International Journal of Diabetes and Clinical Research
The Combination of Canagliflozin with a Half-Dose of Glimepiride Improves Glycemic Control and Blood Glucose Perturbation without Increasing the Risk of Hypoglycemia and Ketosis
Mayuko Harada, Yasuhisa Abe, Yuki Maeda, Michiko Kajikawa, and Mitsuru Hashiramoto*
Division of Diabetes and Endocrinology, Yodogawa Christian Hospital, Japan
*Corresponding author:
Mitsuru Hashiramoto, MD, PhD, Division of Diabetes and Endocrinology, Yodogawa Christian Hospital, 1-7-50 Kuni-Jima, Higashi-Yodogawa-Ku, Osaka 533-0024, Japan, Tel: +81-6-6322-2259, Fax: +81-6-6-6320-6308, E-mail: mhashira-ych@xrj.biglobe.ne.jp
Int J Diabetes Clin Res, IJDCR-3-065, (Volume 3, Issue 3), Original Article; ISSN: 2377-3634
Received: September 09, 2016 | Accepted: September 30, 2016 | Published: October 04, 2016
Citation: Harada M, Abe Y, Maeda Y, Kajikawa M, Hashiramoto M (2016) The Combination of Canagliflozin with a Half-Dose of Glimepiride Improves Glycemic Control and Blood Glucose Perturbation without Increasing the Risk of Hypoglycemia and Ketosis. Int J Diabetes Clin Res 3:065. 10.23937/2377-3634/1410065
Copyright: © 2016 Harada M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aim: To evaluate the efficacy and safety of administering canagliflozin to Japanese type 2 diabetes mellitus (T2DM) patients and reducing the dosage of their existing regimen of sulfonylurea (SU) by half.
Methods: Seventeen Japanese T2DM patients on antidiabetes medication (SU) completed this 12-week prospective single-arm open-label study. First, the dose of SU was reduced by half and a concomitant administration of canagliflozin (100 mg/day) was initiated. Glycemic control and other metabolic parameters were measured at weeks 0, 4, 8 and 12.
Results: At 12 weeks, average HbA1c levels had declined from 7.3 ± 0.3% (56 mmol/mol) to 7.1 ± 0.4% (53 mmol/mol) (p < 0.05). Continuous glucose monitoring showed average blood glucose (BG) levels (mg/dl) reduced from 156.8 ± 23.5 to 129.4 ± 10.4 (p < 0.05) with a standard deviation (SD) of from 45.7 ± 12.6 to 32.4 ± 9.0 (NS). Neither severe hypoglycemia nor other serious adverse events developed during the study period. Serum ketone bodies were transiently elevated from 61 ± 47 to 162 ± 140 (p < 0.05) at 4 weeks but were decreased afterward.
Conclusion: This study showed that a combination of canagliflozin preceded by a reduced dose of SU resulted in an acceptable short-term tolerability and a significant improvement in glycemic control without increasing the risk of hypoglycemia and severe ketosis.
Keywords
SGLT2 inhibitor, Sulfonylurea, Canagliflozin, Continuous glucose monitoring, Hypoglycemia, Ketosis, Ketoacidosis
Abbreviations
T2DM: Type 2 Diabetes Mellitus; SU: Sulfonylurea; SD: Standard Deviation; CV: Cardiovascular; SMBG: Self-Monitoring of Capillary Blood Glucose Levels; CGMs: Continuous Glucose Monitoring System; SGLT: Sodium Glucose Transporter; BG: Blood Glucose; eGFR: estimated Glomerular Filtration Rate; FPG: Fasting Plasma Glucose; SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HOMA: Homeostasis Model Assessment; LDL-C: Low Density Lipoprotein Cholesterol; HDL-C: High Density Lipoprotein Cholesterol; TG: Triglyceride; FFA: Free Fatty Acid; Ht: Hematocrit; Alb: Albumin; Cr: Creatinine; AEs: Adverse Events; BMI: Body Mass Index; DKA: Diabetic Ketoacidosis; MAGE: Mean Amplitude of Glycemic Excursions; T1DM: Type 1 Diabetes Mellitus; MODD: Mean of the Daily Difference
Introduction
The application of sulfonylurea (SU) remains one of the most popular approaches in pharmacological therapy for type 2 diabetes mellitus (T2DM) patients because SU exerts potent stimulatory effects on insulin secretion from pancreatic β cells and is relatively inexpensive and well tolerated. Hypoglycemia, however, is a major safety concern that is associated with the use of SUs, because it impairs a patient's quality of life and can become life threatening when protracted.
