Clinical Medical
Reviews and Case Reports
Neonatal Polycythemia: A Review
S Umit Sarici, Murat Ozcan and Demet Altun*
Department of Pediatrics, Ufuk University, Turkey
*Corresponding author: Demet Altun, Department of Pediatrics, Ufuk University Faculty of Medicine, Ankara, Turkey, Tel: +90-312-204-4000, E-mail: draltundemet@gmail.com
Clin Med Rev Case Rep, CMRCR-3-142, (Volume 3, Issue 11), Review Article; ISSN: 2378-3656
Received: September 03, 2016 | Accepted: November 21, 2016 | Published: November 23, 2016
Citation: Sarici SU, Ozcan M, Altun D (2016) Neonatal Polycythemia: A Review. Clin Med Rev Case Rep 3:142. 10.23937/2378-3656/1410142
Copyright: © 2016 Sarıcı SU, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Polycythemia in the newborn is defined as either venous hematocrit or hemoglobin levels above 65% and 22 g/dl, respectively. Its incidence is reported between 1 to 5%. In this article definition and epidemiology of polycythemia, physiological changes in postnatal hematocrit levels, relationship between polycythemia and hyperviscosity, pathophysiology and diagnosis of polycythemia, etiology, clinical signs and complications of polycythemia with a special emphasis on treatment algorithms and long-term neurodevelopmental outcome considering all current review articles and meta-analyses are reviewed.
Keywords
Hyperviscosity, Newborn, Polycythemia, Partial exchange transfusion
Introduction
Definition and epidemiology
Polycythemia in the newborn is defined as either venous hematocrit or hemoglobin levels above 65% and 22 g/dl, respectively [1-4]. Its incidence is reported between 1 to 5% [3-8]. Its risk is higher in newborns born to mothers living in high altitudes whereas the risk decreases in premature newborns born before 34th week of gestation [9-11].
Physiological changes in postnatal hematocrit levels
Hematocrit levels increase in comparison to cord levels making a peak at around 2nd hour of life and a plateau between 2-4 hours of life, and then return to cord blood levels at 12 to 18 hours of life. The postnatal age at which newborns are screened for polycythemia is, thus, very important with this respect. The incidence of polycythemia may increase up to 20% when screened at 2nd hour whereas its incidence may be detected as low as 2% when its screening is performed at 12 to 18 hours postnatally [12].
Capillary and venous hematocrit measurements
Hematocrit measurement can be made in both capillary and venous samples. However erythtrocyte concentration and the ultimately measured hematocrit level show a great variability depending on the route where the blood sample is obtained. Hematocrit measurement in capillary samples gives higher results in comparison to venous samples because of the rouleaux formation and migration of erythrocytes along the vascular wall, and sensitivity to variations in blood flow [9].
Due to the variations in and weaknesses of capillary measurement, gold standard blood sample to be used in the diagnosis of polycythemia is venous blood sample. In general hematocrit level in capillary samples is found 5-15% higher than that in venous samples, and hematocrit level in peripheral blood samples is higher than that in central venous samples. In a study conducted on newborns with a peripheral venous hematocrit level of ≥ 65%, capillary, peripheral venous and central (umbilical) venous hematocrit levels were reported as 75%, 71% and 63%, respectively [4]. In parallel with this finding, only 3-5% of newborns diagnosed to have polycythemia on screening with capillary measurements were found hyperviscosis [3,13,14], whereas approximately 50% of newborns detected as polycythemic in umbilical cord blood samples had hyperviscosity [15].
Warming the heel before obtaining capillary sample via heel-stick may aid in providing a closer correlation between the measurements of capillary and venous blood samples. Capillary samples may be used for screening, however all high values should be confirmed by a venous sample for the diagnosis of polycythemia [10,11] since approximately 18% of those tested and misdiagnosed with an incorrect method at 2nd hour of life would be subjected to a potential harmful and expensive treatment [12].
Methods of Hematocrit Analysis
Two methods are available
Automatized hematologic analysis: Hematocrit value is indirectly calculated from mean corpuscular volume and hemoglobin values in automatic blood analyzers.
Micro-centrifuge method: Blood is collected into heparinized microcapillary tubes and centrifuged at 10000-15000 rpm for 3-5 minutes. Plasma is separated and packed red cell volume is measured to give the hematocrit. An automatized analyzer gives lower values when compared to the centrifugation method [11]. In most of the studies conducted about polycythemia, centrifugation method has been used.
