International Journal of Cancer and Clinical Research
In Vitro Reaction of Cells Derived from Human Normal Lung Tissues to Carbon-Ion Beam Irradiation
Naoko Okano1, Takahiro Oike1, Jun-ichiSaitoh1*, KatsuyukiShirai2, Masato Enari3, TohruKiyono4, MayuIsono2, Kota Torikai2, Tatsuya Ohno2 and Takashi Nakano1
1Department of Radiation Oncology, Gunma University Graduate School of Medicine, Japan
2Gunma University Heavy Ion Medical Center, Japan
3Division of Refractory Cancer Research, National Cancer Center Research Institute, Japan
4Division of Virology, National Cancer Center Research Institute, Japan
*Corresponding author:
Jun-ichiSaitoh, MD, PhD, Department of Radiation Oncology, Gunma University Graduate School of Medicine, 3-s39-22, Showa-machi, Maebashi, Gunma, 371-8511, Japan, Fax: +81-27-220-2897, E-mail: junsaito@gunma-u.ac.jp
Int J Cancer Clin Res, IJCCR-4-078, (Volume 4, Issue 1), Original Article; ISSN: 2378-3419
Received: March 01, 2017 | Accepted: March 28, 2017 | Published: March 30, 2017
Citation: Okano N, Oike T, Jun-ichi S, Shirai K, Enari M, et al. (2017) In Vitro Reaction of Cells Derived from Human Normal Lung Tissues to Carbon-Ion Beam Irradiation. Int J Cancer Clin Res 4:078. 10.23937/2378-3419/1410078
Copyright: © 2017 Okano N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Radiation-induced lung injury (RILI) is a serious concern in carbon-ion radiation therapy (CIRT) for thoracic malignancies. To estimate the induction of RILI after CIRT, translation of evidence in X-ray radiation therapy to CIRT is of great importance. However, the relative biological effectiveness (RBE) of carbon-ion beams in normal lung tissues is not fully elucidated, making the translation difficult. This is in part due to the absence of in vitro assays that can determine the RBE values in normal cells lacking clonogenic ability.
Materials and methods: Immortalized human small airway epithelial cells (iSAECs), human lung fibroblasts HFL-I, and A549 lung cancer cellswere irradiated with X-rays or carbon-ion beams, cultured for 10 days, fixed with methanol, and then subjected to crystal violet staining (CVS). The stained cells were solubilized and the absorbance of the solutions was measured using a spectrophotometer. The data plots were fitted to the linear-quadratic model to generate survival curves.
Results: The absorbance values in the CVS assay were highly correlated withthe number of colonies in the clonogenic survival assay for A549 cells (R = 0.905, P < 0.01), resulting in strong association between the survival curves generated using the two assays. In iSAECs, the D50 values for X-ray and carbon-ion beam irradiation were 8.3 Gy and 2.6 Gy, respectively, and the RBE was 3.2. In HFL-I cells, the D50 values were 3.3 Gy and 1.5 Gy, respectively, and the RBE was 2.2.
Conclusion: The CVS assay may provide an alternative to the clonogenic survival assay for the assessment of the RBE of carbon ion beam irradiationof normal cells lacking clonogenic ability, and will help understand the biological effect of CIRT on human normal lung tissues.
Keywords
Relative Biological Effectiveness (RBE), Carbon-ion beam, Normal lung tissue, Surviving fraction, Colony forming ability
Introduction
The standard treatment for inoperable early-stage non-small cell lung carcinoma (NSCLC) is radiotherapy. In such cases, X-ray stereotactic body radiation therapy (SBRT), which can deliver highly conformal dose to the tumor, shows comparable local control to surgery [1,2]. Carbon-ion beam radiation therapy (CIRT) also shows local control comparable to SBRT and surgery for early-stage NSCLC (i.e., 5 year local control rate of > 90%) [3-5]. Carbon-ion beams have two important properties that contribute to favorable clinical outcomes: a superior dose distribution associated with the sharp penumbra and distal tail-off achieved by the Bragg peak, and stronger cancer-cell-killing activity than X-rays [6,7].
