International Journal of Respiratory and Pulmonary Medicine
Clinical and Radiological Predictor of Polymyositis and Dermatomyositis Associated Interstitial Lung Disease
Tomoo Kishaba1*, Hiroaki Nagano1, Yuichiro Nei1, Shin Yamashiro1 and Harumi Ito2
1Department of Respiratory Medicine, Okinawa Chubu Hospital, Japan
2Department of Radiology, Fukui University, Japan
*Corresponding author: Tomoo Kishaba, Department of Respiratory Medicine, Okinawa Chubu Hospital, 904-2293 Miyazato 281, Uruma, Okinawa, Japan, Tel: +81-98-973-4111, Fax: +81-98-974-5165, E-mail: kishabatomoo@gmail.com
Int J Respir Pulm Med, IJRPM-3-059, (Volume 3, Issue 4), Original Article; ISSN: 2378-3516
Received: August 19, 2016 | Accepted: September 29, 2016 | Published: October 03, 2016
Citation: Kishaba T, Nagano H, Nei Y, Yamashiro S, Ito H (2016) Clinical and Radiological Predictor of Polymyositis and Dermatomyositis Associated Interstitial Lung Disease. Int J Respir Pulm Med 3:059. 10.23937/2378-3516/1410059
Copyright: © 2016 Kishaba T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Polymyositis (PM) and dermatomyositis (DM) often have interstitial lung disease (ILD) and progression of ILD is important cause of death.
Objectives: This study is conducted to clarify the important clinical, radiological factors of PM/DM ILD patients.
Methods: We reviewed medical records, pulmonary function test (PFT), chest high-resolution computed tomography (HRCT) findings from January 1, 2001 to December 31, 2014 retrospectively at Okinawa Chubu Hospital.
Results: We identified 41 patients. Men were 14 and women were 27. Median age was 52 ± 16.3 (17-80). 19 was DM and 22 was PM. Compared to PM patients, DM patients more often had heliotrope rash, Gottron sign, and subungual erythema (47.4% vs. 4.5%, p = 0.0009, 68.4% vs. 9.1%, p < 0.0001, 52.6% vs. 9.1%, p = 0.0016). In terms of radiological findings, lung tip consolidation was more often detected in DM patients. In survival time, DM-ILD patients showed poor survival compared with that of PM patients (44.7 vs. 88.6 months: p = 0.0242). Cox proportional hazard ratio showed diagnosis of DM, Gottron sign, subungual erythema, Anti-MDA5 antibody, and lung tip consolidation were strong predictors of PM/DM-ILD patients [Hazard Ratio (HR): 6.343, p = 0.021, HR: 7.330, p = 0.019, HR: 8.784, p = 0.004, HR: 25.764, p = 0.001, HR: 2.494, p = 0.042].
Conclusions: In our cohort of PM/DM associated ILD patients, diagnosis of DM, Gottron sign, subungual erythema and lung tip consolidation were strong predictors.
Keywords
PM, DM, ILD, Gottron sign, Lung tip consolidation
Introduction
Dermatomyositis (DM) and polymyositis (PM) are two major idiopathic inflammatory myopathies (IIMs), which mainly affect skin, muscle, lung [1,2]. The estimated annual incidence of PM/DM ranges from 6 to 10 per 1 000 000 [3]. In general, PM associated interstitial lung disease (ILD) have rather chronic course [4,5]. And among the PM-ILD patients, autoantibodies against aminoacyl-tRNA synthetases (ARS) syndrome have good prognosis [6].
On the other hand, DM especially clinically amyopathic dermatomyositis (CADM) which have typical rash of DM with little or no definite muscle symptoms or hypomyopathic DM which mild muscle weakness with no elevation of muscle enzyme for > 6 months associated ILD patients have more poor prognosis [7,8]. Approximately one-third of the patients with DM, CADM, PM develop ILD during their clinical course [4,9]. In addition, acute severe forms of ILD sometimes occur in DM or CADM patients. Therefore, ILD is important extra-muscular manifestation of IIMs [10-12]. Regarding DM, CADM, there was some reports about poor prognosis of especially CADM associated ILD patients.
