Journal of Infectious Diseases and Epidemiology
Preliminary Studies of Edible Bird Nest (EBN) Extract Reduced H1N1 Virus Induced Apoptosis on Cultured Cells-In Vitro
Nur Ain A1*, Abdul Rahman O1,2 and Aini I1,2
1Department of Microbiology and Micology, Universiti Putra Malaysia, Malaysia
2Department of Veterinary Medicine, Universiti Putra Malaysia, Malaysia
*Corresponding author: Nur Ain A, PhD, Department of Microbiology and Micology, Universiti Putra Malaysia, 43400 UPM, Serdang, Malaysia, E-mail: ainamin2013@gmail.com
J Infect Dis Epidemiol, JIDE-3-024, (Volume 3, Issue 1), Original Research; ISSN: 2474-3658
Received: October 21, 2016 | Accepted: February 25, 2017 | Published: February 28, 2017
Citation: Nur AA, Abdul RO, Aini I (2017) Preliminary Studies of Edible Bird Nest (EBN) Extract Reduced H1N1 Virus Induced Apoptosis on Cultured Cells-In Vitro. J Infect Dis Epidemiol 3:024. 10.23937/2474-3658/1510024
Copyright: © 2017 Nur AA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The virus infection result in apoptosis of cells included mechanisms to inhibit apoptosis. It seems that both cellular and viral factors are involved depending on types of cells. Edible bird nest (EBN) is a well-known traditional Chinese medicine that contained various bioactive compounds with health benefits in human. However, not much of scientific investigation has been conducted to confirm apoptotic compound in EBN. Hence, this study to investigate the ability of EBN in reducing apoptosis. EBN was first prepared into water extract, based on the established method, without enzymatic treatment. Cytotoxicity of the EBN extract then was tested by MTT assay on MDCK cells. Later, the influenza A virus (strain A/Puerto Rico/8/1934 H1N1)-infected cells were treated with CC50 of EBN extract. This is followed by using Annexin V-propidium iodide flow cytometry for apoptotic event analysis. Results showed that EBN extracts significantly inhibited cells undergoing through late apoptosis (9.84%) and necrosis (0.94%) compared to infected cells, respectively 31.16% (late) and 16.41% (necrosis) with (p < 0.05). In conclusion, EBN inhibits cells that undergo apoptosis following influenza A virus infection. Further study is required to define the mechanisms of EBN as an anti-apoptotic agent in the context of influenza virus infected cells.
Keywords
Influenza A virus, Viral infection, Apoptosis, EBN extract, Flow cytometry
Introduction
Apoptosis is an important physiological necessary for development and maintenance of tissues homeostasis including tissue atrophy, the immune system development and biological tumor [1,2]. It also plays an important function in the pathogenesis of many infectious diseases including those caused by viruses [3-5]. Many virus infections result in apoptosis of host cells and several viruses have evolved mechanisms to inhibit apoptosis. Influenza viruses induce apoptosis through mechanism then both cellular and viral factors depending on the cell type. However, the precise mechanisms still remain unclear.
Nowadays, Edible bird nest (EBN) has turned into an important in Malaysia as a natural food product. In previous study, it was found that vascular growth factor and melatonin are found in EBN and are believed to provide antioxidant and anti-apoptotic properties [6,7]. As apoptosis have been suggested as pivotal events in Influenza virus infection, EBN, the salivary secretion of swiftlets, may have anti-apoptosis relevance in the therapeutic context of viral infection. Hence, this study aimed to investigate the anti-apoptosis effect of EBN.
Materials and Methods
Preparation of EBN extracts
Raw EBN from the swiftlet of Aerodramus genus collected from bird's nest house in Teluk Intan, Perak, Malaysia. The cleaning process was carried out by first soaking after the unprocessed EBN in distilled water until softened. Debris and feathers were removed after which the EBN was subjected to drying process at 70 °C for 16 hrs and followed by grounding and sifting through a mesh (60 μM in pore size). The grounded samples were kept in distilled water at 3 °C in about 16 hours and continued heated for 30-60 min at 60 °C. The extract was then filtered and freeze-dried after 48 hours freezing at -80 °C, and stored for the future use.