Radiation-induced lung injury (RILI) is lethal in some cases, and is a major dose-limiting factor in thoracic irradiation therapy [8]. Previous studies on X-ray SBRT have demonstrated several predictive factors for RILI, e.g., normal lung volume receiving 20 Gy (V20) ˃ 10% and V25 ˃ 4.2% [9-11]. Meanwhile, there have been no established predictive factors for RILI in CIRT. In this regard, comparison of dose-volume parameters as noted above, between SBRT and CIRT cases, is of great importance for estimating the induction of RILI after CIRT. However, the relative biological effectiveness (RBE) of carbon-ion beams in normal lung tissues is largely unknown. It is unclear whether the RBE in normal lung tissues is higher or lower than the clinically-used value based on the in vitro RBE in the human salivary gland cancer cell line HSG [12,13], which makes it difficult to compare the dose-volume parameters between SBRT and CIRT of the lung.
RBE has been evaluated using various in vitro and in vivo experimental methods with different endpoints including cell survival, apoptosis, chromosome aberration, DNA repair capacity in vitro, and tumor shrinkage in vivo [14-22]. The clonogenic survival assay is one of the standard methods for determining RBE [12,13,23-26] because it can be easily performed in vitro, and the results are significantly correlated with tumor response to radiotherapy in the clinic [27]. However, the clonogenic survival assay cannot be used to determine the surviving fraction in cell lines derived from human normal lung tissues because they lack clonogenic ability. Taken together, in the present study, we sought to investigated the RBE of carbon-ion beams in cell lines derived from human normal lung tissues using a crystal violet staining assay (CVS assay) in which the surviving fraction was determined by measuring the absorbance of a solution containing fixed, stained, and lysed cells [28].
Materials and Methods
Cell lines
Small airway epithelial cells (SAEC) isolated from human bronchi, the human embryonic lung fibroblast line HFL-I, and the human lung adenocarcinoma cell line A549 were purchased from Lonza (Basel, Switzerland), Riken Bioresource Center (Ibaraki, Japan) and ATCC (VA, USA), respectively. SAEC cells were cultured in small airway epithelial basal medium supplemented with SAGM Single Quots (Lonza). HFL-I and A549 cells were cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum, 100 U/ml penicillin and 100 μg/ml streptomycin (Life Technologies, CA, USA).
SAECs have a limited proliferative ability and cannot be stably cultured for more than 20 population doublings; therefore, appropriate assessment of radiosensitivity in these cells is impossible. Thus, to enable the continuous culture of SAECs, an immortalized cell line was established as follows: the lentiviral vector plasmids CSII-CMV-cyclinD1, -CDK4R24C and -TERT were constructed by recombination using the Gateway system (Invitrogen, CA, USA), as described previously [29]. Briefly, the cDNAs of human cyclinD1, a mutant form of human CDK4 (CDK4R24C: an inhibitor-resistant form of CDK4, generously provided by Dr. Hara) and human TERT were recombined with a lentiviral vector, CSII-CMV-RfA (a gift from Dr. Miyoshi), by LR reaction to create a Gateway expression plasmid (Invitrogen) according to the manufacturer's instructions. Lentiviral combinations were used to transduce SAEC cells with cyclinD1, CDK4R24C and TERT, and cells were grown without drug selection to generate immortalized SAECs (iSAECs).
The average doubling time for iSAECs, HFL-I and A549 was approximately 48 h, 36 h and 24 h, respectively. For all the cell lines, the cells at passage number from 5 to 10 were used.
Irradiation
X-ray irradiation was performed using a Faxitron RX-650 (100 kVp, 1.14 Gy/min, Faxitron Bioptics, AZ, USA). Carbon-ion beam irradiation was performed at the Gunma University Heavy Ion Medical Center using the same beam specification as those used in the clinic (290 MeV/nucleon, the average linear energy transfer at the center of a 6 cm spread-out Bragg peak of approximately 50 keV/μm).
