International Journal of Neurology and Neurotherapy
Longitudinal White Matter Alteration in Prolonged Disorders of Consciousness due to Traffic Accidents
Hiroaki Abe1,2*, Keigo Shimoji3, Takeo Kondo2, Takanori Kochiyama4, Yoshihide Nagamine5, Satoru Fujiwara5, Yutaka Oouchida2 and Shin-Ichi Izumi2,6
1Department of Rehabilitation Medicine, Kohnan Hospital Tohoku Ryogo center, Sendai, Japan
2Department of Physical Medicine and Rehabilitation, Graduate School of Medicine, Tohoku University, Sendai, Japan
3Department of Diagnostic Radiology, Tokyo Metropolitan Geriatric Hospital, Tokyo, Japan
4Brain Activity Imaging Center, Advanced Telecommunications Research Institute, Kyoto, Japan
5Department of Neurosurgery, Kohnan Hospital Tohoku Ryogo center, Sendai, Japan
6Graduate school of Biomedical Engineering, Tohoku University, Sendai, Japan
*Corresponding author:
Hiroaki Abe, Department of Rehabilitation Medicine, Kohnan Hospital Tohoku Ryogo center, Nagamachi Minami, 4-20-1, Taihaku-ku, Sendai, Japan, Tel: +81-248-2131, Fax: +81-248-1906, E-mail: abehi0827@gmail.com
Int J Neurol Neurother, IJNN-4-064, (Volume 4, Issue 1), Original Research; ISSN: 2378-3001
Received: November 10, 2016 | Accepted: January 20, 2017 | Published: January 23, 2017
Citation: Hiroaki A, PT, Shimoji K, Kondo T, Kochiyama T, Yoshihide Nagamine Y, et al. (2017) Longitudinal White Matter Alteration in Prolonged Disorders of Consciousness due to Traffic Accidents. Int J Neurol Neurother 4:064. 10.23937/2378-3001/1410064
Copyright: © 2017 Hiroaki A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: We analyzed longitudinal white matter alterations by diffusion tensor imaging (DTI) in patients with prolonged disorders of consciousness (PDC).
Participants: Seventeen patients with PDC having an average of 328.8 days post-injury and eleven control participants.
Main measures: All participants underwent two DTI studies. Fractional anisotropy (FA) values were analyzed using whole-brain analysis (WBA) of 7 patients with no marked brain deformities; mean FA values of forceps minor (mFAFM) were subsequently measured by regional-brain analysis (RBA). Relationships between WB and regional FA values and recovery from PDC were evaluated.
Results: WBA in the PDC group showed that FA values were significantly lower in multiple white matter regions (P < 0.05), particularly in part of the forceps minor, in the second scan than they were in the first scan. RBA showed that mFAFM significantly decreased (P < 0.01). Further, WBA revealed that a significant positive correlation was observed between the degree of recovery from PDC and the difference in the number of voxels with FA values > 0.2 (r = 0.65, P < 0.01) between the first and second scans.
Conclusion: Our results showed microstructural white matter changes in patients with PDC, suggesting recovery from PDC with long-term treatment.
Keywords
Traumatic brain injury, Prolonged consciousness disorder, Diffusion tensor imaging, White matter, Longitudinal alteration, Fractional anisotropy, Vegetative state, Minimal consciousness state, Traffic accident, Tract-based spatial statistics
Introduction
Severe traumatic brain injury (TBI) is one of the most common causes of long-term disability conditions, including coma and persistent vegetative or minimally conscious states [1], which are widely accepted to arise from various forms of structural damage, such as diffuse axonal injury (DAI) [1,2]. Patients with severe prolonged disorders of consciousness (PDC),including vegetative states, typically have unfavorable outcomes although a few patients exhibit slow, subtle, and minor clinical changes [3,4]. For the past 3 years, our institution has provided in patients suffering from PDC due to traffic accident-related severe brain injury not only traditional therapies, such as physical therapy, occupational therapy, and nursing care, but also other specific non-traditional treatments, such as music therapy, aroma massage, and exposure to natural environments (i.e., feeling the sunlight, blowing wind, and seasonal temperature changes). Indeed, a few patients have shown slight positive reactions during inpatient residency at our institution.