The cardiovascular (CV) toxicity of hypoglycemia has drawn renewed attention because of recent large-scale clinical trials [1]. Hypoglycemia, particularly when severe, is a powerful stimulant of the sympathetic nervous system, which may potentially trigger CV events such as sudden cardiac arrest, acute myocardial infarction, and cardiac arrhythmias including prolongation of the QT interval and ventricular arrhythmias [2,3]. Hypoglycemia is also proinflammatory and can increase the risk of plaque inflammation, rupture, and CV events [4,5]. Thus, it is suggested that patients with long-standing T2DM, who presumably bear higher CV risks such as progressed atherosclerosis and/or existing CV diseases, may be more susceptible to the cardio toxicity that can be induced by hypoglycemia.
Intensive therapeutic regimens like multiple daily insulin injections combined with frequent self-monitoring of capillary blood glucose levels (SMBG) are believed to be one of the most useful methods of achieving good metabolic control. Such intensive regimens, however, can usually increase the incidence of hypoglycemia over time. Thus, it often becomes difficult to achieve optimal control [6], despite the use of multiple insulin injections and frequent SMBG, partly because of the limitations of the glycemic profile obtained from intermittent finger-sticks [7]. Furthermore, a high incidence of unrecognized hypoglycemia has been detected in diabetes patients treated with insulin [8]. Since the potential danger of these unrecognized bouts of hypoglycemia is well understood, one of the main objectives for glycemic control in patients with diabetes is to detect and prevent these events.
To overcome these limitations, a new sensor system for continuously measuring glucose concentrations in subcutaneous tissue, the continuous glucose monitoring system (CGMs), has come on the market and has revolutionized the monitoring of short-term glucose variability. This system has been quickly validated by a variety of clinical settings in many countries and has provided data showing a correlation between blood and interstitial glucose levels [9-11]. The CGMs has become a powerful system for the detection of unrecognized hypoglycemia, particularly at night and even between meals during the day.
Sodium glucose transporter (SGLT) 2 inhibitors is a new class of oral hypoglycemic agents that increase urinary glucose excretion by inhibiting SGLT2 in the renal proximal tubules, which reduces renal glucose reabsorption and decreases plasma glucose levels in an insulin-independent manner [12]. The glucose-lowering effect of SGLT2 inhibition seems to be self-limiting such that when the blood glucose (BG) concentration decreases to concentrations at which hypoglycemic symptoms are experienced, the remaining SGLT2 activity and the SGLT1 activity can reabsorb much of the filtered glucose, preventing a further loss of glucose. The mechanism of action relies on SGLT2 inhibitors that save either endogenous or exogenous insulin in patients who are treated with insulin secretagogue(s) and/or insulin injection [13]. Thus, we could expect the co-administration of SGLT2 inhibitor with an existing SU regimen would result in a reduction in the dose of SU without serious deterioration of glycemic control.
The aim of this study was to use CGMs to evaluate the efficacy and safety of administering canagliflozin to Japanese T2DM patients who are already receiving an existing regimen of SU, which could then be reduced by one-half. We assumed that the addition of SGLT2 inhibitors could be useful not only to reduce the dose of the preceding SUs, but also for a uniquely extensive and complex impact on metabolism and circulation.