Assessment of cell survival by the crystal violet staining assay (CVS assay)
The sensitivity of iSAECs and HFL-I cells to X-ray or carbon-ion beam irradiation was determined using an assay described by Guzzi, et al. [28] with a minor modification. Cells were seeded on 60 mm dishes (10,000 cells/dish), incubated at 37 °C for 2 days, subjected to X-ray or carbon-ion beam irradiation and incubated at 37 °C for an additional 10 days. Cells were then fixed in 25% methanol, stained with 0.1% crystal violet, and solubilized in 2 ml of 10% acetic acid. The absorbance of the solution at 570 nm was measured with a Multiskan FC (Thermo Fisher Scientific, NY, USA). The D10 and D50 (doses required to reduce the absorbance to 10% and 50%, respectively) were calculated by fitting the absorbance to the linear-quadratic (LQ) model, as follows: absorbance = exp (-αD-βD2), where D is the dose [30].
Clonogenic survival assay
The surviving fractions of A549 cells exposed to X-rays were assessed using the clonogenic survival assay, as previously described [30]. To compare the surviving fractions calculated by the clonogenic survival assay and the absorbance values calculated by the CVS assay in the same samples, after colony counting, the colonies were solubilized in 10% acetic acid and subjected to absorbance measurements according to the protocol of the CVS assay.
Senescence-associated β-galactosidase staining
Senescence-associated β-galactosidase (SA-β-gal) activity in cells exposed to X-rays or carbon-ion beams was assessed using a Senescence β-galactosidase Staining Kit (Cell Signaling Technology, MA, USA) 10 days after irradiation. Blue-stained cells observed by light microscopy were considered positive for the staining. In each experimental condition, at least 300 cells were counted in triplicate.
Statistical analysis
The correlation between the number of colonies in the clonogenic survival assay and the absorbance in the CVS assay in the same samples of irradiated A549 cells was analyzed by Pearson's correlation coefficient. The significance of differences in the percentages of senescent cells was determined by the unpaired two-tailed Student's t-test. P < 0.05 was considered significant. Experiments were performed in triplicate, at least.
Results
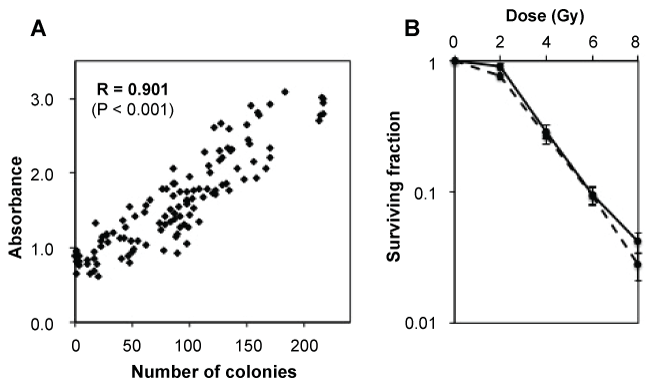
iSAECs and HFL-I cells did not form colonies although they could continuously grow on culture dishes. We therefore examined the radiosensitivity of these cells using the CVS assay [28]. Prior to the assessment of RBE, association between the results of the CVS assay and the clonogenic survival assay was evaluated by comparing the absorbance values assessed by the CVS assay to the number of colonies assessed by clonogenic survival assay in the same set of X-ray-irradiated A549 lung cancer cells, which have clonogenic ability. The absorbance values obtained with the CVS assay were highly correlated to the number of colonies in the clonogenic survival assay (R = 0.905, P < 0.01) (Figure 1A), resulting in strong association between the survival curves generated using the two assays (Figure 1B). These data indicate that the CVS assay can be an alternative to the clonogenic survival assay in the assessment of radiosensitivity.
.