There are validated scoring tools to evaluate the severity of consciousness disorders during the acute stages after TBI [5,6], but there are no appropriate scoring tools for evaluating chronic consciousness disorders. In particular, there are no scoring tools with proven utility for evaluating slight improvements from PDC. Therefore, it is difficult to assess the benefits of providing any intervention to patients with PDC. The "Kohnan score", developed at our institution to resolve this issue [5], reportedly displayed unidimensionality and higher intra- and inter-rater reliability [6]. Using this measure, we found that some patients with PDC exhibited improvement. However, there are no valid predictors of long-term positive clinical response in patients with PDC. Long-term practices without specific clinical goals are provided to patients with PDC by many medical personnel. Therefore, it is important to develop predictors of long-term change elicited through long-term therapeutic intervention within these patients.
Decreased fractional anisotropy (FA) in some regions [7], measured by magnetic resonance diffusion tensor imaging (MR-DTI) [8], recently proved useful for assessing white matter damage in vivo [2,9-20]. Especially, corpus callosum was frequently reported to show significant damage from traumatic brain injury in previous studies [9,11]. Long-term patient outcomes were reportedly associated with the degree of white matter alteration revealed by DTI findings in patients with mild or severe TBI [2,9-20]. Furthermore, several studies reported that longitudinal alteration occurred in patients with severe TBI and that unfavorable outcomes correlated with decreased FA values detected in several brain regions [11,18].
This study analyzed longitudinal alteration in anatomical connections of white matter in patients with PDC due to traffic accidents and evaluated the association of microstructural imaging biomarkers in white matter with clinical markers to determine their potential clinical utility.
Methods
Patients
We retrospectively reviewed 17 patients at our institution with chronic, severe PDC resulting from traffic accident-related injury. All patients with PDC underwent 3.0T MR-DTI studies at admission and after one year of residency. The age range of patients was 23-86 (mean, 58.8 ± 18.3) years, and the median number of days from injury to recruitment for the study, i.e., admission to our institution, median was 264 (IQR: 226 - 391) days. Patient characteristics are presented in table 1. The first MR-DTI scan was performed at admission [266 (230 - 400) days after the initial injury], and the second scan was performed approximately one year after admission [340 (284 - 429) days after the initial injury].
![]()
Table 1: Characteristics of patients with prolonged consciousness disorders.
View Table 1
Furthermore, we prospectively recruited 11 healthy normal volunteers (4 males and 7 females) for comparison. The age range of the healthy participants was 26-60 (41.1 ± 10.8) years (Table 2).
![]()
Table 2: Clinical characteristics of patients with prolonged consciousness disorders.
View Table 2
Standard protocol approvals, registrations, and patient consents
This study was conducted in compliance with the ethical principles that originated in the Declaration of Helsinki regarding biomedical research on human subjects and informed consent regulations. Approval from the institutional ethics committee was obtained prior to the initiating of the study.
PDC assessment
PDC was assessed using the Kohnan score. The Kohnan score was developed to evaluate the severity of consciousness disorder, with a particular focus on the persistent vegetative state. (Appendix) [6]. This score comprises even parameters, with each parameter divided into 5 grades: extreme (10 points), severe (9 points), moderate (7 or 8 points), mild (5 points) and slight (0 points). We additionally assessed general functional recovery using the Extended Glasgow Outcome Scale (GOSE) [21], which ranges from 1 to 8, with higher scores suggesting better functional outcomes. These assessments were performed at admission and at 1 year after admission.
Image acquisition
MRI was performed using a 3.0 T Signa Excite HD scanner (General Electric, Milwaukee, WI, USA). The general scan parameters for MR-DTI were as follows: echo time, 59 ms; repetition time, 9,000 ms; flip angle, 90°; slice thickness, 3 mm with no gap; field of view, 28.8 × 28.8 cm; acquisition matrix, 96 × 96; image matrix, 256 × 256 with a voxel size of 1.125 × 1.125 × 3.0 mm; number of excitations, 1; and bandwidth, 250 kHz. Images were obtained with 15-directional diffusion encoding (b value, 1,000 s/mm2 in each direction) and without diffusion encoding (b value, 0 s/mm2). A total of 46 axial section images covering the entire cerebrum were obtained. The inferior MR-DTI slices were positioned at the medulla oblongata during acquisition.