In addition, ILD causes substantial morbidity and resulting in mortality of approximately 50% in some series [13]. There are substantial heterogeneity exists within the spectrum of IIMs, from mild chronic course to fulminant rapidly progressive course [14]. Some reports have shown clinical characteristics of PM. DM associated ILD. However, little is known about both clinical and radiological findings of each phenotype, the aim of this retrospective study were evaluating clinical and radiological predictors of PM/DM patients.
Methods
Patient inclusion: We identified 41 patients diagnosed with PM (n = 22), DM (n = 15), and CADM (n = 4) associated with ILD patients from 2000 April to 2014 December at Okinawa Chubu Hospital. The diagnosis of PM/DM was based on the criteria of Bohan and Peter [1]. 1) systemic muscle weakness, 2) increased serum muscle enzyme levels, 3) electromyographic (EMG) evidence of myopathic changes, 4) typical histologic findings in muscle biopsies, and/or 5) characteristic dermatologic manifestations of DM. CADM, or dermatomyositissine myositis, is defined by the absence of clinically significant muscle symptoms and normal muscle enzymes such as creatine kinase (CK) level [7,12,15,16] for periods of > 6 months, and is associated with an acute severe form of interstitial lung disease (ILD) [9]. All clinical information including pulmonary function test, radiological findings were baseline at diagnosis.
Clinical information: We reviewed clinical symptoms such as fever, cough, dyspnea, myalgia, arthralgia, mechanic hand, rash, subungual erythema, erythema, heliotrope rash, Gottron sign, finger swelling and Raynaud phenomenon. In terms of laboratory findings, we evaluated WBC, CRP, CPK, GOT, GPT, ALP, γ-GTP, LDH, Sialylated carbohydrate antigen KL-6 (KL-6), surfactant protein-D (SP-D), ferritin. Auto-antibodies including Jo-1 [17] and anti-melanoma differentiation-associated gene 5 (MDA-5) antibody [18,19]. If patient underwent broncho-alveolar lavage (BAL), we checked cell population such as neutrophils, lymphocytes, eosinophils and CD4/8 ratio in broncho-alveolar lavage fluid (BALF). In physiology findings, we reviewed forced vital capacity (FVC), percent predicted FVC, forced expiratory volume in 1 second (FEV1), percent predicted FEV1, total lung capacity (TLC), percent predicted TLC, diffusing capacity or transfer factor of the lung for carbon monoxide (DLco) and percent predicted DLco.
Radiological findings: Chest high resolution computed tomography (HRCT) findings without contrast material were reviewed. These images comprised 1.5 mm collimation sections at 10 mm intervals. We evaluated the presence of consolidation, ground-glass opacity, reticular opacity, traction bronchiectasis and lung tip consolidation at below 1 cm of right diaphragm. Definition of ground-glass opacity was hazy increased attenuation of the lung that did not obscure the underlying vessels. Consolidation was defined as homogeneous increase in pulmonary parenchymal attenuation that obscured the underlying vessels. Definition of reticular opacity was regular interlacing linear shadows separated by a few millimeters. Traction bronchiectasis was defined as Irregular bronchial dilatation within or around areas with parenchymal abnormality. Definition of lung tip consolidation was thick consolidation > 3 mm with connection of both right diaphragm and pleura. Extent of consolidation, ground-glass opacity, reticular opacity, traction bronchiectasis was defined as follows; 0: none, 1: < 25%, 2: 25% < 50% >, 3: 50% < 75% >, 4: > 75% [20]. Extent of lung tip consolidation was defined as 0: none, 1: one thick consolidation, 2: more than two thick consolidations. Thick was defined over 3 mm. Lung tip consolidation looks like migratory bird. Therefore, we call it migratory bird sign. The score of each finding was defined as total sum divided into patient's number.
Our institutional board waived informed consent because of retrospective study.