Figure 1: Association between the results of the CVS assay and those of the clonogenic survival assay. (A) Correlation between the number of colonies in the clonogenic survival assay and the absorbance values in the CVS assay in the same samples of irradiated A549 cells (n = 120). The correlation was analyzed by using Pearson correlation coefficient; (B) Representative survival curves obtained from the clonogenic survival assay (solid line) and the CVS assay (dotted line) (n = 3, each). Average ± SD is shown.
View Figure 1
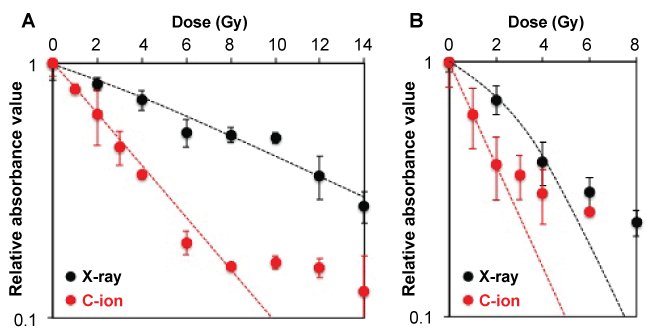
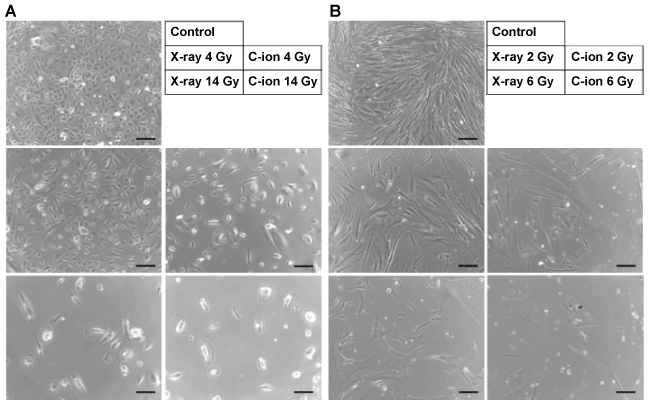
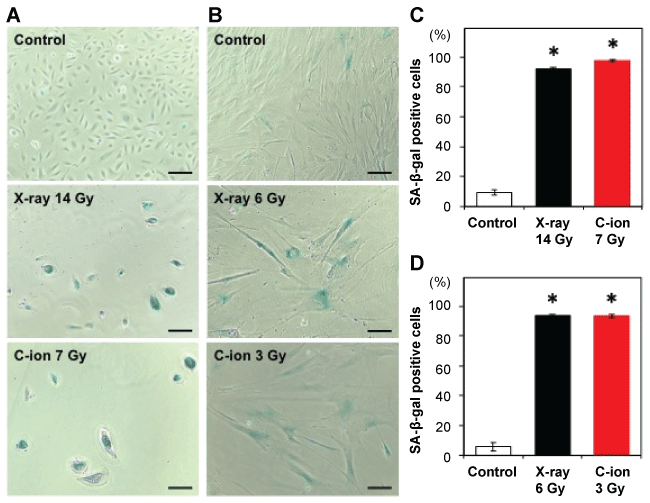
Assessment of the sensitivity of iSAECs and HFL-I cells to irradiation by the CVS assay (Figure 2) showed that at low doses, X-ray or carbon-ion beam irradiation decreased the absorbance values in a dose-dependent manner. However, the gradient of the fitting curve decreased with increasing doses and the absorbance values could not be fitted to the LQ model (10-14 Gy in iSAECs receiving carbon-ion beam irradiation, and 6-8 Gy and 3-6 Gy in HFL-I cells receiving X-ray and carbon-ion beam irradiation, respectively). On microscopic observation, the numbers of iSAECs and HFL-I cells exposed to high doses of X-rays or carbon-ion beams were lower than those estimated by the CVS assay (Figure 2). Furthermore, surviving cells after high-dose irradiation lost proliferative ability over time and showed enlarged morphology (Figure 3). Since enlarged morphology with lost proliferative ability is characteristic of cells undergoing senescence, which is one of the major modes of proliferative cell death associated with ionizing irradiation [30-35], the induction of senescence in iSAECs and HFL-I cells receiving high-dose X-ray or carbon-ion beam irradiation was investigated. The number of SA-β-gal positive iSAECs and HFL-I cells increased significantly 10 days after irradiation with high doses of X-rays or carbon-ion beams (Figure 4). These findings indicate that the high staining intensity of the samples could be attributed to senescent cells with enlarged bodies that accumulated on the culture plates in response to high-dose irradiation, leading to the overestimation of the surviving fraction in the CVS assay.