Subjects, Materials and Methods
Patients and study design
This was a 12-week prospective, single-arm, open-label study to evaluate the efficacy, safety and tolerability of canagliflozin, added concomitantly with SUs, in Japanese T2DM patients who are already controlling their condition with SUs. The study was conducted from January 2015 to December 2015. This study was performed in accordance with the 2nd Helsinki Declaration and the International Conference on Harmonization/Good Clinical Practice Guidelines and was approved by the institutional ethics committee of Yodogawa Christian Hospital. The study was registered in the University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR, Japan) (ID: UMIN000022718) (https://upload.umin.ac.jp/cgi-open-bin/ctr/index.cgi?function=02). All participants freely provided informed consent in writing.
Inclusion and exclusion criteria
Inclusion criteria included the following: age range 20-80 years; type 2 diabetes treated with SU; and, HbA1c ≥ 6.5% (47 mmol/mol) and < 9.0% (75 mmol/mol) at the time of screening. Key exclusion criteria included an estimated glomerular filtration rate (eGFR) < 45 ml/min/1.73 m2 (using the MDRD equation); acute coronary syndrome, stroke, or transient ischemic attack within 3 months prior to consent; severe liver dysfunction.
Treatment and interventions
At the beginning of the study, the dose of previously prescribed SU, irrespective of the type and dose, was reduced by half and a concomitant administration of canagliflozin, 100 mg/day, was initiated. Canagliflozin was taken once daily after breakfast. Study visits were at the baseline and at weeks 4, 8, and 12 of treatment. Glycemic control (HbA1c, glycoalbumin) and other metabolic parameters were measured at the baseline and at weeks 4, 8, and 12 of treatment. Daily profiles of BG and glycemic variability were evaluated via a CGMs (iPro2, Medtronix, Dublin, Ireland) at the baseline and at week 12 of treatment with canagliflozin administration (Figure 1). CGMs measurement was based on enzymatic electrode method and was calibrated by SMBG conducted four times per day before each meal and the bedtime. Statistical analysis of the data was performed using a Wilcoxon and Dunn test.
Endpoints and assessments
The primary endpoints were the week-12 changes from the baseline values for HbA1c, glycoalbumin, fasting plasma glucose (FPG), and in the daily profiles of BG and glycemic variability, as evaluated via CGMs. The exploratory secondary endpoints were the week-12 values: changes from the baseline in weight, systolic blood pressure (SBP), diastolic blood pressure (DBP), homeostasis model assessment (HOMA)-R, HOMA-β, fasting CPR index, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), triglyceride (TG), free fatty acids (FFA), hematocrit (Ht), creatinine (Cr), uric acid, serum ketone bodies, and CRP. HOMA-R was calculated using the formula: HOMA-R = [glucose (mg/dl) x insulin (μU/mL)/405], using fasting values [14]. HOMA-β was calculated using the formula: 360 × insulin (μlU/ml)/fasting glucose (mg/dl) -63, using fasting values [14]. CRP was measured by latex agglutination turbidimetry. Safety endpoints included vital signs, clinical laboratory parameters, and adverse events (AEs). AEs included all events with an onset after the first dose and up to 7 days after the last dose of study medication. Confirmed hypoglycemic AEs were defined as AEs with plasma glucose ≤ 70 mg/dl and/or requiring assistance. Events consistent with urinary tract infection, genital infection, volume depletion, and hypersensitivity reactions were assessed.
Statistical analysis
Data are presented as the mean ± S.E.M. Statistical analysis was performed using Stat Flex ver. 6.0 software (Artech Co., Ltd, Osaka, Japan). Statistical significance was determined using the Wilcoxon and Dunn tests.
Results
Patient demographics and baseline characteristics
Of the 22 patients who gave their consent to participate, 21 began the run-in period. Four patients were withdrawn from the study, which included two follow-up failures and one deviation from protocol. One patient discontinued study treatment due to anxiety about the reduction in body weight and blood pressure. There were 17 patients who completed the study. Patient profiles, glycemic and other metabolic markers were gathered as part of the baseline, and these are shown in table 1. The mean age was 65.0 ± 11.9 years, the mean disease duration was 10.0 ± 4.4 years, the mean body mass index (BMI) was 28.0 ± 2.9 kg/m2, the mean HbA1c was 7.3 ± 0.3% (56 mmol/mol), and the mean fasting C-peptide index was 2.0 ± 0.5. All participants received glimepiride, and the baseline value for average dose was 1.0 ± 0.5 mg/day. Table 1 also shows the number of participants who received other antidiabetes agent(s) at the beginning of the study: insulin injection, 4; metformin, 14; thiazolidinedione, 12; dipeptidyl peptidase-4 (DPP-4) inhibitor, 10; alpha-glucosidase inhibitor, 1.