Statistical Analysis
Continuous variables are presented as means ± standard deviations, and categorical variables are presented as percentages. The Chi-square and Fisher's exact tests were used to analyze categorical data, and the unpaired t-test and Mann-Whitney U test were used for continuous data. Cox regression analysis was used to identify significant variables predictive of mortality.
.
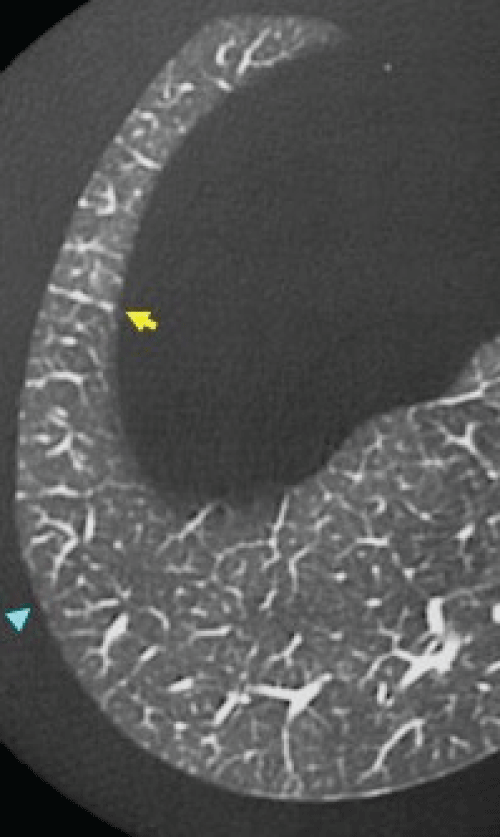
Figure 1 Representative image of Lung Tip consolidation.
Arrow indicates two thick consolidation and migratory birds sign.
View Figure 1
The Kaplan-Meier survival curves and the log-rank test were used to evaluate survival. The level of statistical significance was set at P < 0.05. All analyses were performed using Stata Data Analysis and Statistical Software STATA version 11.0; (Stata Corp., College Station, TX, USA).
.
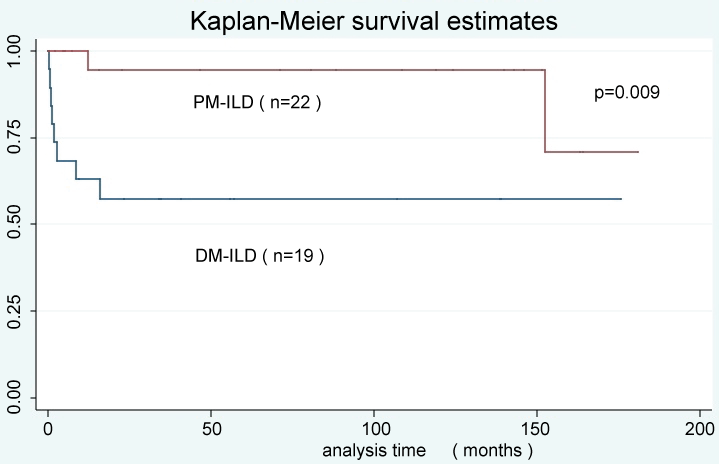
Figure 2 Survival curve of between PM and DM associated ILD patients.
DM-ILD patients showed poor prognosis compared to PM-ILD patients.
View Figure 2
![]()
Table 1: Clinical characteristics of combined PM/DM associated ILD patients.
