity: Lessons Learned from a Ten Year Experience
Pamela J Kennedy1, Sajata Outin2, Tomas R Rodriguez3, Lauren Polansky3, Margaret McCarron3 and Karen R Siener3, Ann C Moen3*
1Centers for Disease Control and Prevention, Atlanta, USA
2Centers for Disease Control and Prevention, McKing Consulting Corporation, Atlanta, USA
3Centers for Disease Control and Prevention, Time Solutions, LLC, Atlanta, USA
*Corresponding author:
Ann C Moen, Centers for Disease Control and Prevention, 1600 Clifton Road, NE, MSA20, Atlanta, Georgia 30329, USA, Tel: +404-639-4652, Fax: +404-639-2334, E-mail: alc3@cdc.gov
J Infect Dis Epidemiol, JIDE-3-026, (Volume 3, Issue 1), Review Article; ISSN: 2474-3658
Received: October 24, 2016 | Accepted: March 06, 2017 | Published: March 08, 2017
Citation: Kennedy PJ, Outin S, Rodriguez TR, Polansky L, McCarron M, et al. (2017) Building Surveillance Capacity: Lessons Learned from a Ten Year Experience. J Infect Dis Epidemiol 3:026. 10.23937/2474-3658/1510026
Copyright: © 2017 Kennedy PJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
As Global Health Security partners endeavor to support capacity building globally, it is important to examine prior efforts for building surveillance and laboratory systems for emerging infectious diseases. CDC provides financial support and technical assistance to foreign governments for the development or improvement of influenza surveillance networks in over fifty countries through cooperative agreements. In 2004, nine countries were awarded a first round of capacity building agreements and seven of these countries transitioned to sustainability agreements in 2009. The seven countries had active World Health Organization (WHO) National Influenza Centers (NIC) and were committed to developing plans for sustainability of their influenza surveillance program. The objective of this review is to document the development and enrichment of influenza programs and lessons learned from the first countries completing ten years of support.
Keywords
Surveillance network, Capacity building agreement, World Health Organization, Sustainability, Influenza
Introduction
The World Health Organization (WHO) identified the need for cooperation from its member states in detecting and responding to health risks to avoid or mitigate a public health event of international significance and worked with 194 countries globally to gain agreement on implementation of the International Health Regulations (2005) (IHR) in 2007 [1]. The IHR requires Member States to strengthen core surveillance and response capacities at the local, regional and national levels, as well as at designated international ports, airports and ground crossings. IHR (2005) contains several key themes including maintenance of established systems to ensure ongoing ability to respond to public health threats [2]. Member states recognize the essential nature of not only building capacity to address emergency issues but the value in building a sustainable system that will aid in identifying, tracking, and addressing emerging infectious diseases.
In 2004, ongoing outbreaks of avian influenza A (H5N1) in domestic and wild bird populations and sporadic associated human cases in Asia heightened the concern that an influenza pandemic might emerge. These events prompted the U.S. Centers for Disease Control and Prevention to develop an international program to improve global pandemic preparedness and enhance capacity for laboratory and epidemiologic surveillance of influenza. Specifically, capacity in this context is defined as the ability to track and monitor disease out break and geographical spread within a country or region. Increased capacity equates to increased identification of and response to disease outbreaks. CDC approached their efforts from the perspective of "country-ownership" and focused on building capacity within the country's Ministries of Health (MOH) to handle disease monitoring and reporting. The program has 3 five-year phases, "capacity building" to assist countries in developing core functions, "sustainability" designed to plan for their systems in the absence of external funding and a "maintenance" phase with minimal ongoing funding. All three phases include technical assistance. During inception a set of principles was used to guide program development. Core to these principles was country ownership and use of National MOH staff and systems to build surveillance and create efforts that would be sustainable in the long term. A second core principle was to build upon the WHO Global Influenza Surveillance and Response System (GISRS) with use of regional and global WHO guidelines and recommendations. Support was provided to the first nine countries through a cooperative agreement mechanism in 2004 with 7 of the countries continuing their participation through 2014.