Clinical characteristics of the patients at the end of the study (Table 1)
Canagliflozin administration for 12 weeks resulted in significant reductions in HbA1c 7.3 ± 0.3% (56 mmol/mol) at the baseline to 7.1 ± 0.4% (53 mmol/mol) at 12 weeks, p < 0.05), glycoalbumin (20.0 ± 2.3 to 19.4 ± 2.4 %, P < 0.01), BMI (28.0 ± 2.9 to 27.4 ± 3.1 kg/m2, P < 0.01), SBP (135 ± 14 to 125 ± 11 mmHg, P < 0.05), and uric acid (5.3 ± 0.8 to 4.8 ± 0.6 mg/dl, P < 0.05). Ht was slightly increased only at 8 weeks. FFA was elevated moderately throughout the study, which presumably was indicative of lipolysis (Figure 2). There were no significant differences in lipid levels, FPG, HOMA-R, HOMA-β, fasting C-peptide index, Cr, or CRP.
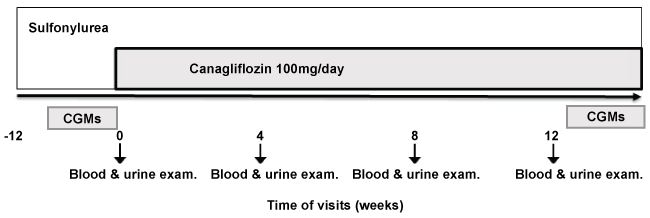
.
Figure 2: Changes in serum total ketone bodies and free fatty acid. Changes in serum total ketone bodies (μmol/l) from the individual subjects were plotted (solid line)and the average values of each time point were indicated in the bold line; the dotted line indicates the average serum free fatty acid levels (mEq/l). *p < 0.05 (Dunn test) vs. baseline.
View Figure 2
![]()
Table 1: Patients' characteristics: anthropometry and laboratory tests.
View Table 1
In order to gain insights into the relationship between SGLT2 inhibition and diabetes ketoacidosis (DKA), as well as for safety, we measured serum ketone bodies every 4 weeks during each hospital visit. Total ketone bodies were transiently elevated from 61 ± 47 to 162 ± 140 (p < 0.05) at 4 weeks but were decreased afterward (Table 1). In some patients, the level of serum ketone reached as high as several hundred μmol/L 4 to 8 weeks after the administration of canagliflozin, but it had returned to the baseline level by 12 weeks. Other patients, however, showed no significant changes throughout the study period (Figure 2).