View Table 1
Results
The clinical characteristics of PM/DM associated ILD patients (14 men, 27 women) are summarized in table 1. Mean age was 52 (17-80). Mean partial pressure of oxygen (PaO2) was 76.2 mmHg and partial pressure of carbon dioxide (PaCO2) was 31.3 mmHg. In BALF findings, mean neutrophils, lymphocytes, eosinophils and CD4/8 were 19.2% (0-81), 38.8% (4-93), 1.2% (0-4), and 1.2 (0.1-1.3) respectively. We compared clinical, physiological findings between PM-associated ILD and DM including CADM-associated ILD patients. PM-associated ILD patients tended to show more elevation of CPK, LDH and KL-6 (4544 vs 1956, 726 vs 517, 1812 vs 748) (Table 2). However, it did not show statistical significance. In autoantibody, four patients had positive for Anti-MDA-5 antibody and seven patients showed positive for Jo-1 antibody. When we evaluated survival time between PM-associated ILD patients and anti-MDA-5 negative DM-ILD patients, there was still statistically significance (88.6 months vs 52.9 months, p-value = 0.04). We also evaluated clinical symptoms and signs between PM and DM ILD patients. As shown in table 3, DM patients had more heliotrope rash (47.4% vs. 4.5%, p < 0.001), Gottron sign (68.4% vs. 9.1%, p < 0.001), subungual erythema (52.6% vs. 9.1%, p = 0.002), generalized rash (84.2% vs. 42.9%, p = 0.006).
![]()
Table 2: Clinical characteristics between PM and DM associated ILD patients.
View Table 2
In PFT, PM-ILD patients tended to show decreased %FVC, TLC and % DLco (71.4 vs. 78.3, 84.6 vs. 90.9, 65.6 vs. 94.8), however, it did not show statistically significance (Table 4). In terms of radiological findings, DM-associated ILD patients showed more lung tip consolidation than PM-ILD patients. (0.44 vs. 0.11, p = 0.025) (Table 5). Among five CADM-associated ILD patients, 80% of the patients had lug tip consolidation, on the other hand, 14% of the PM-ILD patients and 43% of the DM-associated ILD patients had lung tip consolidation. Representative image of lung tip consolidation is shown in figure 1. We call this finding migratory bird sign. Regarding treatment, 39 patients received high dose prednisolone initially. Especially, we did steroid pulse with immunosuppressants such as cyclosporine or tacrolimus for CADM associated ILD patients. In maintenance phase, PM-associated ILD patients more often took prednisolone alone (54.5% vs. 33.3%) and DM-ILD patients received more prednisolone plus tacrolimus (38.9% vs. 9.1%). Among the four anti-MDA-5 antibody positive patients, three patients (75%) died finally despite intensive therapy. Kaplan-Meier survival curve showed DM associated ILD group showed poor prognosis compared with that of PM related ILD group (88.6 months vs. 44.7 months, p = 0.009) (Figure 2). Overall, ten patients died during observation period. Cox proportional hazard model showed Gottron sign, subungual erythema, lung tip consolidation, diagnosis of DM and Anti-MDA-5 antibody[hazard ratio(HR); 7.33: p = 0.020, HR; 8.78: p = 0.004, HR; 2.494: p = 0.042, HR; 6.343: p = 0.021, HR; 25.76: p = 0.001, respectively]were strong predictors of PM/DM associated ILD patients at our cohort (Table 6).
![]()
Table 3: Clinical symptoms and signs of between PM and DM associated ILD patients.
View Table 3
![]()
Table 4: Pulmonary function test between PM and DM associated ILD patients.
View Table 4
Discussion
In this study, we clarified the clinical characteristics, physiological findings, radiological findings and prognosis of 41 PM/DM including CADM associated ILD patients at our cohort. Our result showed DM-specific signs such as Gottron sign and subungual erythema, anti-MDA-5 antibody were predictor of mortality. In addition, we also focused on radiological findings and lung tip consolidation or migratory bird sign was a useful predictor of mortality in DM associated ILD patients, which is a novel finding. There were a few studies which evaluated the prognostic factors for PM/DM ILD [5,8,16]. Therefore, our study contributes to meaningful information for evaluation of PM/DM associated ILD.
![]()
Table 5: Radiological findings between PM and DM associated ILD patients.
View Table 5
![]()
Table 6: Cox proportional Hazard Model for PM/DM associated ILD patients.