Relationship between Polycythemia and Hyperviscosity
Hyperviscosity and polycythemia are different definitions although they are often used interchangeably. Polycythemia is the abnormal increase in erythrocyte mass, and defined as a venous hematocrit level of > 65% in newborns. Viscosity is, on the other hand, the property of the liquid which provides the resistance of that liquid against placing between the two layers of the fluid. Viscosity equals to the ratio of powers effective on the unit square of the liquid or sliding tension to sliding velocity. Sliding resistance of the liquid or its response to changes in sliding velocity is somewhat different from other liquids as whole blood is not a homogenous liquid [9]. The relationship between hematocrit and viscosity is linear below the hematocrit levels of 60%, whereas progressive increases over 65% show a logarithmic relationship [4,13]. This means even a small increase in hematocrit over the levels of > 65% causes a dramatic increase in viscosity. Hyperviscosity is the decrease of blood fluidity, and defined as a viscosity above the 2 standard deviation of the mean. Hyperviscosity syndrome, on the other hand, is a syndrome of circulatory disorder developing secondary to increase in resistance against blood flow [9,11]. Except for the hematocrit level, there are a lot of factors affecting whole blood viscosity. Leukocytes, platelets, plasma proteins, immunoglobulins and coagulation factors are the other cellular elements which affect the whole blood viscosity. Hyperviscosity has been detected in 47% of newborns with polycythemia [15], whereas polycythemia has been detected as the etiologic factor in only 24% of newborns with hyperviscosity [11,15,16]. It is not easy to measure viscosity, and Wells-Brookfield viscometer is used with this aim. However most of the neonatal intensive care units do not have this device, and high hematocrit level and the presence of polycythemia-associated symptoms are taken into account as the determinator of hyperviscosity [9,11].
Pathophysiology
Polycythemia may cause some symptoms and complications due to the metabolic and hemodynamic effects of hyperviscosity, microcirculatory hipoperfusion, decreased organ blood flow and multisystem organ dysfunction, destruction of increased erythrocyte mass, and hyper/hypovolemia. Tissue hypoxia, acidosis and hypoglycemia develop secondarily to regional effects of hyperviscosity. Tissue blood supply and oxygenation are disrupted. Oxygen carrying capacity is determined by the hemoglobin level and blood flow, it is optimal at the normal hematocrit levels, and oxygen transport is decreased at low hematocrit levels as the oxygen binding capacity decreases [17]. Increase of blood volume, while the hematocrit is the same, increases blood flow and oxygen transport by decreasing peripheral vascular resistance with vasodilatation. This mechanism constitutes the basis of treatment of polycythemia [7]. With polycythemia/hyperviscosity, decreases in cerebral blood flow, arterial glucose concentration, cerebral glucose up take and disturbed cerebral glucose distribution have been reported [17,18].
Decreased microcirculation has been deemed responsible for the morbidity associated with polycythemia. Thrombi ocurring in microcirculation may cause symptoms in central nervous system, kidneys, surrenal glands, cardiopulmonary and gastrointestinal systems [19]. Drew, et al. [15] have demonstrated that the primary factor determining the neurological prognosis in polycythemia/hyperviscosity syndrome is hyperviscosity [20]. As a conclusion perfusion and tissue oxygenation are disturbed, plasma glucose concentration decreases, cerebral glucose uptake is disturbed and risk of cerebral morbidity is increased with microthrombi formation as the viscosity increases [7,17,21]. In neonatal polycythemia increased destruction of increased erythrocyte mass with a relatively shorter erythrocyte life span primarily contributes to hyperbilirubinemia. Hypervolemia may lead to congestive heart failure, pulmonary edema and cardiopulmonary failure, and hypovolemia may cause hypoxic-ischemic organ injury [12].
Etiology
Although the etiology of polycythemia is multifactorial, there are two primary mechanisms: passive (erythrocyte transfusion) and active (increased intrauterine erythropoiesis) (Table 1) [22]. Polycythemia secondary to excess erythrocyte transfusion to the fetus (Passive polycythemia) may occur due to delayed clamping of the cord, acute fetal distress and intrapartum hypoxia, twin-to-twin transfusion syndrome, materno-fetal transfusion and holding the baby below the level of introitus [9,11].
![]()
Table 1: Etiology of neonatal polycythemia [22].