CGMs analyses
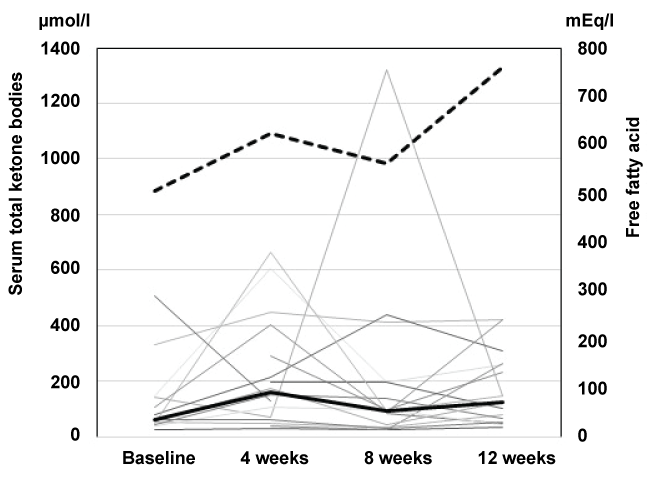
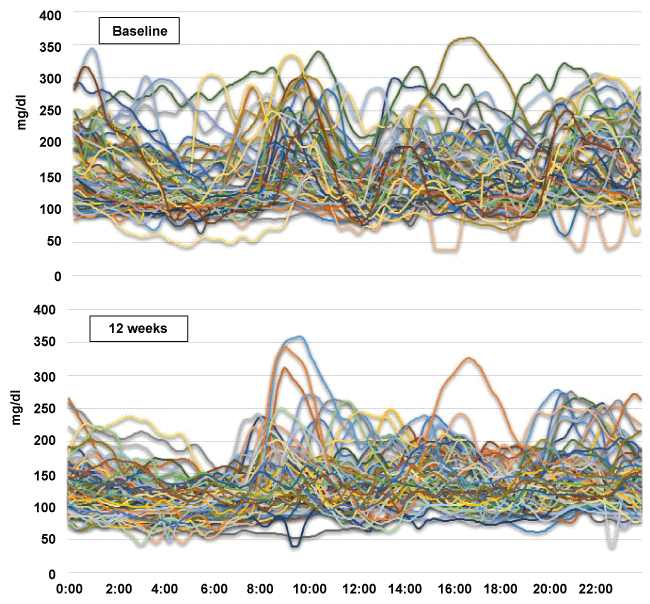
The changes in daily BG profiles are shown by plotting the raw data from the CGMs readings of all subjects at baseline and at week 12 of the treatment (Figure 3). Canagliflozin was kept administered at week 12 at the time of CGMs analysis (Figure1). The average duration of CGMs was 3.3 ± 1.4 days at baseline and 4.5 ± 1.5 days at week 12. The range of BG levels narrowed and the BG perturbation became less prominent at 12 weeks, compared with the baseline, showing a substantial improvement in glycemic profile throughout the day. By CGMs analyses, the average BG level (mg/dl) was reduced from 156.8 ± 23.5 to 129.4 ± 10.4 (p < 0.05), and SD was reduced from 45.7 ± 12.6 to 32.4 ± 9.0 (NS), and mean amplitude of glycemic excursions (MAGE) was reduced from 96.5 ± 30.6 to 73.4 ± 18.9 (NS) (Table 1). CGMs analyses also showed that the time spent at the target glucose level (70-180 mg/dl) was increased from 71 to 83% ( p < 0.05), and time above 180 mg/dl was shortened from 29 to 12% (p < 0.01) at 12 weeks, as shown figure 4. Taken together, the SU-canagliflozin combination was very useful in converging the daily glycemic perturbation into a very narrow range of BG levels throughout the day.
.
Figure 3: Raw data from the CGMs readings. Raw data from the CGMs readings of all subjects before (upper panel) and 12 weeks after canagliflozin administration (lower panel) were plotted for comparison.
View Figure 3
.
Figure 4: Distribution from the CGMs readings.
Proportion of CGMs readings above 180 mg/dl, in a target range of 70 to 180 mg/dl, and below 70 mg/dl, before (white bar) and 12 weeks after canagliflozin administration (hatched bar). *p < 0.01, #p < 0.05 (Wilcoxon test).
View Figure 4
Safety and tolerability
Events indicative of osmotic diuresis, thirst (n = 8) and polyuria (n = 5), were the most-frequent AEs. Urinalysis showed 3 patients to have urinary tract infections, but no kidney infections were reported. One patient developed infectious enteritis and exhibited ketosis, which was detected by an increase in serum ketone bodies as high as 1,322 μmol/L, at week 8 (Figure 2). The patient recovered after only a 2-day cessation of canagliflozin and eventually completed the study. One patient reported hypoglycemic symptoms, but the SMBG at the time of the episode was 80 mg/dl. Regarding hypoglycemia, CGMs analyses showed that the times below 70 mg/dl and below 55 mg/dl had not changed significantly either before or after the study period (Figure 4). Changes in the laboratory parameters are shown in table 1. There were no significant changes in the Ht, eGFR, or urine albumin-to-creatinine ratio. No other serious adverse events were observed during the study period.