.
Figure 2: Sensitivity of iSAECs and HFL-I cells to X-ray and carbon-ion beam irradiation assessed by using the CVS assay. (A) iSAECs; (B) HFL-I. Average ± SD is shown. The fitting curves were generated based on the linear-quadratic model using the data obtained at the dose ranges listed in Table 1. C-ion, carbon-ion beams.
View Figure 2
.
Figure 3: Representative micrographs of iSAECs and HFL-I cells exposed to X-ray and carbon-ion beam irradiation. (A) iSAECs; (B) HFL-I. Micrographs taken 10 days after irradiation are shown. Scale bar, 100 μm. C-ion, carbon-ion beams.
View Figure 3
.
Figure 4: Induction of senescence by X-ray and carbon-ion beam irradiation of iSAECs and HFL-I cells. The cells were exposed to X-ray or carbon-ion beam irradiation and then assessed using the senescence-associated β-galactosidase (SA-β-gal) staining 10 days after irradiation. (A,B) Representative micrographs of iSAECs (A) and HFL-I (B) cells. SA-β-gal positive cells are stained in blue. Scale bar, 100 μm; (C,D) The percentage of SA-β-gal positive iSAECs (A) and HFL-I cells (B). Average ± SD is shown. Asterisks indicate significant differences (P < 0.001) compared with the non-irradiated controls. C-ion, carbon-ion beams.
View Figure 4
Previous studies have demonstrated that the surviving fraction of irradiated normal cells can be fitted to the LQ model in vitro [12]. Hence, in the present study, we employed a dose range in which the absorbance values could be fitted to the LQ model to calculate the RBE (0-14 Gy and 0-8 Gy in iSAECs, and 0-4 Gy and 0-2 Gy in HFL-I cells for X-ray and C-ion beam irradiation, respectively). Among these dose ranges, the D10 and D50 which have been shown to be reliable in previous studies, were used as endpoints to determine the RBE [22-26,36]. The calculated D10, D50 and the RBE values are shown in Table 1. In iSAECs, the RBE values at D10 and D50 were 2.4 and 3.2, respectively. In HFL-I cells, the surviving fraction in the < 40% region could not be fitted to the LQ model, indicating that the D10 could not be obtained reliably. The RBE in HFL-I cells at D50 was 2.2.
![]()
Table 1: D10, D50 and RBE in iSAEC and HFL-I cells.
View Table 1
Discussion
The results of the present study demonstrate strong association between the survival curves generated by the CVS assay and those generated by the clonogenic survival assay for A549 cancer cells. Using the CVS assay, the RBE in normal lung cells lacking clonogenic ability could be calculated at least at D50.
In Japan, the irradiation dose used in CIRT is based on the RBE value obtained using HSG cells, a human salivary gland cancer line [12,13]. However, previous studies have demonstrated that RBE varies depending on the histological type [14], highlighting the difficulty in using RBE values generated using HSG cells for determining the optimal radiation dose in CIRT for each type of cancer. To overcome this problem, considerable research has been expended in determining the RBEs of CIRT in different cancer and normal tissue types to establish optimal CIRT treatment planning strategies. Since most cell lines derived from human normal tissues lack clonogenic ability, determination of RBE in human normal tissues using the clonogenic survival assay has been difficult. Furthermore, to the best of our knowledge, currently there is no consensus on which in vitro assays provide data broadly in association with those of the clonogenic survival assay. The results of the present study provide insights into these issues.Although the RBE at D10 determined using the CVS assay was not considered reliable because iSAECs and HFL-I cells enter senescence at high doses of irradiation, RBE at D50, where the surviving fractions could be fitted the LQ model, may be more reliable for the determination of RBE using this assay. Besides, in the present study, the D50 of iSAECs and HFL-I cells is within the dose range used in fractionated radiotherapy in the clinic, indicating that D50 may be an appropriate endpoint for RBE in the CVS assay. Since the CVS assay is simple and convenient, it can be used to screen the radiosensitivity of cells derived from a panel of normal organs, as well those of biopsy samples.