View Table 6
We described diagnosis of DM and DM-specific clinical signs such as Gottron sign and subungual erythema were strong predictors of mortality in DM-ILD at our cohort [21,22]. Physicians who diagnose ILD should have comprehensive overview of physical findings to distinguish from connective tissue disease (CTD) associated ILD especially in middle-aged patients. Among all CTDs, CADM have aggressive presentation of ILD [23,24]. Therefore, we should look for DM-specific physical findings such as Gottron sign, Heliotrope rash carefully. Subungual erythema is often detected systemic sclerosis (SSc) or PM-limited SSc overlap syndrome [25,26]. However, SSc or overlap syndrome is possible to be excluded by detailed clinical information or specific antibody such as anti-PM/Scl autoantibodies [22]. So, rapid clinical course with subungual erythema and DM-specific sign will be alarming signs for DM-associated ILD patients. Both Gottron sign and subungual erythema are associated extensive vasculitis. Therefore, poor prognosis of DM-associated ILD can be explained by these specific physical findings.
In autoantibodies, we showed anti-MDA 5 autoantibody-related ILD was poor prognosis and this antibody was strong predictor of mortality. This antibody is specific for dermatomyositis and may be found approximately 10% of adult PM/DM patients [27-29]. Both DM specific signs and anti-MDA 5 autoantibody are useful predictor of poor prognosis for DM associated ILD at our cohort. On the other hand, anti-tRNA-synthestase (Anti-ARS) autoantibodies was first described by Marguerie, et al. and this antibody associated ILD showed good prognosis compared with that of anti-MDA 5 autoantibody-related ILD in our study. Anti-ARS autoantibodies especially anti-Jo1 antibody is most frequently found in PM patients [14]. Recently ten anti-ARS autoantibodies including PL-7, PL-12 have reported in several studies [30,31]. Both PL-7 and PL-12 often have ILD. However, it requires more evidence. Connors et al. proposed revised Anti-ARS syndrome criteria with major items (arthritis, myositis, and ILD) and minor criteria (unexplained fever, Raynaud phenomenon and Mechanic's hand) [4].
This criteria is easy to use and practical for daily practice. These syndrome patients have good response with prednisolone. However, they often have relapse. Therefore, long-term careful follow-up will be required. Anti-MDA 5 autoantibody-related ILD and Anti - ARS syndrome have quite different clinical course. CADM and Anti-MDA 5 autoantibody positive patients are more prevalent in Japanese patients compared with non-Asian patients [32]. We should pay attention to detailed history and specific symptoms, signs such as Gottron sign, subungual erythema for PM/DM associated ILD patients.
In pulmonary function test (PFT), especially our DM ILD patient had preserved pulmonary function including %FVC, %TLC and %DLco at initial stage.
In PM/DM patients, restrictive disorder is affected by ILD or muscle involvement. Therefore, interpretation of PFT is rather difficult. Even if initial PFT is preserved, PM/DM associated ILD can develop acute deterioration during clinical course. Therefore, we should monitor both clinical, physiological and radiological changes carefully.
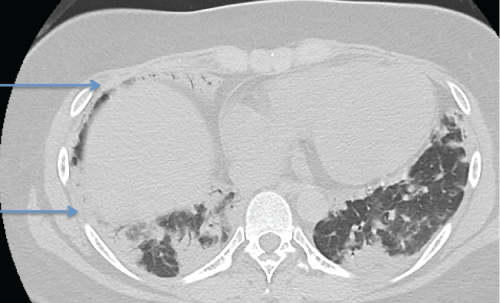
In terms of radiological findings, chest HRCT is crucial for evaluation of CTD-ILD. Among PM/DM patients, interstitial changes are frequently located in bilateral lower lobes [33,34]. GGO, consolidation, reticular opacity, traction bronchiectasis and linear opacities are frequent HRCT findings of PM-DM ILD. Linear opacity was found all our PM/DM ILD patients. Therefore, we thought this may not be possible prognostic discriminator and excluded our analysis. In Anti-ARS syndrome often have volume loss of lower lung field and peri-bronchovascular consolidation [31]. These HRCT findings correspond pathologically to organizing pneumonia (OP) or non-specific interstitial pneumonia (NSIP) or variant of organizing pneumonia with supervening fibrosis [35,36]. GGO and reticulation correspond to reversible fibrosis. We focused on lung tip consolidation. Because such findings were often seen in our CADM associated ILD. On the other hand, this consolidation was not detected in normal population (Figure 3). We think grade of lung tip consolidation was rather mild in PM-associated ILD patients. Therefore, more extensive lung tip consolidation of CADM patients caused strong limitation of movement of diaphragm and overall lower lung field. With these findings, we provide more intensive treatment immediately. We built up hypothesis that these lung tip consolidation or migratory bird sign may limit the movement of right diaphragm. In addition, there are more gravity dependent and more respiratory movement for removal of exudates in this extreme basal area. If persistent inflammation or fibrosis continue in this area, this will lead to severe restrictive disorder or profound dyspnea. Our study showed lung tip consolidation predicted mortality of CADM associated ILD patients. Therefore, our proposed hypothesis might be correct.