Madin darby canine kidney (mdck) cell culture
MDCK cell line was purchased from the American type culture Collection (ATCC, CCL-34™) and grown in Dulbecco's Modified Eagle's Medium (Gibco, UK) supplemented with 10% fetal bovine serum (Gibco,UK), antibiotic-antimycotic (Gibco,UK). The cells then were seeded into a sterile 96-well flat bottom plate (Nunc, USA) and maintained at 37 °C humidified incubator with 5% CO2 (Galaxy,UK) for 2-3 days until 70%-80% confluency is achieved.
Virus propagation
Influenza A virus, strain A/Puerto Rico/1934 (H1N1) was purchased from the American Type Culture Collection (ATCC,® VR-95™) and propagated in MDCK cells, then the stock virus was titrated by TCID50. In obtained 100 TCID50, the resultant virus titer was further diluted with 100 °l taken from the amount of TCID50 then it was to be used as a constant positive control in the following procedure.
Cell viability based on combination treatment
This procedure was performed by using Maximal Non-Cytotoxic Concentration of EBN and constant virus titer (100 TCID50). Briefly, the virus was first inoculated into the MDCK cells and later followed by the EBN extract replacement for 1 hrs followed by change with media. All the samples were incubated at 37 °C in 5% CO2 incubator.
After 48 hrs of post-treatment incubation, the plates were exposed to the MTT assay. Treated cells were subjected to MTT reagent (Sigma Aldrich, USA) in which reacts to quantify the viable cells. Pure DMSO solution (Merck, Germany) was added to the cells after 2-3 hr exposure to the MTT reagent to solubilize the formazan precipitation. Absorbance of the solution was measured spectrophotometrically by using ELISA reader at 540 nm wavelength (Bio Tek Instruments EL800, USA).
Apoptotic cells analysis
The protocol of Annexin V-FITC Apoptosis Detection Kit (BD Pharmingen, USA) by Rieger, et al. (2011) was performed in this procedure. Briefly, upon completion of treatments, cells were harvested and washed with binding buffer. Then, Annexin V and propidium iodide (PI) were added and incubated in dark for 15 min. Subsequently, washing was performed twice with binding buffer which then incubated for 15 min at 37 °C. Finally, the samples were reads for analysis with Becton Dickson FacsCalibur Flow Cytometer (BD Biosciences, USA).
Statistical analysis
Data was collected as triplicate for each experiment. The results were expressed as mean ± standard deviation. Statistical significance was assessed with Anova test and P value < 0.05 was considered significant.
Result and Discussion
The determination of toxicity of Edible Bird Nest (EBN) extract
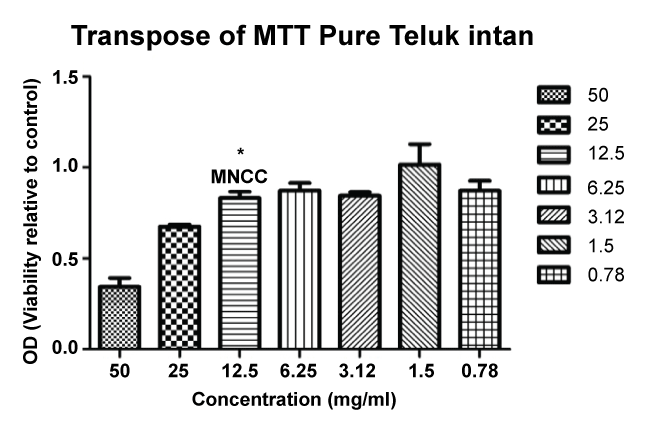
Toxicity study was performed with addition of EBN extract to determine the concentration wide effect as well as CC50 of the extract on MDCK culture. CC50 is the 50% of cytotoxic concentration that can lead to the death of 50% of the host cells. In order to determine the effect of EBN treatment at lower concentration was determined by MNCC, which is the maximal dose for cell toxicity with no cytotoxic effect.