We have described in detail capacity gains for influenza laboratory and surveillance systems in 35 of the 39 countries we supported between 2004 and 2013 [3]. We found dramatic improvement in the quality assurance of RT-PCR diagnostics, participation in vaccine strain selection, and reporting of data to GISRS FluNet. Thirty-two (94%) countries established > 3 surveillance sites while in the program, and 28 (80%) countries added additional pathogens to the routine platforms developed or enhanced through capacity strengthening. Overall, 34 (97%) countries reported that they were able to meet their needs through this program. In this paper, we seek to describe our program and lessons learned from the first seven countries completing ten years in our program of technical and financial support. Our approach is to review and describe the program, funding opportunities and assessment and monitoring tools. In addition, we conducted phone interviews using standard questions with the seven grantees to record their thoughts, successes, and plans for the future. We use this review and feedback as the basis to develop lessons learned from the first ten years of our capacity building program for both internal improvement and sharing with other organizations.
Funding opportunity announcements
Funding was made available in 2004, with a published Funding Opportunity Announcement (FOA), "Development of Influenza Surveillance Networks". The purpose of this capacity building FOA was to establish or enhance an active influenza surveillance network that uses standardized data collection instruments, operational definitions, and laboratory diagnostic tests to enhance surveillance for influenza at three or more hospital or clinical sites within the country [4]. Countries were required to have a designated WHO National Influenza Center (NIC) laboratory and preference was given to countries with identified human cases of H5N1 or countries in geographic proximity to H5N1outbreaks or cases of human infection [5]. Nine countries China, India, Indonesia, Malaysia, Mongolia, Pakistan, Philippines, Republic of South Korea, and Thailand received funding. In 2004, the capability levels to conduct and support influenza surveillance varied widely, though each country had a WHO recognized NIC. The cooperative agreement period was for five years with funding renewable on a yearly basis. Median funding over the entire five year capacity building cycle was $400,000 with a yearly median range from $220,000 to $700,000. Eight of nine countries completed the initial five-year capacity building phase and had an opportunity to apply for a second five-year cooperative agreement titled "Developing Sustainable Influenza Surveillance Networks". Malaysia opted not to complete the first cycle and ended their agreement early. The second phase built on capacity established and/or gained during the first five years and provided support for the countries as they planned for sustainability and country ownership for ongoing program support. Seven of the eight eligible countries applied and in the initial year received median award of $554,831. The emergence of A (H1N1) in mid-2009 resulted in increased funding during the first year of the sustainability phase however, funding decreased annually over the next five years. Under this phase, countries continued to enhance their systems while they developed detailed plans to provide full financial support for timely surveillance of novel, pandemic and seasonal influenza and to ensure sustainable systems [6].
Capacity-Building Approach
Regional management and technical support
A key component of the program and a core principle was provision of technical assistance to countries. CDC carried out technical support through dedicated follow-up from CDC staff and enhanced collaboration with WHO regional offices. CDC assigned a project officer for each of the six WHO Regions; this person was responsible for coordinating technical and financial assistance to program grantees and working with both the country and WHO to implement strategies to accomplish country objectives [7]. In addition, China, Thailand, India, Philippines, and Indonesia had a CDC Influenza staff epidemiologist placed in-country to work collaboratively with Ministries of Health and WHO. Financial and technical support varied by country based on needs and included activities to monitor influenza activity, create an evidence base for decision making and evaluate development of policies for prevention and control. Collaboration between CDC and regional WHO offices was important to ensure a unified approach to improving influenza surveillance in the countries.
Capacity assessments and monitoring tools
Capacity assessments in conjunction with other technical collaborators, such as Association of Public Health Laboratories (APHL) and Council for State and Territorial Epidemiologists (CSTE), were central to establishing a baseline level of capacity, identifying training needs and monitoring progress. CDC developed two tools to document performance levels and training needs for surveillance and laboratory as well as a third tool to assess the ability of countries to respond to pandemics [8]. All assessments were voluntary, facilitated by CDC personnel and conducted in collaboration with the countries.