In treatment, PM related ILD patients can be managed with prednisolone alone especially anti-ARS syndrome group or with prednisolone and immunosuppressants such as azathioprine [13]. Over half of our PM-ILD cohort was controlled with prednisolone alone. However, regarding PM-associated ILD patients, PSL + FK506 improved mortality recently. On the contrary, DM associated ILD especially CADM ILD patients have poor prognosis as shown by our study.
Based on our useful predictor, when we diagnose CADM-ILD, we should start intensive therapy such as steroid pulse with calcineurin inhibitor such as cyclosporine or tacrolimus and intravenous cyclophosphamide, azathioprine [33,37-39].
We have limitations. First, it was s single-center retrospective study. Therefore, it did not represent overall PM/DM ILD patients. However, clinical symptoms, signs were comparable with previous reports. Second, some data were missing such as Ferritin and anti-ARS autoantibody. However, most important issue is tracing clinical course and catch-up prognostic signs or marker for PM/DM ILD. Third, radiological point of view, detecting clinical meaning of lung tip consolidation is novel finding. All facilities cannot perform chest HRCT. However, it is important to create close cooperation between private clinic and special center.
In conclusion, Gottron sign, subungual erythema, lung tip consolidation, diagnosis of DM and Anti-MDA-5 antibody are useful predictor of mortality in PM/DM associated ILD patients. Further studies are needed to evaluate the multi-dimension of PM/DM ILD patients.
References
-
Bohan A, Peter JB (1975) Polymyositis and dermatomyositis (first of two parts). N Engl J Med 292: 344-347.
-
Ernste FC, Reed AM (2013) Idiopathic inflammatory myopathies: current trends in pathogenesis, clinical features, and up-to-date treatment recommendations. Mayo Clin Proc 88: 83-105.
-
Dobloug C, Garen T, Bitter H, Stjärne J, Stenseth G, et al. (2015) Prevalence and clinical characteristics of adult polymyositis and dermatomyositis; data from a large and unselected Norwegian cohort. Ann Rheum Dis 74: 1551-1556.
-
Connors GR, Christopher-Stine L, Oddis CV, Danoff SK (2010) Interstitial lung disease associated with the idiopathic inflammatory myopathies: what progress has been made in the past 35 years? Chest 138: 1464-1474.
-
Marie I, Hatron PY, Dominique S, Cherin P, Mouthon L, et al. (2011) Short-term and long-term outcomes of interstitial lung disease in polymyositis and dermatomyositis: a series of 107 patients. Arthritis Rheum 63: 3439-3447.
-
Koreeda Y, Higashimoto I, Yamamoto M, Takahashi M, Kaji K, et al. (2010) Clinical and pathological findings of interstitial lung disease patients with anti-aminoacyl-tRNA synthetase autoantibodies. Intern Med 49: 361-369.
-
Sontheimer RD (2002) Would a new name hasten the acceptance of amyopathic dermatomyositis (dermatomyositis siné myositis) as a distinctive subset within the idiopathic inflammatory dermatomyopathies spectrum of clinical illness? J Am Acad Dermatol 46: 626-636.
-
Fujisawa T, Hozumi H1, Kono M1, Enomoto N1, Hashimoto D1, et al. (2014) Prognostic factors for myositis-associated interstitial lung disease. PLoS One 9: e98824.