The obtained values were transformed as a ratio of the control. The linear regression analysis then was used to calculate the concentration of EBN contributed to the cell cytotoxicity with p < 0.05. Then, followed by Tukey HSD post hoc analysis which showed the first concentration of EBN without significant difference in contrast to negative control considered as MNCC with the obtained value (12.5 mg/ml) as figure 1. The MNCC concentration was used in the following procedure for cell treatment.
.
Figure 1: The MNCC value for EBN concentration. Asterisk (*) show the first non-significant difference of the cytotoxicity with (p < 0.05) of the negative control (PR8).
View Figure 1
Based on the result, it is likely similar with the previous study [8], which showed that crude EBN extract is more cytotoxic compared to the water soluble substances.
The determination on titrations of virus particles on infected culture cell
Virus titrations are commonly using hemagglutination assay (HA) in order to quantify the concentration of infectious and non-infectious virions through binding of erythrocytes [9]. Then, either by plaque assay [10], or as tissue culture infectious dose (TCID50) is commonly used to determine the concentration of infectious virus [9].
In this study, the daily virus titration upon the stage of virus propagation is shown in table 1. The onset of virus production was detected to increase over the day. The result showed that the virus titer started to increase after the propagation. Prior to the result, the virus showed the highest titer as 256 (28) in day 2 and 3.
![]()
Table 1: Virus propagation in MDCK cells.
View Table 1
Subsequently, at day 3, the supernatant of the propagated viruses were harvested and used for determination of TCID50. The determination of TCID50 based on HA result is revealed as table 2. The Karber formula [11] then is used to calculate TCID50 of the virus according to the result.
![]()
Table 2: TCID50 based on HA.
View Table 2
According to the result, TCID50/ml of the harvested virus was around 103. At dilution of 103 of the virus stock, 100 TCID50/ml which is equal to 0.01-0.5 MOI was used in the following procedures.
Overall, the titrations of virus particles from culture supernatant depend on how much of infectious cells are able to release their virus particles. The comparatively high viral titre will reflect the high infectious infectivity of virus (TCID50). This could be concluded that the increase of virus titre as well as virus infectivity.
EBN extracts improves cell viability in H1N1 virus-challenged MDCK cells
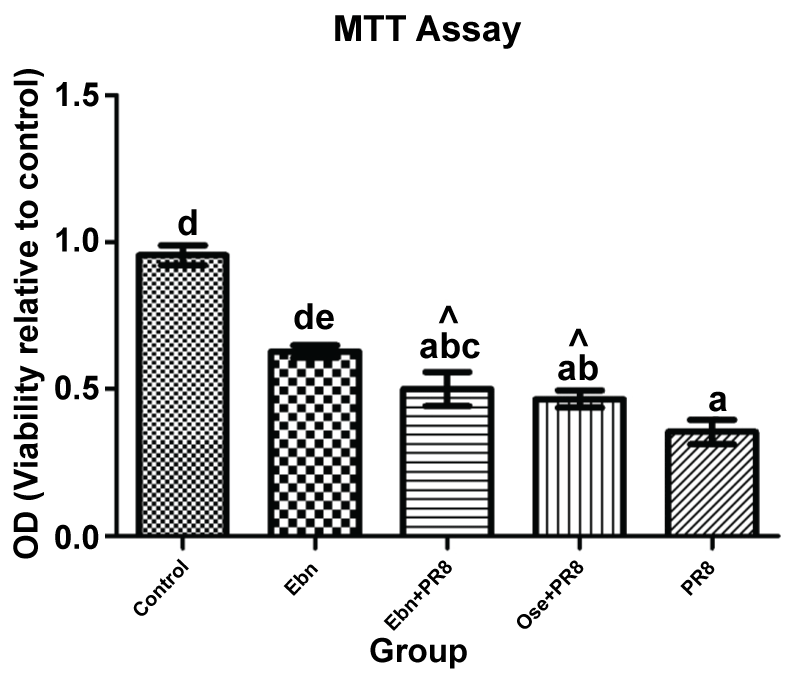
MTT assay was carried out in order to quantify the cell viability from those observed in the infected MDCK cells treated with EBN extract. Based on the observation, in general, all treatment groups seem did not show significantly in cell improvement compared to control, whilst upon challenge with IAV infection (PR8), show the increasing of cell viability after 48 hour of post-treatment. The data for cell viabilities in combination treatment is shown in figure 2.