National inventory of core capabilities for pandemic influenza preparedness and response
The first tool, the National Inventory of Core Capabilities for Pandemic Influenza Preparedness and Response provided a format to document and assess preparedness capabilities over time [9]. Taking two years to fully develop, pilot and implement, the tool enabled collection of data in 12 critical capabilities, each with four proxy indicators, for responding to a pandemic. Capabilities included topics such as country planning, epidemiology, laboratory, routine surveillance and infection control. Proxy indicators looked at more details. For example, laboratory indicators included information about the laboratory network, bio-safety levels and routine processing of specimens, methods and participation in the WHO GISRS. A description of the development of the tool and results is published [5,9]. Capability indicators were scored on a scale of 0, indicating little or no capability, to a 3, indicating competency for the indicator. These facilitated assessments helped guide the countries and Project Officers in identifying technical assistance needs and supporting activities or developing plans to address gaps. The data showed that countries made progress in building capacity and maintained that capacity year over year (Figure 1).
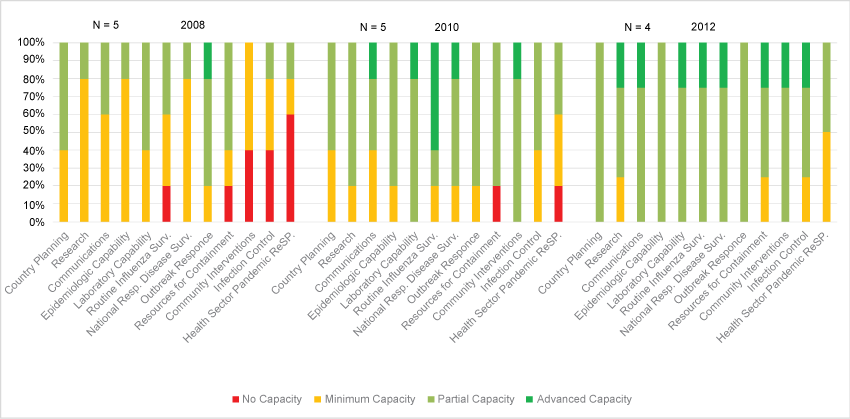
.
Figure 1: Aggregate country scores for pandemic preparedness and response, 2008, 2010 and 2012.
View Figure 1
International influenza laboratory capacity review tool (lab tool)
CDC, in conjunction with the APHL, developed and implemented the International Influenza Laboratory Capacity Review Tool (Lab Tool) for use beginning in 2009 [8]. This assessment tool documents laboratory system strengths and challenges. Sections of the tool include general laboratory information, virology, laboratory management and bio-safety indicators and is published and downloadable at https://www.cdc.gov/flu/international/tools.htm. During 2010, five of the countries requested and completed assessments. Laboratory strengths and specific recommendations for improvement were shared with laboratory personnel in each respective country. Four of the countries requested repeat assessments which were conducted in 2013 and 2014. Initial assessor recommendations ranged from suggestions to improve quality control procedures, suggestions for equipment purchases to recommendations for training to improve staff skills. During the second assessments, initial recommendations were reviewed with a combined 93% completion rate. The development of the tool and impact of the assessments has been previously published and showed the assessments substantially helped countries complete recommendations for improvement [10].
Surveillance assessment and review tool (surveillance tool)
The Surveillance Assessment and Review Tool (Surveillance Tool) [8] was developed in 2010 to assess the strengths and weaknesses of a country's influenza surveillance system. The tool reviews national level surveillance as well as sentinel surveillance sites and provides an overall picture of the functioning and management of the surveillance system. Questions range from inquiries about case report forms to questions on how surveillance data is collected and stored both at surveillance sites and on an aggregate national level. Assessors visit surveillance sites and discuss process and procedures with site staff. Results including recommendations for improvements to data collection and management were documented. Of the nine countries in the first cohort, five requested surveillance reviews and were provided summary reports.