-
Gerami P, Schope JM, McDonald L, Walling HW, Sontheimer RD (2006) A systematic review of adult-onset clinically amyopathic dermatomyositis (dermatomyositis sine myositis): a missing link within the spectrum of the idiopathic inflammatory myopathies. J Am Acad Dermatol 54: 597-613.
-
Cottin V, Thivolet-Bejui F, Reynaud-Gaubert M, Cadranel J, Delaval P, et al. (2003) Interstitial lung disease in amyopathic dermatomyositis, dermatomyositis and polymyositis. Eur Respir J 22: 245-250.
-
Fujisawa T, Suda T, Nakamura Y, Enomoto N, Ide K, et al. (2005) Differences in clinical features and prognosis of interstitial lung diseases between polymyositis and dermatomyositis. J Rheumatol 32: 58-64.
-
Suda T, Fujisawa T, Enomoto N, Nakamura Y, Inui N, et al. (2006) Interstitial lung diseases associated with amyopathic dermatomyositis. Eur Respir J 28: 1005-1012.
-
Saketkoo LA, Ascherman DP, Cottin V, Christopher-Stine L, Danoff SK, et al. (2010) Interstitial Lung Disease in Idiopathic Inflammatory Myopathy. Curr Rheumatol Rev 6: 108-119.
-
Lega JC, Fabien N, Reynaud Q, Durieu I, Durupt S, et al. (2014) The clinical phenotype associated with myositis-specific and associated autoantibodies: a meta-analysis revisiting the so-called antisynthetase syndrome. Autoimmunity Rev 13: 883-891.
-
Sontheimer RD, Miyagawa S (2003) Potentially fatal interstitial lung disease can occur in clinically amyopathic dermatomyositis. J Am Acad Dermatol 48: 797-798.
-
Tanizawa K, Handa T, Nakashima R, Kubo T, Hosono Y, et al. (2013) The prognostic value of HRCT in myositis-associated interstitial lung disease. Respir Med 107: 745-752.
-
Marguerie C, Bunn CC, Beynon HL, Bernstein RM, Hughes JM, et al. (1990) Polymyositis, pulmonary fibrosis and autoantibodies to aminoacyl-tRNA synthetase enzymes. Q J Med 77: 1019-1038.
-
Sato S, Hirakata M, Kuwana M, Suwa A, Inada S, et al. (2005) Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum 52: 1571-1576.
-
Tansley SL, Betteridge ZE, Gunawardena H, Jacques TS, Owens CM, et al. (2014) Anti-MDA5 autoantibodies in juvenile dermatomyositis identify a distinct clinical phenotype: a prospective cohort study. Arthritis Res Ther 16: R138.
-
Sumikawa H, Johkoh T, Colby TV, Ichikado K, Suga M, et al. (2008) Computed tomography findings in pathological usual interstitial pneumonia: relationship to survival. Am J Respir Crit Care Med 177: 433-439.
-
Fathi M, Dastmalchi M, Rasmussen E, Lundberg IE, Tornling G (2004) Interstitial lung disease, a common manifestation of newly diagnosed polymyositis and dermatomyositis. Ann Rheum Dis 63: 297-301.
-
(2015) "Idiopathic inflammatory myopathies and the lung." Jean-Christophe Lega, Quitterie Reynaud, Alexandre Belot, Nicole Fabien, Isabelle Durieu and Vincent Cottin. Eur Respir Rev 2015; 24: 216-238. Eur Respir Rev 24: 545.
-
Hamaguchi Y, Kuwana M, Hoshino K, Hasegawa M, Kaji K, et al. (2011) Clinical correlations with dermatomyositis- specific autoantibodies in adult Japanese patients with dermatomyositis: a multicenter cross-sectional study. Arch Dermatol 147: 391-398.
-
Mukae H, Ishimoto H, Sakamoto N, Hara S, Kakugawa T, et al. (2009) Clinical differences between interstitial lung disease associated with clinically amyopathic dermatomyositis and classic dermatomyositis. Chest 136: 1341-1347.