Discussion
In this 12 week prospective study, we showed that an administration of canagliflozin after a reduced dose of glimepiride improved both average glycemic parameters and daily BG profiles. Since we assumed that SGLT2 inhibitor added to an existing prescription that included SU might enhance the risk of hypoglycemia, we planned to cut the dose of SU by half at the initiation of a concomitant application of SGLT2 inhibitor. The combination of canagliflozin with a half-dose of glimepiride resulted in better glycemic control, as shown by the improvement in HbA1c and glycoalbumin, without increasing the frequency of hypoglycemia. These results suggest a good starting point for general practitioners when they intend to add SGLT2 inhibitors to a previous prescription such as SU that requires a dosage adjustment for safety reasons.
By using CGMs analyses, we found that the concomitant administration of canagliflozin improved not only the markers for long-span glycemic control (i.e. HbA1c and glycoalbumin) but also improved the daily glycemic profile. These results are essentially similar to those in previous reports that have investigated the monotherapy of SGLT2 inhibitors and the combination therapy with SGLT2 inhibitors to show significant improvement in glucose perturbations in T2DM patients [15-17].
More importantly, this study showed that a SU-canagliflozin combination could have a favorable effect on daily glycemic control without increasing hypoglycemia. We also realized that CGMs analyses could be very efficient in detecting unrecognized hypoglycemia in some patients, particularly on the night of the baseline and even at the end of a study period (data not shown). The CV toxicity of hypoglycemia (see Introduction) makes it crucial to achieve better glycemic control while avoiding hypoglycemia in the clinical treatment of T2DM patients. This is attributable to a reduction in the dose of SU and also to the properties of SGLT2 inhibitors that reduce the potential to induce hypoglycemia when used alone or to resist hypoglycemia even when used in combination with insulin injection or the administration of insulin secretagogue(s). The latter includes diminished SGLT2 activity in the proximal tubules, which is caused by the physiological decline in GFR during sympathetic nervous system activation from hypoglycemia [18] and by a putative compensatory increase in hepatic gluconeogenesis from the increase in glucagon secretion, which is further enhanced by SGLT2 inhibition [19]. In addition, a diminished paracrine inhibition by insulin [20] and decreased SGLT2 mediated glucose transport into β-cells [19] are also suggested to be among the mechanisms that elevate plasma glucagon concentrations in patients taking SGLT2 inhibitors.
Recently, the U.S. Food and Drug Administration (FDA) issued a Drug Safety Communication, warning of the increased risk of DKA associated with the use of SGLT2 inhibitors, based on a search of the U.S. FDA Adverse Event Reporting System (FAERS), [21]. Published and preliminary data from the large clinical studies, which includes the Empagliflozin Cardiovascular Outcome Event Trial study (EMPA-REG), the canagliflozin cardiovascular Assessment Study (CANVAS), and the Dapagliflozin Effect on Cardiovascular Events (DECLARE), indicated that the frequency of reported events suggestive of DKA is less than 0.1% in T2DM participants. This compares to the frequency observed for general T2DM patients who have not been treated with SGLT2 inhibitors [22]. In this sense, the FDA Communication might be somewhat of an exaggeration by suggesting that DKA can often develop in SGLT2-treated T2DM patients. However, a more likely scenario could be that these reported DKA cases were insulin-treated or insulin-deficient T2DM patients or type 1 diabetes, as was suggested by Rosenstock D and co-workers [22].