.
Figure 2: Effect of combination treatment on strain A/Puerto Rico/8/1934 (H1N1)-challeged MDCK cells cell viability in 48 hour. Cell viability was assessed with MTT assay and data shown are means ± SD by Tukey HSD pairwase comparison between groups (p < 0.05). *abcde Values with different superscript differ significantly difference with infected group (PR8) (p < 0.05) indicated by caret (^).
View Figure 2
Based on the result, both treatment groups, EBN and Oseltamivir showed significant increased cell viabilities compared to infected group (PR8). However, EBN treated MDCK challeged virus (EBN+PR8) is more produced the cell viability compared to Oseltamivir (OSE+PR8). Thus, the inreasing of cell viability in EBN treated infected cells could be related to previous study done by [12], showed that EBN able to promote cell division in rabbit corneal keratocytes and acid hydrolysates of EBN has been shown in making proliferation of human colonic adenocarcinoma (CaCO-2) cells [13]. As comparison, in previous study done by Hinshaw, et al. (1994) shown the cytotoxicity assay has a lower level of cell viabilities in MDCK cells at 48 to 72 hour after infected with Influenza A and B.
EBN extracts reduce late apoptotic event in HINI virus challenged MDCK cells
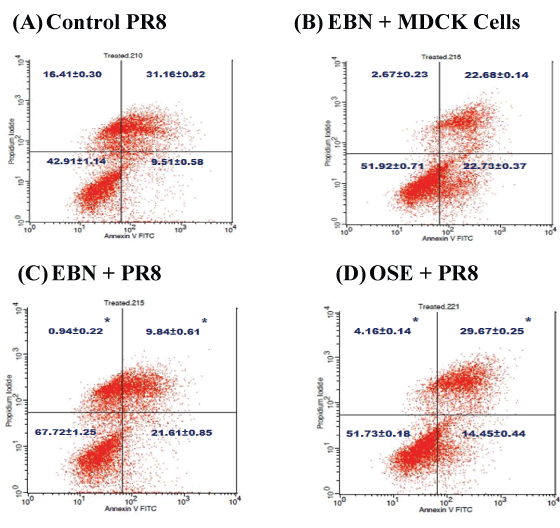
Apoptotic event was investigated by Annexin V-PI double staining method to identify the mode of cell death. Late apoptosis, represented by cell stained negatively with Annexin V due to phosphatidylserine translocation towards outer membrane surface followed by loss of membrane integrity, was found to be the major cell death mechanism in infected cell with H1N1 virus for 48 hours (Figure 3). It accounted for about 31% of the cell population which is in concordance with other reports [14,15].
.
Figure 3: A) Assessment of cell death using a Flow Cytometry analysis; B) The apoptosis detection on MDCK Cells with and without EBN treatment for Virus A/Puerto Rico/8/1934 (H1N1); C) Cell treated for 48 hour was analyzed by Annexin V-propodium iodide double staining, (D) Representative plots of propodium iodide versus Annexin V-FITC fluorescence signals are visualized (A-D). The data represents the percentage mean ± standard deviation of three independent experiments (treatment groups) carried in triplicate. (*) show a significant difference with the values of p < 0.05 compared to control.