-
Raijmakers R, Renz M, Wiemann C, Egberts WV, Seelig HP, et al. (2004) PM-Scl-75 is the main autoantigen in patients with the polymyositis/scleroderma overlap syndrome. Arthritis Rheum 50: 565-569.
-
D'Aoust J, Hudson M, Tatibouet S, Wick J; Canadian Scleroderma Research Group, Mahler M, et al. (2014) Clinical and serologic correlates of anti-PM/Scl antibodies in systemic sclerosis: a multicenter study of 763 patients. Arthritis Rheumatol 66: 1608-1615.
-
Ghirardello A, Bassi N, Palma L, Borella E, Domeneghetti M, et al. (2013) Autoantibodies in polymyositis and dermatomyositis. Curr Rheumatol Rep 15: 335.
-
Hall JC, Casciola-Rosen L, Samedy LA, Werner J, Owoyemi K, et al. (2013) Anti-melanoma differentiation-associated protein 5-associated dermatomyositis: expanding the clinical spectrum. Arthritis Care Res (Hoboken) 65: 1307-1315.
-
Labrador-Horrillo M, Martinez MA, Selva-O'Callaghan A, Trallero-Araguas E, Balada E, et al. (2014) Anti-MDA5 antibodies in a large Mediterranean population of adults with dermatomyositis. J Immunol Res 2014: 290797.
-
Takato H, Waseda Y, Watanabe S, et al. (2013) Pulmonary manifestations of anti-ARS antibody positive interstitial pneumonia -With or without PM/DM. Respiratory Medicine 107: 128-133.
-
Hervier B, Devilliers H, Stanciu R, Meyer A, Uzunhan Y, et al. (2012) Hierarchical cluster and survival analyses of antisynthetase syndrome: phenotype and outcome are correlated with anti-tRNA synthetase antibody specificity. Autoimmun Rev 12: 210-217.
-
Chen Z, Cao M, Plana MN, Liang J, Cai H, et al. (2013) Utility of anti-melanoma differentiation-associated gene 5 antibody measurement in identifying patients with dermatomyositis and a high risk for developing rapidly progressive interstitial lung disease: a review of the literature and a meta-analysis. Arthritis Care Res (Hoboken) 65: 1316-1324.
-
Marie I, Hachulla E, Chérin P, Dominique S, Hatron PY, et al. (2002) Interstitial lung disease in polymyositis and dermatomyositis. Arthritis Rheum 47: 614-622.
-
Douglas WW, Tazelaar HD, Hartman TE, Hartman RP, Decker PA, et al. (2001) Polymyositis-dermatomyositis-associated interstitial lung disease. Am J Respir Crit Care Med 164: 1182-1185.
-
Lee JW, Lee KS, Lee HY, Chung MP, Yi CA, et al. (2010) Cryptogenic organizing pneumonia: serial high-resolution CT findings in 22 patients. AJR Am J Roentgenol 195: 916-922.
-
Travis WD, Costabel U, Hansell DM, King TE Jr, Lynch DA, et al. (2013) An Official American Thoracic Society/European Respiratory Society Statement: Update of the International Multidisciplinary Classification of the Idiopathic Interstitial Pneumonias. Am J Respir Crit Care Med 188: 733-748.
-
Selva-O'Callaghan A, Labrador-Horrillo M, Muñoz-Gall X, Martínez-Gomez X, Majó-Masferrer J, et al. (2005) Polymyositis/dermatomyositis-associated lung disease: analysis of a series of 81 patients. Lupus 14: 534-542.
-
Kameda H, Nagasawa H, Ogawa H, Sekiguchi N, Takei H, et al. (2005) Combination therapy with corticosteroids, cyclosporin A, and intravenous pulse cyclophosphamide for acute/subacute interstitial pneumonia in patients with dermatomyositis. J Rheumatol 32: 1719-1726.
-
Wilkes MR, Sereika SM, Fertig N, Lucas MR, Oddis CV (2005) Treatment of antisynthetase-associated interstitial lung disease with tacrolimus. Arthritis Rheum 52: 2439-2446.