Two MR-DTI scans were performed in healthy, normal participants; the second scan was performed 1 year after the first scan. Scan parameters for MR-DTI in normal participants were different from those in patients with PDC because of an MRI equipment upgrade. The scan parameters were as follows: echo time, 65.8 ms; repetition time, 15,000 ms; flip angle, 90°; slice thickness, 3 mm with no gap; field of view, 28.8 × 28.8 cm; acquisition matrix, 96 × 96; image matrix, 256 × 256 with a voxel size of 1.125 × 1.125 × 3.0 mm; number of excitations, 1; and bandwidth, 1953.12 kHz. Images were obtained with 15-directional diffusion encoding (b value, 1,000 s/mm2 in each direction) and without diffusion encoding (b value, 0 s/mm2). A total of 46 axial section images covering the entire cerebrum were obtained. Other settings were the same as in patients with PDC.
We performed a whole-brain (WB) statistical analysis of FA maps with a tract-based spatial statistics (TBSS) technique using the diffusion toolbox implemented in the Oxford Centre for Functional MRI of the Brain(FMRIB) software library (FSL:http://www.fmrib.ox.ac.uk/fsl/). Images underwent eddy current distortion correction [22] and skull-stripping using the Brain Extraction Tool [23]. The FA maps of each patient were aligned into the Montreal Neurological Institute spaces using nonlinear registration algorithms with FNIRT in FSL. The FA values of each patient were subsequently projected onto a mean FA skeleton, which represented centers of white matter tracts common to all patients. The resulting FA skeleton data were used in the following voxel-wise WB statistical analysis. The longitudinal changes in FA values between the first and second scans were evaluated using a permutation-based randomized test and inference using the threshold-free cluster enhancement method implemented in FSL. The statistical threshold for all image analyses was set at P < 0.05, with family-wise errors corrected for multiple comparisons of the voxel-wise WB analysis. We also measured the mean FA value in WB (mFAWB) and the number of voxels in which FA values were > 0.2 in WB (VsFA0.2) in the FA skeleton.
After TBSS analysis, we performed a regional brain assessment with a tract-specific analysis (TSA) of FA values using dTV II and Volume-One 1.72 software, developed by Masutani, et al. (http://www.volume-one.org/).
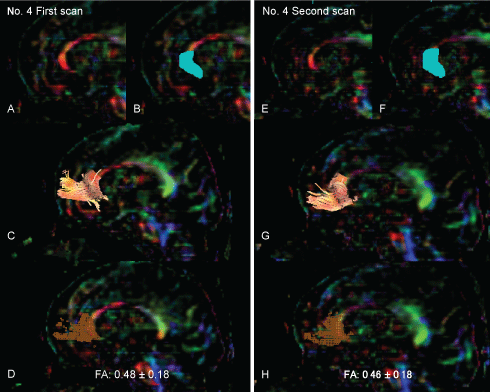
White matter tractography was assessed in all subjects using the region-of-interest (ROI) method. Three-dimensional anisotropy contrast (3DAC) color-coded maps were used to precisely and objectively position ROIs in white matter tracts (Figure 1A and Figure 1E). The relatively large target ROI, including the entirety of the genu of the corpus callosum, was manually placed on a reconstructed mid-sagittal section of the 3DAC image (Figure 1B and Figure 1F). Drawn tractography was generated using the threshold values of line-tracking termination as FA > 0.18 (Figure 1C and Figure 1G). This tractography was considered to be the forceps minor (FM). The dTV II software provides a track-line voxelization function that extracts the tracking line of the white matter tract to 3D voxels while preserving original tensor parameters. Voxelization along the FM tract was also performed (Figure 1D and Figure 1H). All voxels, including the tracked lines, were evaluated, and the mean FA values in the registered voxels within the FM core (mFAFMs) were measured [7,8]. In all subjects, we also measured FA values regarding mFAWB and VsFA0.2 in the whole brain, including both the white and gray matter regions. Therefore, we were unable to extract only white matter because some patients had too much deformity to perform accurate segmentation.
.
Figure 1: The drawing strategy for tractography and voxelization along the tractography of the forceps minor. Both panels show the tractography of the forceps minor and voxelization along the tract of the forceps minor in the representative subject (patient No. 4).
The left panel shows the tractography in the first scan. The right panel shows the tractography at the second scan. The FA value of the forceps minor decreased from 0.48 ± 0.18 in the first scan to 0.46 ± 0.18 in the second scan.