In this study, we measured total ketone bodies in the patients' blood every 4 weeks after the initiation of canagliflozin and found that ketone bodies were elevated transiently as high as several hundred μmol/L at 4 to 8 weeks after canagliflozin add-on. That would be far below the standard ketone level (usually several thousands) of DKA development in T2DM patients. The transient elevation of total ketone bodies seen in some patients (Figure 2) might reflect a rapid and drastic decline in BG level after the addition of a SGLT2 inhibitor, accompanied by lipolysis that is obviously enhanced by the elevation of glucagon, along with subsequent metabolic compensation. On the other hand, it would not be overstatement to suggest that SGLT2 inhibitors are, among the list of anti-diabetes medications, the most ketogenic class of anti-diabetes since this drug promotes the excretion of a large portion of glucose into urine, and easily invites relative deficiencies in the sources of cellular energy. Because facilitative glucose transport from the blood stream into muscle and fat cells requires sufficient glucose and workable insulin, relative deficiencies in BG and/or viable insulin should readily boot a compensatory program to synthesize ketone bodies as alternative cellular energy sources at the expense of lipolysis. Thus, we designed this study initially not to withdraw SU totally but to cut the dose of SU by half at the time canagliflozin is added because we intended to secure at least some workable insulin for the coming glucose-deficient state. As a result, this study may suggest the very important notion that we should be very careful not to discontinue the already-prescribed insulin secretagogue(s) or insulin injection totally because of an excessive fear of hypoglycemia, but for sufficient prevention of ketogenesis when SGLT2 inhibitor is added. In this sense, the strategy of combination therapy for T2DM patients constituted by insulin secretagogue(s) or insulin and SGLT2 inhibitor requires careful steering between the ketogenesis that is developed by excessive deficiencies in both glucose and workable insulin, and the hypoglycemia that is obviously provoked by too much insulin.
Global clinical studies that are expanding the use of SGLT2 inhibitors for type 1 diabetes mellitus (T1DM) patients are ongoing [23], and these are based on many pilot studies using a variety of SGLT2 inhibitors concomitantly with multiple insulin injection therapy and/or continuous subcutaneous insulin injection in T1DM patients. These have successfully shown better glycemic control, significant reduction in insulin dosage, and reductions in body weight without increasing hypoglycemia [24]. However, it should be emphasized that sufficient care and constant surveillance for the development of ketosis and ketoacidosis is warranted when using SGLT2 inhibitors in T1DM patients.
In conclusion, we showed that the addition of canagliflozin following a reduction in the dose of an existing regimen of SU in T2DM patients was tolerable and significantly improved glycemic control and daily glucose perturbation without increasing the risk of hypoglycemia and ketone production. Long-term studies will be required to establish the efficacy and safety of such a combination strategy.
Contributor Statements and Conflicts of Interest
M. Harada conducted the study, analyzed the data, and wrote the manuscript. Y.A, Y.M, and M.K. recruited patients and contributed to the discussion. M. Hashiramoto designed and conducted the study, analyzed the data, and wrote and edited the manuscript.
M. Hashiramoto has received lecture fees from the Takeda Pharmaceutical Company Ltd. and from the Mitsubishi Tanabe Pharma Corporation. No other potential conflicts of interest relevant to this article were reported.
References
-
Mannucci E, Monami M, Lamanna C, Gori F, Marchionni N (2009) Prevention of cardiovascular disease through glycemic control in type 2 diabetes: a meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis 19: 604-612.
-
Marques JL, George E, Peacey SR, Harris ND, Macdonald IA, et al. (1997) Altered ventricular repolarization during hypoglycaemia in patients with diabetes. Diabet Med 14: 648-654.
-
Adler GK, Bonyhay I, Failing H, Waring E, Dotson S, et al. (2009) Antecedent hypoglycemia impairs autonomic cardiovascular function: implications for rigorous glycemic control. Diabetes 58: 360-366.
-
Dotson S, Freeman R, Failing HJ, Adler GK (2008) Hypoglycemia increases serum interleukin-6 levels in healthy men and women. Diabetes Care 31: 1222-1223.
-
Packard RR, Libby P (2008) Inflammation in atherosclerosis: from vascular biology to biomarker discovery and risk prediction. Clin Chem 54: 24-38.
-
American Diabetes Association (1996) Self monitoring of blood glucose (Consensus Statement). Diabetes Care 19 : 62-66.
-
Boland EA, Monsod T, Delucia M, Brand CA, Fernando S, et al. (2001) Limitations of conventional methods of self monitoring of blood glucose: lessons learned from 3 days of continuous glucose sensing in pediatric patients with type 1 diabetes. Diabetes Care 24: 1858-1862.