A549 cells irradiated with high doses of X-ray or carbon-ion beams mostly floated in the culture media. On the other hand, most iSAECs and HFL-I cells irradiated with such high doses showed senescence and remained attached to the culture dishes (Figure 3).The differences in the mechanisms of cell death between cancer and non-cancerous cells might affect the different absorbance values in the high-dose region of the CVS assay. Previously, we reported that, after X-ray irradiation for the D10, apoptosis, necrosis and mitotic catastrophe were induced in A549 cancer cells while senescence was less frequently induced [37]. In contrast, in another fibroblast line HFL-III, X-ray irradiation for the D10 predominantly induced senescence. Thus, predominant induction of senescence but not apoptosis, necrosis and mitotic catastrophe resulting in the floating of the cells, may be a common nature of normal cells after irradiation. This should be considered in assessing the radiosensitivity of normal cells.
With HFL-1 cells, survival curves did not fit the linear-quadratic model at doses higher than 2 Gy (Figure 2B). This is probably because fibroblasts responded to ionizing irradiation by entering senescence (Figure 4B and Figure 4D). Since senescent cells remain attached to culture dishes, surviving fractions estimated by the CVS assay may be overestimated, resulting in inconsistency with the linear-quadratic model. Taken together, our results indicate that care should be taken when assessing the in vitro radiosensitivity of fibroblasts at high doses of irradiation.
The limitation of the present study was as follows. Since SAECs have limited proliferative ability, it was necessary to immortalize them to determine their radiosensitivity. The immortalization process included transduction with human cyclinD1, CDK4 and TERTcDNAs. CyclinD1 and CDK4 are involvedin cell cycle regulation whereas TERT is involvedin telomere function. Therefore, there exists the possibility that the changes induced in these key proliferative pathways by these transductions could have affected iSAEC responses to ionizing radiation. However, it should also be noted that there was no significant difference in the cell doubling time between SAECs and iSAECs (data not shown).
In summary, we showed the possibility that the CVS assay could provide an alternative to the clonogenic survival assay for the assessment of RBE in normal cells lacking clonogenic ability. By using this method, the RBE values in iSAECs at D10 and D50 were calculated as 2.4 and 3.2, respectively; the RBE value in HFL-I cells at D50 was 2.2. Further investigation is necessary to obtain a comprehensive understanding of the risks and benefits of CIRT for thoracic malignancies.
Acknowledgement
The authors thank Dr. Miyoshi of Riken BioResource Center for providing lentiviral vector CSII-CMV-RfA. This work was supported by Grants-in-Aid from the Japan Society for the Promotion of Science for Young Scientists (B) KAKENHI [25861069] and [10643471].
References
-
Grills IS, Mangona VS, Welsh R, Chmielewski G, McInerney E, et al. (2010) Outcomes after stereotactic lung radiotherapy or wedge resection for stage I non-small-cell lung cancer. J Clin Oncol 28: 928-935.
-
Onishi H, Shirato H, Nagata Y, Hiraoka M, Fujino M, et al. (2011) Stereotactic body radiotherapy (SBRT) for operable stage I non-small-cell lung cancer: can SBRT comparable to surgery? Int J Radiat Oncol Biol Phys 81: 1352-1358.
-
Miyamoto T, Yamamoto N, Nishimura H, Koto M, Tsujii H, et al. (2003) Carbon ion radiotherapy for stage I non-small cell lung cancer. Radiother Oncol 66: 127-140.