View Figure 1
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) 24.0 for Mac (IBM SPSS; Chicago, IL, USA). Comparisons over time for Kohnan scores, GOSE, mFAWB, VsFA0.2, and mFAFM of the groups were conducted using either the paired t-test or the Wilcoxon rank-sum test based on the results of the Shapiro-Wilk test. The Spearman rank correlation coefficient was used to evaluate the relationships among age, the number of days from injury to admission, degree of recovery from PDC as determined by change in the Kohnan score during the first year after admission, and FA values. The FA values used for comparison were FAFM, mFAWB, and VsFA0.2 in the first scan and the differences in mFAFM, mFAWB, and VsFA0.2 between the first and second scans. We also calculated the partial correlation coefficient between each FA values that showed a significant relationship with the degree of recovery from PCD according to the Spearman rank correlation coefficient and with that from PCD with age and gender as covariates.
Results
In this study, 15 patients had an at-admission GOSE score of 2 (vegetative state), and two patients had an initial GOSE score of 3 (severe disorder). However, the Kohnan scores at admission showed more variability (range, 29-68), ranging from the minimally conscious state (< 39) to the completely vegetative state (> 65). After one year, there was no significant change in GOSE scores (P = 0.16, Wilcoxon rank-sum test): 13 patients had a GOSE score of 2, whereas 4 patients had a GOSE score of 3. In contrast, a subset of PDC patients showed a significant improvement as evidenced by the Kohnan score (60.0 ± 11.5 vs. 52.3 ± 21.4, P < 0.01; Wilcoxon rank-sum test) (Table 3). The mean difference in the Kohnan scores assessed in the first and second scans was 7.7 ± 10.5.
![]()
Table 3: Characteristics of seven patients with prolonged consciousness disorders evaluated by TBSS.
View Table 3
In patients with PDC, mFAWB and VsFA0.2 values in the first scan were 0.25 ± 0.04 and 225,646.76 ± 56,386.77, respectively, whereas those in the second scan were 0.24 ± 0.03 and 204,747.41 ± 38,384.88, respectively (Table 3). Patients with PDC showed no significant difference over time in mFAWB (P = 0.12, paired t-test) value, including gray matter comparison. In contrast, they showed significant difference over time in VsFA0.2 (P = 0.02, Wilcoxon rank-sum test) value. There were no significant differences in mFAWB (P = 0.16, paired t test) and VsFA0.2 (P = 0.16, paired t-test) values between the first and second scans in normal, healthy participants (Table 2).
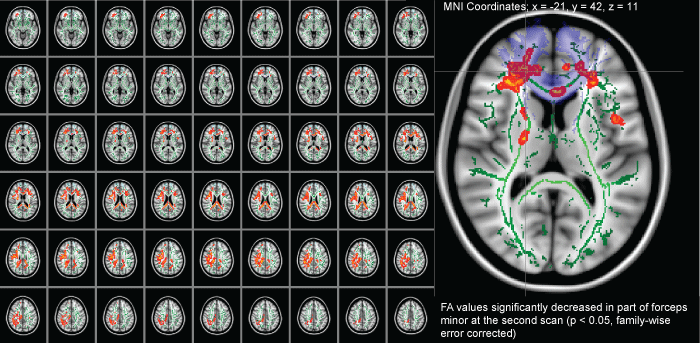
In some patients exhibiting marked brain atrophy, extended cerebral ventricles, and/or brain deformities, their imaging scans showed considerably missed registrations. Therefore, 10patients with severe brain deformities were excluded. Thus, TBSS included five male and two female patients with a mean age of 59.7± 8.4 years and a mean of 264 (188-323) days from injury to admission (Table 3). TBSS revealed that the FA values in the second scan were significantly lower than those in the first scan in numerous, small white matter regions (Figure 2). In particular, TBSS revealed a significant decrease over time in FA values for parts of the genu of the corpus callosum. Similarly, mFAWB in white matter skeleton (WMS) (0.37 ± 0.032 vs. 0.35 ± 0.018, P < 0.05) and VsFA0.2 in WMS (144,269.57 ± 4,794.88 vs. 138,945.43 ± 4,764.62, P < 0.01) values were significantly lower in the second scan than those in the first scan (Table 3). In contrast, by TBSS, there were no significant WB changes in the 11 normal participants.
.