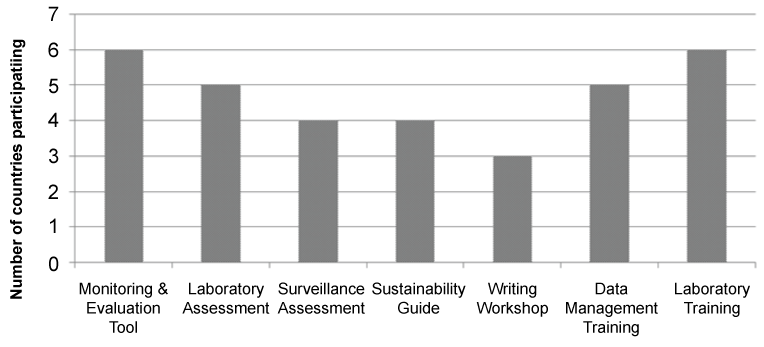
The use of these assessment tools helped identify three key training needs within the countries assessed. To meet these needs, courses in data management for influenza surveillance, laboratory management, and writing workshops were offered at a regional level with country representation based on need. The data management trainings provided attendees with hands on practice and technical assistance in building data management systems for their surveillance data. Similarly, the laboratory trainings were conducted to provide specific technical, hands on training in laboratory techniques and laboratory management. The writing workshops provided assistance on data analysis and writing skills to facilitate publication. These trainings enhanced technical skills and information sharing. Overall satisfaction with the trainings, were confirmed via participant end of course evaluations including both quantitative and qualitative sections.
Participation in assessments and trainings was voluntary and countries were able to choose participation based on their needs, priorities and schedules. Figure 2 represents the participation level of the countries in the technical training and assessments offered. Overall, participation in the technical assistance offered was high.
International influenza program sustainability guide and framework (guide)
An International Influenza Program Sustainability Guide and Framework (Guide) to assist grantees with the development of their sustainability plans was developed by CDC. The Guide provides a framework for sustainability planning and implementation and consists of six elements: Program Capacity, Strategic Planning, Partnerships, Funding, Communications, and Program Evaluation. The Guide was shared with countries when they were awarded sustainability cooperative agreements.
Of the seven countries completing the sustainability phase, four developed sustainability plans and/or held national meetings with key stakeholders to discuss program sustainability, both technical and financial. The actions most commonly identified to foster sustainability included plans to reduce staff, reduce or expand the number of surveillance sites, publish data from past years to influence future policy decisions, and integrate the influenza surveillance system into another respiratory or public health program. All countries have active influenza surveillance systems with all seven reporting results to WHO FluNet for the 2015 season [11].
Results and Lessons Learned
Capacity building phase
Determine baseline status by conducting capacity assessments early: During the capacity building phase, most countries focused on hiring personnel, improving laboratory testing and establishing sentinel sites to collect surveillance data. As CDC funded a larger group of countries in 2006 (24 countries) the need for assessment tools to document status and determine needs became very clear. Several of the tools developed were not ready until the originally funded countries were in their fourth or fifth year.
Because the tools were developed and implemented four to six years into the program, a baseline assessment was not conducted prior to CDC assistance and therefore progress for the 2004 group is harder to measure and document. A first critical lesson learned was that conducting these assessments in the first years of the capacity building phase provides a framework for future work and allows countries to build yearly objectives and effective work plans. Project Officers now offer assessments to countries during the initial years of the grant cycle to aid in guiding investments and technical assistance. The assessment tool data identifies areas needing improvement not only for individual countries but when in aggregate, across regions. The circulation of H5N1 necessitated a rapid start to our program. In hindsight, work on the development of assessment tools should have commenced concurrently with the start of the program to facilitate understanding of baseline needs and to document status.
Discuss sustainability earlier: A second lesson learned was that discussions about program sustainability need to begin during the initial capacity building phase. Five countries noted that earlier discussions would have provided more time to plan for sustainability of a surveillance network that meets country objectives and goals and to continue with minimal financial support. If sustainability is a program goal, organizations should begin to discuss sustainability from inception and to identify stakeholders who can work as a team to create a sustainability plan as early as possible in the funding cycle. Initial discussions about sustainability provide several advantages including developing and maintaining relationships with potential program champions, identifying and fostering additional partnership opportunities, and ensuring the proposed funding plan adapts to trends and unanticipated funding shortfalls.
Standardizing a five-year review: A five-year review was required of each country as they entered the sustainability phase. No format was provided and the review was meant to function as a self-evaluation of program goals, successes, achievements, areas of improvement and strategy moving forward. Outcomes recommended included a summary of the system, successes and challenges to date, and lessons learned to form the basis for making sure their system met country goals and was positioned to "right-size" for sustainability.