View Table 1
In acute fetal distress and peripartum hypoxia transcapillary leakage of plasma occurs and blood flow from placenta to fetus increases, and all these result in increased plasma volume and erythrocyte mass in the fetus [9]. Clamping of the umbilical cord later than 3 minutes after delivery of the baby is defined as "delayed cord clamping" [11]. Carpasso, et al. (2003) have reported a significant decrease in the incidence of polycythemia in newborns whose umbilical cord was clamped early (in first 10 seconds of life) when compared to those whose umbilical cord was clamped late (in 11th to 120th seconds of life) [23]. Therefore, early cord clamping and holding the baby at the level of introitus at the time of delivery could play a role in prevention from polycythemia by minimizing materno-fetal transfusion. On the other hand, however, no statistically significant differences were reported in the hematocrit values of newborns whose umbilical cord was clamped early or late [24]. Twin-to-twin transfusion syndrome is seen in approximately 10% of recipients of monochorionic twins [25].
Polycythemia secondary to increased intrauterine erythropoiesis (active polycythemia) is usually observed in cases of placental insufficiency, intrauterine hypoxia and situations associated with the fetus. Maternal hypertension [26], preeclampsia, maternal diabetes mellitus (type 1 diabetes mellitus and gestational diabetes) [27], maternal cyanotic heart disease, intrauterine growth retardation, postmaturity, living at high altitude and maternal smoking are all associated with this mechanism.
Situations associated with fetus may develop secondarily to problems either in fetus or maternal diseases (Table 1). The incidence of polycythemia in newborns of diabetic mothers varies between 22 to 29%, and polycythemia shows a close correlation with macrosomia and neonatal hypoglycemia in these babies [9,11]. There is an increased risk of polycythemia in diseases with a genetic inheritance such as trisomy 18 and trisomy 13 [28], trisomy 21 [29], and Beckwith-Wiedemann syndrome. Congenital hypothyroidsm, neonatal thyrotoxicosis and congenital adrenal hyperplasia are the other causes of polycythemia associated with the fetus [9,11].
Another etiologic classification of polycythemia is the one based on the volume status of plasma: normovolemic, hypervolemic and hypovolemic [12].
Normovolemic polycythemiaThere is an increase in erythrocyte mass while intravascular volume is normal. It is seen in intrauterine growth retardation, maternal hypertension, maternal diabetes mellitus, and in situations associated with placental insufficiency and/or chronic intrauterine hypoxia such as maternal smoking and postmaturity.
Hypervolemic polycythemia
There is an increase in plasma volume in association with an increased erythrocyte mass. It is usually seen in cases of acute transfusion such as maternofetal transfusion and twin-to-twin transfusion.
Hypovolemic polycythemia
It is due to the relative increase of erythrocyte mass in comparison to plasma volume. This situation usually develops due to intravascular dehydration [12].
Clinical Signs and Complications
Most of the newborns (74-90%) are asymptomatic [30,31]. In symptomatic newborns polycythemia may affect many organs and systems. Hyperviscosity, decrease of tissue perfusion, and metabolic complications such as hypoglycemia and hypocalcemia are responsible for clinical signs [10,32]. Nonspecific signs and symptoms such as apnea, cyanosis, feeding problems, vomiting, irritability, jitteriness, tremor, lethargy, respiratory distress and seizures may be seen [16]. The most commonly encountered problems in severely symptomatic newborns with polycythemia are central nervous system disorders [11,32,33].
Cardiopulmonary complications (Cardiomegaly, increase in pulmonary vascular resistance and decrease in cardiac output) with tachycardia and tachypnea may develop [32].
Although polycythemia and hyperviscosity have been suggested responsible for the pathogenesis of necrotizing enterocolitis in term and near-term newborns [34,35], partial exchange transfusion itself, performed to lower the hematocrit, has been reported to cause necrotizing enterocolitis [36-38].
Renal problems encountered in polycythemia are decrease in glomerular filtration rate, oliguria, hematuria, proteinuria and renal vein thrombosis [16,39].
Of the metabolic problems, the most commonly encountered is hypoglycemia (12-40%). In addition to cerebral blood flow, glucose carrying capacity also decreases in polycythemia. As a result plasma glucose concentration, especially venous one is lower than normal [9]. Hypocalcemia and hyperbilirubinemia may also be seen in polycythemic newborns [9,11]. The level of calcitonin gene related peptide (CGRP) has been shown to be high in polycythemic newborns. This peptide regulates vascular tonus stimulating vasodilatation and leads to hypocalcemia, and high levels of CGRP suggest its role in response to polycythemia [40].