Figure 2: A comparison of longitudinal diffusion tensor imaging after traumatic axonal injury in patients during the chronic phase (at admission) and during the extended chronic phase (1 year after admission). The images (red-yellow) depict instances where diffusion tensor imaging measures during the chronic phase were more than those of the extended chronic phase. The green tracts are representative of the skeleton created by tract-based spatial statistics. The light violet regions are representative of the forceps minor as specified by the Johns Hopkins University white matter tractography atlas provided with the FSL. The FA values significantly decreased in part of the forceps minor in the second scan (panels B) as compared with that in the first scan (0.46 ± 0.18 vs. 0.48 ± 0.18, P < 0.05, family-wise error correction).
View Figure 2
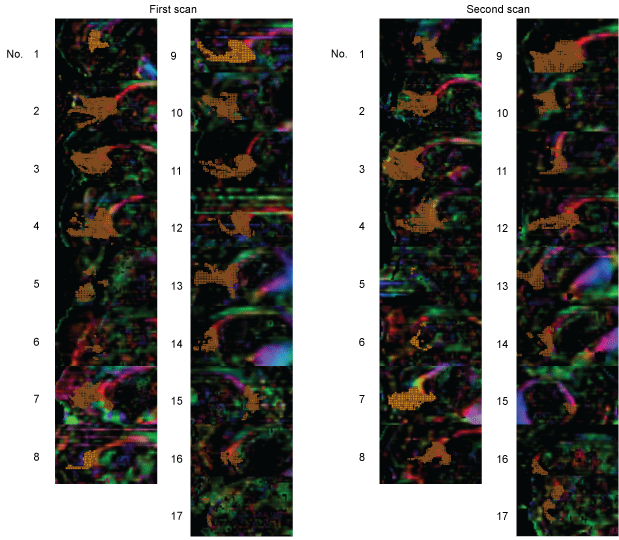
Following TBSS, regional analysis using TSA was performed to determine the FA values of FM for all 17 patients with PDC. The results indicated a significant decrease in mFAFM over time (0.39 ± 0.1 vs. 0.37 ± 0.1, P < 0.001, Figure 3).
.
Figure 3: Voxelization along the FM tract was performed on 17 patients. In patient 15, the right-sided view is presented as left-side tractography because drawing the forceps minor was not possible with this patient.
View Figure 3
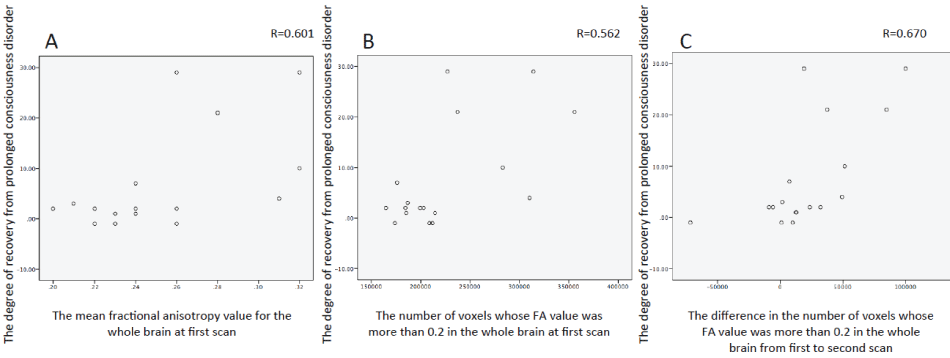
In the correlation analysis of all 17 patients, no significant correlations were observed between the degree of recovery from PDC and the difference in mFAFM over time (Table 4). However, a significant positive correlation was observed between the degree of recovery from PDC and time-dependent changes in VsFA0.2 (r = 0.56, P = 0.02) between the first and second scans, and VsFA0.2 (r = 0.67, P < 0.01), mFAWB (r = 0.60, P = 0.01) at first scan (Figure 4). Similarly, a significant positive correlation was observed between the degree of recovery from PDC and time-dependent changes in VsFA0.2 (r = 0.61, P = 0.02) between the first and second scans and between VsFA0.2 (r = 0.62, P = 0.02) and mFAWB (r = 0.60, P = 0.02) at first scan according partial correlation analysis. There was a significant correlation between the change in VsFA0.2 values and the VsFA0.2 value in the first scan (r = 0.67, P < 0.01).
.
Figure 4: Analysis of positive correlation between the degree of recovery from prolonged consciousness disorder and the mean fractional anisotropy value for the whole brain at first scan (A, r = 0.601, P = 0.011). Analysis of positive correlation between the degree of recovery from prolonged consciousness disorder and the number of voxels whose FA value was more than 0.2 in the whole brain at first scan (B, r = −0.562, P = 0.019). Analysis of positive correlation between the degree of recovery from prolonged consciousness disorder and the difference in the number of voxels whose FA value was more than 0.2 in the whole brain from first to second scan (C, r = 0.670, P = 0.003).