View Figure 3
Generally, EBN treatment cell alone did not reduced apoptotic event (Figure 3B) but it effectively reduced late apoptotic injury in cells challeged with H1N1 virus (Figure 3C) (9.84%) compared to infected group (Figure 3A) with 31.16% of cell death population. Meanwhile, the commercial drug treatment (Figure 3D) also show reduced the apoptotic event but slightly in cell death population (29.67%) compared to EBN. The results suggest that EBN is a potential anti-apoptotic agent, which act by inhibiting apoptosis since previous finding stated that the efficiency of virus growth to induce apoptosis was active at the late stage infection or apoptosis occur [16].
Conclusion
In conclusion, this study has been successfully demonstrated that EBN extract confer apoptosis reduction in infected cells by influenza A viruses. Particularly, EBN demonstrated apoptosis reduction by ameliorate cell viability and attenuate of cell death population.
Acknowledgement
Our sincere gratitude to Dr Fadzillah A'ini, from Department of Veterinary Services, Putrajaya, Malaysia, who kindly provided EBN sample. This project was supported by Coe Swiftlet grants no.6371400 from Ministry of Agriculture and Agro-Based Industry, Putrajaya, Malaysia.
References
-
Jacobson MD, M Weil, MC Raff (1997) Programmed cell death in animal development. Cell 88: 347-354.
-
Young LS, CW Dawson, AG Eliopoulos (1997) Viruses and apoptosis. Br Med Bull 53: 509-521.
-
Razvi ES, RM Welsh (1995) Apoptosis in viral infections. Adv Virus Res 45: 1-60.
-
Ludwig S, S Pleschka, T Wolff (1999) A fatal relationship influenza virus interactions with the host cell. Viral Immunol 12: 175-196.
-
Roulston A, RC Marcellus, PE Branton (1999) Viruses and apoptosis. Annu Rev Microbiol 53: 577-628.
-
Mehraein F, Talebi R, Jameie B, Joghataie MT, Madjd Z (2011) Neuroprotective effect of exogenous melatonin on dopamenergic neurons of the substantia nigra in ovariectomized rats. Iran Biomed J 15: 44-50.
-
Falk T, Yue X, Zhang S, McCourt AD, Yee BJ, et al. (2011) Vascular endothelial growth factor-B is neuroporotective in an vivo rat model of Parkinson's Disease. Neurosci Lett 496: 43-47.
-
Yew MY, Koh RY, Chye SM, Othman I, Ng KY (2014) Edible bird nest ameliorates oxidative stress-induced apoptosis in SH-SY5Y human neuroblastoma cells. BMC Compliment Altern Med 14: 391.
-
Mahy B, Kangro H (1996) Virology Methods Manual. Academic Press.
-
Huprikar J, Rabinowitz S (1980) A simplified plaque assay for influenza viruses in Madin-Darby Canine Kidney (MDCK) cells. J Virol Methods 1: 117-120.
-
Karber G (1931) 50% endpoint calculation. Arch Exp pathol pharmacol 162: 480-483.
-
Zainal Abidin F, Hui CK, Luan NS, Mohd Ramli ES, Hun LT, et al. (2011) Effect of edible bird's nest (EBN) on cultured rabbit corneal keratocytes. BMC Complement Altern Med 11: 94.
-
Aswir AR, Wan Nazaimoon WM (2011) Effect of edible bird's nest on cell proliferation and tumor necrosis factor-alpha (TNF-α) release in vitro. Int Food Res J 18: 1123-1127.
-
Ikeda Y, Tsuji S, Satoh A, Ishiku M, Shirasawa T, et al. (2008) Protective effects of astaxanthin on 6-hydroxydopamine-induced apoptosis in human neuroblastoma SH-SY5Y cells. J Neurochem 107: 1730-1740.
-
Wei L, Sun C, Lei M, Li G, Yi L, et al. (2013) Activation of Wnt/beta-catenin pathway by exogenous Wnt1 protects SH-SY5Y cells against 6-hydroxydopamine toxicity. J Mol Neurosci 49: 105-115.
-
Teodoro JG and Branton PE (1997) Regulation of apoptosis by viral gene products. J Virol 71: 1739-1746.