Thrombocytopenia, low antithrombin III levels, and more rarely development of thrombosis are hematological problems encountered in polycythemia [9,11]. As the density of the erythrocytes increases they accumulate in the center of blood flow, and thrombocytes, which are lighter, migrate to periphery of the vascular wall (thrombocyte margination). This causes the number of thrombocytes to be counted lower than the actual count (relative thrombocytopenia) [7,32].
Is Routine Screening Necessary?
Screening is not necessary in asymptomatic conditions [12,22]. Screening may be considered in symptomatic cases and some selected high-risk groups (Small-for-gestational age and large-for-gestational age newborns, newborns of diabetic mothers, monochorionic twins) in the presence of symptoms suggestive of polycythemia. Capillary hematocrit measurement is the method of choice for screening.
Treatment
In clinical practice one of the most commonly encountered causes of polycythemia is dehydration. Therefore dehydration, which may develop secondary to causes such as fever, feeding problems, vomiting and diarrhea should be excluded before establishing a diagnosis of polycythemia [9]. Polycythemia due to dehydration may be present in a period extending into the 2-3 days of life in contrast to most of the early causes of polycythemia. Birth weights and actual weights of the babies should be compared, and dehydration should be suspected in case of weight loss over 7% in the first five days of life [22]. In case of dehydration, this situation should be corrected by increasing fluid intake, and hematocrit measurement should be repeated there after [9,11]. All polycythemic newborns should be investigated and followed for neurologic, gastrointestinal and cardiopulmonary signs and commonly encountered complications such as hypoglycemia and hyperbilirubinemia [9,11,22].
Treatment options in polycythemic patients depend on whether they are symptomatic or not and their hematocrit levels (Figure 1) [22].
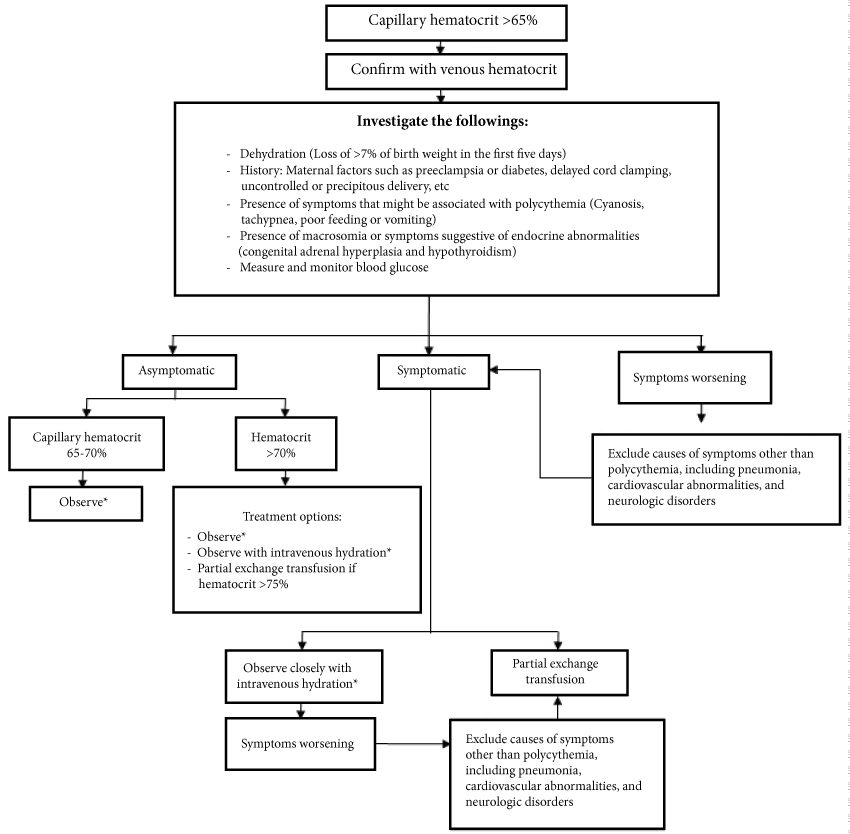
.
Figure 1: Treatment algorithm in neonatal polycythemia [22].
*Observation consists of monitoring intake, weight and urine output; follow up blood glucose as indicated based on initial results; monitor for symptoms.
View Figure 1
Asymptomatic polycythemia
Treatment option in asymptomatic polycythemic patients depends on the hematocrit level (Plethora is not considered as a symptom).