View Figure 4
![]()
Table 4: Correlation between the degree of recovery from prolonged consciousness disorder and variables for 17 patients.
View Table 4
Discussion
We explored the evolution of damaged white matter by examining MR-DTI changes in patients with PDC obtained at admission to our institution and again at 1year after admission. Seven patients with severe complicated DAI who demonstrated subcortical white matter lesions and had not undergone invasive treatment were evaluated. Results showed decreased FA values in multiple regions of some WMSs. Siminally, mFAWB in WMS and VsFA0.2 in WMS values significantly decreased. In particular, the decreased FA voxels were concentrated in the corpus callosum (i.e., FM) compared with no such significant changes detected in normal participants upon using TBSS. TSA performed on 17 patients with PDC revealed that mFAFM was significantly decreased during the year after admission; however, no significant correlations were observed among the degree of recovery from PDC, the difference in the mFAFM values over time and the mFAFM value in the first scan. In contrast, a significant correlation was observed between the degree of recovery from PDC and the time-dependent changes in VsFA0.2, mFAWB, and VsFA0.2 values in the first scan. Currently, there are no valid predictors of long-term positive clinical response in patients with PDC. Several clinicians provide long-term care without established, specific clinical goals for patients with PDC. Therefore, it is critical to develop predictors of long-term changes elicited through long-term therapeutic intervention in these patients. Our results demonstrated microstructural white matter changes occurring in patients with PDC, suggesting that the assessment of white matter changes facilitates the development of valid long-term outcome predictors.
Although previous studies have evaluated chronic DAI, we used TBSS, a voxel-based refinement of MR-DTI data analysis, in this study. Spatially, diffusion changes observed in the chronic stage occurs across various white matter regions, including the expected areas (i.e., corpus callosum), as revealed by histopathological and radiological studies of DAI [2,9-11,13-20]. This finding is consistent with prior reports of decreased FA values during the chronic phase of DAI [11,14,15,18-20]. Sidaros, et al. examined longitudinal microstructural white matter alteration in individuals with severe TBI in the late sub-acute and chronic stages and investigated potential correlations with 1-year clinical outcomes [11]. They found decreased FA values in all the investigated white matter regions, such as the posterior aspect of the corpus callosum (PCC), posterior limb of internal capsule (PLIC), centrum semiovale (CSO), and cerebral peduncle (CP) in the late sub-acute stage, approximately 8 weeks post-injury. Follow-up MR-DTI, which was performed approximately 12 months post-injury, showed FA normalization in PLIC and CSO, primarily in patients with unfavorable outcomes (i.e., GOSE score of 4 [moderate disability] or 5 [low disability]). Similarly, FA values remained depressed in PCC and CP, particularly in patients with unfavorable outcomes (i.e., GOSE scores ≤ 3 [severe disability]). Although TBSS showed decreased FA values in multiple WMS regions, there were differences between the subjects in previous studies and our study. Our subjects generally had more severe outcomes; all our patients had GOSE scores ≤ 3, indicating more severe disability than those in previous studies [2,9-15,18-20]. Thus, inter-study differences in results may reflect DAI severity.
Our first scan data were collected 334.2 ± 176.7 days after injury, thus reflecting chronic white matter damage. Furthermore, longitudinal white matter alteration, indicated by the difference in VsFA0.2 values between the first and second scans, was associated with the degree of PDC recovery. In contrast, our results showed no correlation between the degree of PDC recovery and the change in mFAFM, which suggested that patients with PDC have multiple regions with white matter injury. Thus, we speculated that an overall white matter evaluation is a better indicator of recovery than a focal area evaluation in PDC.