-
Bolli GB, Perriello G, Fanelli CG, De Feo P (1993) Nocturnal blood glucose control in type 1 diabetes mellitus. Diabetes Care 16: 71-89.
-
Rebrin K, Steil GM, Van Antwerp WP, Mastrototaro JJ (1999) Subcutaneous glucose predicts plasma glucose independent of insulin: implications for continuous montoring. Am J Physiol 277: E561-571.
-
Schmidt F, Sluiter WJ, Schoonen AJM (1993) Glucose concentration in subcutaneous extracellular space. Diabetes Care 16: 695-700.
-
Sternberg F, Meyerhoff C, Mennel FJ, Bischof F, Pfeiffer EF (1995) Subcutaneous glucose concentration in humans: real estimation and continuous monitoring. Diabetes Care 18: 1266-1269.
-
Abdul-Ghani MA, DeFronzo RA (2008) Inhibition of renal glucose reabsorption: a novel strategy for achieving glucose control in type 2 diabetes mellitus. Endocr Pract 14: 782-790.
-
Abdul-Ghani MA, DeFronzo RA, Norton L (2013) Novel hypothesis to explain why SGLT2 inhibitors inhibit only 30-50% of filtered glucose load in humans. Diabetes 62: 3324-3328.
-
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, et al. (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28: 412-419.
-
Kentaro Yamada, Hitomi Nakayama, Satoko Yoshinobu, Seiko Kawano, Munehisa Tsuruta, et al. (2015) Effects of a sodium glucose co-transporter 2 selective inhibitor, ipragliflozin, on the diurnal profile of plasma glucose in patients with type 2 diabetes: A study using continuous glucose monitoring. J Diabetes Investig 6: 699-707.
-
Rimei Nishimura, Yuko Tanaka, Koiwai K, Inoue K, Hach T, et al. (2015) Effect of empagliflozin monotherapy on postprandial glucose and 24-hour glucose variability in Japanese patients with type 2 diabetes mellitus: a randomized, double-blind, placebo-controlled, 4-week study. Cardiovasc Diabetol 14: 11.
-
Yuta Nakamura, Yoshiko Nagai, Terashima Y, Nishine A, Ishii S, et al. (2015) Better response to the SGLT2 inhibitor dapagliflozin in young adults with type 2 diabetes. Expert Opin Pharmacother 16: 2553-2559.
-
Patrick AW, Hepburn DA, Swainson CP, Frier BM (1992) Changes in renal function during acute insulin-induced hypoglycaemia in patients with type 1 diabetes. Diabet Med 9: 150-155.
-
Bonner C, Kerr-Conte J, Gmyr V, Queniat G, Moerman E, et al. (2015) Inhibition of the glucose transporter SGLT2 with dapagliflozin in pancreatic alpha cells triggers glucagon secretion. Nat Med 21: 512-517.
-
Maruyama H, Hisatomi A, Orci L, Grodsky GM, Unger RH (1984) Insulin within islets is a physiologic glucagon release inhibitor. J Clin Invest 74: 2296-2299.
-
U.S. Food and Drug Administration. Drug Safety Communication: FDA warns that SGLT2 inhibitors for diabetes may result in a serious condition of too much acid in the blood.
-
Rosenstock J, Ferrannini E (2015) Euglycemic Diabetic Ketoacidosis: A Predictable, Detectable, and Preventable Safety Concern With SGLT2 Inhibitors. Diabetes Care 38: 1638-1642.
-
Henry RR, Rosenstock J, Edelman S, Mudaliar S, Chalamandaris AG, et al. (2015) Exploring the potential of the SGLT2 inhibitor dapagliflozin in type 1 diabetes: a randomized, double-blind, placebo-controlled pilot study. Diabetes Care 38: 412-419.
-
Argento NB, Nakamura K (2016) Glycemic Effects of Sglt-2 Inhibitor Canagliflozin in Type 1 Diabetes Patients Using the Dexcom G4 Platinum Cgm. Endocr Pract 22: 315-322.