Although each country conducted a five-year review, the actual format differed from country to country with some preparing and presenting a verbal presentation, some writing an overall summary report, and some including the information in a presentation format with slides. The countries documented and discussed successes that included establishing laboratory and epidemiologic surveillance, recruiting staff, and electronic reporting. Identified obstacles include political challenges, minimal publications, and underperforming sites. A third lesson learned was the need for formalizing this review process and detailing the desired review objectives and outcomes as well as increasing documentation of the process.
Publishing surveillance results: Countries applying for sustainability funding were required to publish and share surveillance results from the first five-year cooperative agreement. Eight countries published surveillance results after their first five-year agreement although only seven continued with the second phase of funding [12-19]. Countries were encouraged to publish surveillance results to increase knowledge and understanding of influenza locally and globally. In addition to publications, countries were encouraged to present their findings at international and national conferences and meetings.
As countries began to develop abstracts and publications, the need for data management and training on how to outline and write a scientific publication was identified. The writing workshop and the data management workshop were developed to address these needs and improve a skills gap. As a program, the lesson learned was the need to better document all of these activities, with special focus on any activities that were required as part of the cooperative agreement. Although activities were completed, a formal documentation process did not exist.
Sustainability phase
A questionnaire was developed and phone interviews were conducted with grantees to document their thoughts, successes, and plans for the future (Appendix). Three key issues were identified from these interviews regarding sustainability; 1) the need for government support, 2) the need for skilled, trained staff to maintain the sentinel site system, and 3) the need to discuss sustainability and right-sizing earlier in the grant cycle, preferably at inception.
Based on responses, six of the seven countries completing the entire ten year project phase indicate they had built a sustainable surveillance system. Due to political issues at the central government level, the remaining country felt that the surveillance work would be transitioned to the provincial level which puts sustainability in question. Four countries indicated that the laboratory system would be the easiest part of the system to maintain with the WHO designated NIC as the focal point. Government support was identified as pivotal to building and maintaining a long lasting system with agreement that building relationships with other organizations should start during the capacity building phase.
All countries from the first cohort indicated the importance of skilled, trained and committed staff to the maintenance of a long-term sentinel site system. The ability to maintain trained staff to continue the work was a common concern among all the countries. Stable Ministry of Health (MOH) funding to maintain staff salaries and supplies was also a concern. This common concern also highlights the importance of countries starting or maintaining Field Epidemiology and Laboratory Training Programs (FELTP) as a source of trained staff within countries [20].
Reliable, actionable data are a key critical outcome of an effective surveillance system. Countries were prompted to focus on establishing quality surveillance sites rather than simply establishing numerous surveillance sites, especially if the country was unable to support them. Collecting quality data enabled countries to develop burden of disease estimates, strengthen their surveillance systems, and guide influenza prevention strategies. The lesson learned was that maintaining fewer sites with quality data was more conducive to long-term sustainability.
Right-sizing is the concept of establishing the optimum amount of sentinel sites to provide adequate influenza surveillance. This is a concept that five out of the seven countries indicated they would have liked to discuss and incorporate into their plans from the beginning of the capacity building grant. The remaining two countries used the concept as they identified and built their systems. The ability to build a system based on the concept of rightsizing allows the countries to use funds more effectively and develop a more efficient, sustainable system. Global guidance on "right-size" strategies similar to U.S. efforts would be beneficial to countries trying to make sure their surveillance systems meet minimal requirements for National and Global contributions to influenza surveillance [21].
Conclusion
During the project timeframe, many global entities, including CDC, were conducting technical support for H5N1 and supporting improvements to the WHO Global Influenza Surveillance and Response System (GISRS). It is clear that collective global efforts have supported influenza surveillance improvements. After ten years of support, all countries established and/or enhanced laboratory and epidemiologic surveillance for influenza. Systems and skills gained and maintained by National MOHs were a positive contribution to capacity building efforts and were important for the detection of emerging infectious diseases such as influenza. Addressing sustainability and defining long-term success (> 10 years) for public health programs is challenging. Discussions about program sustainability led by the MOH are critical as MOHs are responsible for maintaining systems that meet the public health priorities of their governments and comply with IHR. Lessons learned from review of the support for this 2004 initial group of grantees have served to improve our support for subsequent groups of grantees as they enter the capacity building, sustainability and maintenance phases. These lessons also may be valuable for others embarking on establishing capacity building programs for global health security.