These results suggested the existence of progressive microstructural changes in the white matter of patients with chronic PDC due to severe DAI. PDC was shown to cause microstructural alteration in white matter. The time-dependent change in VsFA0.2 values positively correlated with the degree of recovery from PDC, indicating that a time-dependent decrease in FA values could lead to a better outcome in PDC. These results are inconsistent with those of several previous reports, indicating that a time-dependent decrease in FA values was related to poor outcomes as assessed by cognitive function [18] and GOSE [11]. These contradictory findings may reflect the difference in PDC severity between studies; most of our patients had GOSE scores of ≤ 3 and showed subtle and minor clinical changes. Although significant improvements were revealed by the Kohnan scores, the changes were not significant based on GOSE scores, suggesting that improvements in our patients were lower than those reported in previous studies [11,18]. Conversely, these divergent findings might reflect the difference in the investigated phase. Compared to previous studies, we included patients with PDC in the extended chronic phase. Longitudinal FA value change may differ between the sub-acute (> 7.5 months) [2,9,14-15] and extended chronic phases (> 1 year) as well as between the chronic (approximately within 1 year) [11,17] and extended chronic phases (> 1 year). Moreover, there was a significant correlation between the VsFA0.2 value in the first scan and time-dependent change in the VsFA0.2 value. Our results suggested that a faster assessment of white matter damage is a better predictor of long-term outcome. The change in VsFA0.2 value strongly correlated with the initial VsFA0.2 value, which might indicate that an achievement of recovery from PDC correlated with less DAI. In patients with PDC, the degree of improvement was very small, as detected by the absence of change in GOSE scores. In patients with PDC, the induction of a severe inactive state due to brain injury might lead to secondary neurodegeneration that might be detected by MR-DTI. In the patients with severe whole brain damage, whole brain FA values are very low from the very early stage. Therefore, these patients might have small voxels that might contribute to reduced FA values. In contrast, long-term changes in FA values might result from inactivity in only patients with comparatively less brain damage. MR-DTI was able to detect microstructural changes. Based on our findings, we speculate that only patients with decreased FA values in multiple voxels over an extended time might show improvement from PCD. However, the underlying mechanism behind longitudinal alteration in white matter remains unclear. Thus, further longitudinal studies are warranted that combine MR-DTI with volumetric measurements and other neuroimaging modalities, such as magnetoencephalography. The potential of MR-DTI use as a prognostic tool needs further investigation in studies with larger number of subjects.
In conclusion, our results showed microstructural white matter changes occurring over time in patients with PDC and suggested that FA values are useful indices of white matter alteration in these patients. As a noninvasive modality, MR-DTI provides in vivo quantitative pathophysiological information. Tracking microstructural changes in white matter over time has the potential to measure neuroplasticity and repair after TBI and may eventually be utilized to monitor therapeutic responses, which requires further study.
Disclosures
Dr. Hiroaki Abe, Dr. Shimoji Keigo, Dr. Takeo Kondo, Dr. Takanori Kochiyama, Dr. Yoshihide Nagamine, Dr. Satoru Fujiwara, Dr. Yutaka Oouchida, and Prof. Shin-Ichi Izumi report no disclosures.
Funding Sources
This study was supported by a medical research grant on traffic accidents from The General Insurance Association of Japan.
Acknowledgments
The authors would like to thank the patients and normal healthy volunteers who participated in this study. The authors would also like to thank Tomohiro Chiba and the rest of the imaging sta for their skilled MRI acquisition. This work was partially supported by JSPS KAKENHI Grant Number 26120007. This work was supported by JSPS KAKENHI Grant Number JP16H06280, Grant-in-Aid for Scientific Research on Innovative Areas- Resource and technical support platforms for promoting research ‘Advanced Bioimaging Support’. We also thank Enago for the English language review.
References
-
Meythaler JM, Peduzzi JD, Eleftheriou E, Novack TA (2001) Current concepts: diffuse axonal injury-associated traumatic brain injury. Arch Phys Med Rehabil 82: 1461-1471.
-
Ljungqvist J, Nilsson D, Ljungberg M, Sorbo A, Esbjornsson E, et al. (2011) Longitudinal study of the diffusion tensor imaging properties of the corpus callosum in acute and chronic diffuse axonal injury. Brain Inj 25: 370-378.
-
Luaute J, Maucort-Boulch D, Tell L, Quelard F, Sarraf T, et al. (2010) Long-term outcomes of chronic minimally conscious and vegetative states. Neurology 75: 246-252.
-
Estraneo A, Moretta P, Loreto V, Lanzillo B, Santoro L, et al. (2010) Late recovery after traumatic, anoxic, or hemorrhagic long-lasting vegetative state. Neurology 75: 239-245.
-
Fujiwara S, Nakasato N, Ogasawara K (1994) Evaluation of the severity of prolonged consciousness disturbances after head injury: A scoring system developed in our department. Proceedings of the 2nd Annual Meeting of the Society for Treatment of Coma 2: 173-183.