-
Miyamoto T, Baba M, Yamomoto N, Koto M, Sugawara T, et al. (2007) Curative treatment of stage I non-small -cell lung cancer with carbon ion beams using hypofractionated regimen. Int J Radiat Oncol Biol Phys 67: 750-758.
-
Miyamoto T, Baba M, Sugane T, Nakajima M, Yashiro T, et al. (2007) Carbon ion radiotherapy for stage I non-small cell lung cancer using a regimen of four fractions during 1 week. J Thorac Oncol 2: 916-926.
-
Kraft G (1998) Radiotherapy with heavy ions: radiobiology, clinical indications and experience at GSI, Darmstadt. Tumori 84: 200-204.
-
Ohno T (2013) Particle radiotherapy with carbon ion beams. EPMA J 4: 9.
-
Yamashita H, Nakagawa K, Nakamura N, Koyanagi H, Tago M, et al. (2007) Exceptionally high incidence of symptomatic grade 2-5 radiation pneumonitis after stereotactic radiation therapy for lung tumors. Radiat Oncol 2: 21.
-
Barriger RB, Forquer JA, Brabham JG, Andilino DL, Shapiro RH, et al. (2012) A dose-volume analysis of radiation pneumonitis in non-small cell lung cancer patients treated with stereotactic body radiation therapy. Int J Radiat Oncol Biol Phys 82: 457-462.
-
Matsuo Y, Shibuya K, Nakamura M, Narabayashi M, Sakanaka K, et al. (2012) Dose-volume metrics associated with radiation pneumonitis after stereotactic body radiation therapy for lung cancer. Int J Radiat Oncol Biol Phys 83: 545-549.
-
Takeda A, Ohashi T, Kunieda E, Enomoto T, Sanuki N, et al. (2010) Early graphical appearance of radiation pneumonitis correlates with severity of radiation pneumonitis after stereotactic body radiotherapy (SBRT) in patients with lung tumors. Int J Radiat Oncol Biol Phys 77: 685-690.
-
Kanai T, Endo M, Minohara S, Miyahara N, Koyama-Ito H, et al. (1999) Biophysical characteristics of HIMAC clinical irradiation system for heavy-ion radiation therapy. Int J Radiat Oncol Biol Phys 44: 201-210.
-
Kagawa K, Murakami M, Hishikawa Y, Abe M, Akagi T, et al. (2002) Preclinical biological assessment of proton and carbon ion beams at Hyogo ion beam medical center. Int J Radiat Oncol Biol Phys 54: 928-938.
-
Ando K, Kase Y (2009) Biological characteristics of carbon-ion therapy. Int J Radiat Biol 85: 715-728.
-
Sasaki H, Yatagai F, Kanai T, FurusawaY, Hanaoka F, et al. (1997) Dependence of induction of interphase death of Chinese hamster ovary cells exposed to accelerate heavy ions on linear energy transfer. Radiat Res 148: 449-454.
-
Aoki M, Furusawa Y, Yamada T (2000) LET dependency of heavy-ion induced apoptosis in V79 cells. J Radiat Res 41: 163-175.
-
Kawata T, Durante M, Furusawa Y, George K, Takai N, et al. (2001) Dose--response of initial G2-chromatid breaks induced in normal human fibroblasts by heavy ions. Int J Radiat Biol 77: 165-174.
-
Monobe M, Ando K (2002) Drinking beer reduces radiation-induced chromosome aberrations in human lymphocytes. J Radiat Res 43: 237-245.
-
George K, Durante M, Willingham V, Wu H, Yang TC, et al. (2003) Biological effectiveness of accelerated particles for the induction of chromosome damage measured in metaphase and interphase human lymphocytes. Radiat Res 160: 425-435.
-
Takahashi A, Matsumoto H, Furusawa Y, Ohnishi K, Ishioka N, et al. (2005) Apoptosis induced by high-LET radiations is not affected by cellular p53 gene status. Int J Radiat Biol 81: 581-586.