Acknowledgments
We are grateful to all country representatives and others who planned and participated in both assessments and trainings. Without them, program goals could not have been accomplished.
Disclaimer
The opinions expressed by authors contributing to this journal do not necessarily reflect the opinions of the Centers for Disease Control and Prevention or the institutions with which the authors are affiliated.
References
-
World Health Organization: International Health Regulations (2005) Frequently asked questions about the International Health Regulations.
-
World Health Organization (2005) International Health Regulations. (2nd edn).
-
Polansky LS, Outin-Blenman S, Moen AC (2016) Improved Global Capacity for Influenza Surveillance. Emerg Infect Dis 22: 993-1001.
-
Centers for Disease Control and Prevention (2004) Development of Influenza Surveillance Networks.
-
Moen A, Kennedy PJ, Cheng PY, MacDonald G (2014) National Inventory of Core Capabilities for Pandemic Influenza Preparedness and Response: results from 36 countries with reviews in 2008 and 2010. Influenza Other Respir Viruses 8: 201-208.
-
Centers for Disease Control and Prevention (2009) Developing Sustainable Influenza Surveillance Networks and Response to Avian and Pandemic Influenza by National Health Authorities outside the United States.
-
(2013) USA Jobs. Public Health Advisor.
-
Centers for Disease Control and Prevention (2013) Evaluation and Capacity Review Tools.
-
MacDonald G, Moen AC, St Louis ME (2014) The national inventory of core capabilities for pandemic influenza preparedness and response: an instrument for planning and evaluation. Influenza Other Respir Viruses 8: 189-193.
-
Lucinda E A Johnson, Sarah A Muir-Paulik, Pam Kennedy, Steven Lindstrom, Amanda Balish, et al. (2015) Capacity building in national influenza laboratories-use of laboratory assessments to drive progress. BMC Infectious Disease 15: 501.
-
World Health Organization (2017) Influenza.
-
Chadha MS, Broor S, Gunasekaran P, Potdar VA, Krishnan A, et al. (2012) Multi site Virological Influenza Surveillance in India: 2004-2008. Influenza Other Respir Viruses 6: 196-203.
-
Kosasih H, Roselinda, Nurhayati, Klimov A, Xiyan X, et al. (2013) Surveillance of Influenza in Indonesia, 2003-2007. Influenza Other Respir Viruses 7: 312-320.
-
Burmaa A, Kamigaki T, Darmaa B, Nymadawa P, Oshitani H (2014) Epidemiology and impact of influenza in Mongolia, 2007-2012. Influenza Other Respir Viruses 8: 530-537.
-
Badar N, BashirAamir U, Mehmood MR, Nisar N, Alam MM, et al. (2013) Influenza Virus Surveillance in Pakistan during 2008-2011. PLoS ONE 8: e79959.
-
Chittaganpitch M, Supawat K, Olsen SJ, Waicharoen S, Patthamadilok S, et al. (2012) Influenza viruses in Thailand: 7 years of sentinel surveillance data, 2004-2010. Influenza Other Respir Viruses 6: 276-283.
-
Lee JS, Shin KC, Na BK, Lee JY, Kang C, et al. (2007) Influenza surveillance in Korea: Establishment and first results of an epidemiological and virological surveillance scheme. Epidemiol Infect 135: 1117-1123.
-
Yang P, Duan W, Lv M, Shi W, Peng X, et al. (2009) Review of an influenza surveillance system, Beijing, People’s Republic of Chica. Emerg Infect Dis 15: 1603-1608.
-
Furuse Y, Suzuki A, Kishi M, Galang HO, Lupisan SP, et al. (2010) Detection of novel respiratory viruses from influenza-like illness in the Philippines. J Med Virol 82: 1071-1074.
-
Nguku P, Oyemakinde A, Sabitu K, Olayinka A, Ajayi I, et al. (2014) Training and Service in Public Health, Nigeria Field Epidemiology and Laboratory Training, 2008-2014. Pan Afr Med J 18: 2.
-
(2013) Association of Public Health Laboratories. Influenza Virologic Surveillance Right-Size Roadmap.