-
Fujiwara S, Nakasato N, Nagamine Y, Yoshimoto T, Suematsu K, et al. (1997) Reliability and factorial structure of a rating scale for persistent vegetative state. No To Shinkei 49: 1139-1145.
-
Yasmin H, Nakata Y, Aoki S, Abe O, Sato N, et al. (2008) Diffusion abnormalities of the uncinate fasciculus in Alzheimer's disease: diffusion tensor tract-specific analysis using a new method to measure the core of the tract. Neuroradiology 50: 293-299.
-
Yasmin H, Aoki S, Abe O, Nakata Y, Hayashi N, et al. (2009) Tract-specific analysis of white matter pathways in healthy subjects: a pilot study using diffusion tensor MRI. Neuroradiology 51: 831-840.
-
Wang JY, Bakhadirov K, Abdi H, Devous MD Sr, Marquez de la Plata CD, et al. (2011) Longitudinal changes of structural connectivity in traumatic axonal injury. Neurology 77: 818-826.
-
Wang JY, Bakhadirov K, Devous MD Sr, Abdi H, McColl R, et al. (2008) Diffusion tensor tractography of traumatic diffuse axonal injury. Arch Neurol 65: 619-626.
-
Sidaros A, Engberg AW, Sidaros K, Liptrot MG, Herning M, et al. (2008) Diffusion tensor imaging during recovery from severe traumatic brain injury and relation to clinical outcome: a longitudinal study. Brain 131: 559-572.
-
Greenberg G, Mikulis DJ, Ng K, DeSouza D, Green RE (2008) Use of diffusion tensor imaging to examine subacute white matter injury progression in moderate to severe traumatic brain injury. Arch Phys Med Rehabil 89: S45-50.
-
Perlbarg V, Puybasset L, Tollard E, Lehericy S, Benali H, et al. (2009) Relation between brain lesion location and clinical outcome in patients with severe traumatic brain injury: a diffusion tensor imaging study using voxel-based approaches. Hum Brain Mapp 30: 3924-3933.
-
Perez AM, Adler J, Kulkarni N, Strain JF, Womack KB, et al. Longitudinal white matter changes after traumatic axonal injury. J Neurotrauma 31: 1478-1485.
-
Kumar R, Husain M, Gupta RK, Hasan KM, Haris M, et al. (2008) Serial changes in the white matter diffusion tensor imaging metrics in moderate traumatic brain injury and correlation with neuro-cognitive function. J Neurotrauma 26: 481-495.
-
Hulkower MB, Poliak DB, Rosenbaum SB, Zimmerman ME, Lipton ML, et al. (2013) A decade of DTI in traumatic brain injury: 10 years and 100 articles later. Am J Neuroradiol 34: 2064-2074.
-
Newcombe VF, Williams GB, Scoffings D, Cross J, Carpenter TA, et al. (2010) Aetiological differences in neuroanatomy of the vegetative state: insights from diffusion tensor imaging and functional implications. J Neurol Neurosurg Psychiatry 81: 552-561.
-
Farbota KD, Bendlin BB, Alexander AL, Rowley HA, Dempsey RJ, et al. (2012) Longitudinal diffusion tensor imaging and neuropsychological correlates in traumatic brain injury patients. Front Hum Neurosci 6: 160.
-
Sugiyama K, Kondo T, Oouchida Y, Suzukamo Y, Higano S, et al. (2009) Clinical utility of diffusion tensor imaging for evaluating patients with diffuse axonal injury and cognitive disorders in the chronic stage. J Neurotrauma 26: 1879-1890.
-
Arenth PM, Russell KC, Scanlon JM, Kessler LJ, Ricker JH (2014) Corpus callosum integrity and neuropsychological performance after traumatic brain injury: a diffusion tensor imaging study. J Head Trauma Rehabil 29: E1-E10.
-
Jennett B. Snoek J, Bond MR, Brooks N (1981) Disability after severe head injury: observations on the use of the Glasgow Outcome Scale. J NeurolNeurosurg Psychiatry 44: 285-293.
-
Behrens TE, Woolrich MW, Jenkinson M, Johansen-Berq H, Nunes RG, et al. (2003) Characterization and propagation of uncertainty in diffusion-weighted MR imaging. Magn Reson Med 50: 1077-1088.
-
Smith SM (2002) Fast robust automated brain extraction. Hum Brain Mapp 17: 143-155.