-
Hirayama R, Furusawa Y, Fukawa T, Ando K (2005) Repair kinetics of DNA-DSB induced by X-rays or carbon ions under oxic and hypoxic conditions. J Radiat Res 46: 325-332.
-
Terato H, Tanaka R, Nakaarai Y, Nohara T, Doi Y, et al. (2008) Quantitative analysis of isolated and clustered DNA damage induced by gamma-rays, carbon ion beams, and iron ion beams. J Radiat Res 49: 133-146.
-
Blakely EA, Ngo F, Curtis SB, Tobias CA (1984) Heavy-ion radiobiology: Celular studies. Adv Radiat Biol 11: 295-389.
-
Furusawa Y, Fukutsu K, Aoki M, Itsukaichi H, Eguchi-Kasai K, et al. (2000) Inactivation of aerobic and hypoxic cells from three different cell lines by acceralated (3)He-, (12)C- and (20)Ne-ion beams. Radiat Res 154: 485-496.
-
Shao C, Aoki M, Furusawa Y (2001) Medium-mediated bystander effects on HSG cells co-cultivated with cells irradiated by X-rays or a 290 MeV/u carbon beam. J Radiat Res 42: 305-316.
-
Weyrather WK, Kraft G (2004) RBE of carbon ions: experimental data and the strategy of RBE calculation for treatment planning. Radiother Oncol 73: S161-S169.
-
Torres-roca JF (2012) A molecular assay of tumor radiosensitivity: a roadmap towards biology-based personalized radiation therapy. Per Med 9: 547-557.
-
Guzzi F, Zanchetta D, Cassoni P, Guzzi V, Francolini M, et al. (2002) Localization of the human oxytosin receptor in cavaolin-1 enriched domains turns the receptor-mediated inhibintion of cell growth into a proliferative response. Oncogene 21: 1658-1667.
-
Sasaki R, Narisawa-Saito M, Yugawa T, Fujita M, Tashiro H, et al. (2009) Oncogenic transformation of human ovarian surface epithelial cells with defined cellular oncogenes. Carcinogenesis 30: 423-431.
-
Oike T, Ogiwara H, Torikai K, Nakano T, Yokota J, et al. (2012) Garcinol, a histone acetyltransferase inhibitor, radiosensitizes cancer cells by inhibiting non-homologous end joining. Int J Radiat Oncol Biol Phys 84: 815-821.
-
Hall EJ, Giaccia AJ (2011) Radiobiology for the Radiologist. (7th edn), Lippincott Williams & Wilkins, Philadelphia (PA).
-
Hendry JH, West CM (1997) Apoptosis and mitotic cell death: their relative contributions to normal-tissue and tumour radiation response. Int J Radiat Biol 71: 709-719.
-
Roninson IB, Broude EV, Chang BD (2001) If not apoptosis, then what? Treatment-induced senescence and mitotic catastrophe in tumor cells. Drug Resist Updat 4: 303-313.
-
Gewirtz DA, Holt SE, Elmore LW (2008) Accelerated senescence: an emerging role in tumor cell response to chemotherapy and radiation. Biochem Pharmacol 76: 947-957.
-
de Bruin EC, Medema JP (2008) Apoptosis and non-apoptotic deaths in cancer development and treatment response. Cancer Treat Rev 34: 737-749.
-
Mehnati P, Morimoto S, Yatagai F, Furusawa Y, Kobayashi Y, et al. (2005) Exploration of "over kill effect" of high-LET Ar- and Fe-ions by evaluating the fraction of non-hit cell and interphase death. J Radiat Res 46: 343-350.
-
Oike T, Komachi M, Ogiwara H, Amornwichet N, Saitoh Y, et al. (2014) C646, a selective small molecule inhibitor of histone acetyltransferase p300, radiosensitizes lung cancer cells by enhancing mitotic catastrophe. Radiother Oncol 111: 222-227.