Asymptomatic patients with a peripheral venous hematocrit level between 65-70% should be observed for intake, weight, and urine output providing sufficient hydration and glucose intake. Serum bilirubin and glucose levels should be checked when necessary. While monitoring for the development of symptoms, a venous hematocrit mesurement should be repeated in 12-24 hours. If the repeated hematocrit is below 70% and no symptoms develop, the same policy should be continued for 24 hours and a hematocrit measurement should be repeated.
For the patients with a peripheral venous hematocrit level above 70%, there are a few treatment options. Some clinicians prefer to continue observation with or without providing intravenous hydration. Some other clinicians prefer partial exchange transfusion (PET) in asymptomatic patients only if venous hematocrit is above 75% [41,42], whereas some others advise PET if venous hematocrit is above 70% even in asymptomatic cases (less common) [12,43,44].
Symptomatic polycythemia
The optimal management of symptomatic polycythemic newborns has not been exactly established and there are various approaches. Some clinicians perform PET to lower hematocrit in symptomatic cases with a peripheral venous hematocrit level above 65% [41]. Neonatal hematocrit and blood viscosity peaks between two and four hours after birth, and also considering that most of the studies reporting no favourable long-term benefits of PET on neurodevelopmental outcome have been performed PET after 6 hours of age, PET should be done as soon as possible (in the first 2-4 hours of life) after a decision is made to perform PET [12,22]. On the other hand some clinicians prefer close observation with intravenous hydration. The main aim of intravenous hydration is to prevent the development of hypoglycemia, a common complication of polycythemia. Intravenous fluid should be provided for the first 24 to 48 hours of age at a rate of at least 100 ml/kg per day, including glucose at a rate 6 to 8 mg/kg per min. With this approach a PET is performed only if there is worsening of symptoms, such as persistent hypoglycemia, or persistent cyanosis/apnea, or gastrointestinal symptoms [22].
PET treatment
Isovolumetric PET reduces hematocrit without causing hypovolemia. Although PET acutely demonstrates hemodynamic improvements in cerebral blood flow, cardiac index and oxygen transport [45-51], its long-term effects on psychomotor and neurodevelopmental outcome have not clearly been established and are under debate.
PET can be performed via peripheral or central routes. In peripheral route, peripheral arteries and veins are used. Blood is removed from an arterial catheter, and normal saline is simultaneously infused into a peripheral vein. In central route, blood is removed from an umbilical venous catheter and normal saline is simultaneously infused into a peripheral vein. Umbilical venous catheter also can be used for both removing blood and infusing fluid [11], and this method is the most commonly used method in practice.
The exchange volume is calculated using the following formula:
*Desired hematocrit is usually 55%.
**Blood volume is 80-90 ml/kg in term babies and 90-100 ml/kg in preterm babies.
In general the exchange volume is 15-20 ml/kg [22].
Crystalloids such as normal saline or ringer’s lactate or colloids such as fresh frozen plasma or 5% albumin can be used for PET. These two types of fluids provide a similar efficacy in lowering hematocrit [11], and normal saline is the fluid of choice to be used in PET since it does not carry the risk of transfusion associated infections, and less expensive and easily available [11,52].
Prognosis
In acute phase the incidence of seizures and intracerebral hemorrhages is higher in polycythemic babies in comparison to normal babies. The early neonatal behaviour of polycythemic babies assessed by Brazelton Behavioral Assessment Scale demonstrated abnormalities of hypotonia, poor state control and irritability [53]. The long-term neurodevelopmental outcome of polycythemic infants remains controversial. Malanand de V Heese (1980) reported no neurodevelopmental differences between polycythemic and normal infants on follow-up at 8 months of age [54]. Delaney-Black, et al. (1989) reported speec hand fine motor abnormalities in polycythemic infants at 2 years of age. In the same children at 7 years of age they noted lower spelling and arithmetic achievement test results and gross motor skills than normal control children [55].
Long-term effects of PET on neurodevelopmental outcome in comparison to conservative treatment have been studied in limited number of studies [30,44,48,53,54,56,57]. In none of these studies a positive effect of PET on long-term neurodevelopmental outcome could be demonstrated. Dempsey and Barrington (2006) have performed a systematic meta-analysis on five of these studies [30,48,53,54,56], and investigated whether PET has positive short-term or long-term neurodevelopmental effects in polycythemic infants [37]. The authors reported no improvement in long-term neurologic outcome (mental developmental index, incidence of developmental delay, and incidence of neurologic diagnoses) after PET in symptomatic or asymptomatic newborns. There was also no improvement in early neurobehavioral assessment scores (Brazelton Neonatal Behavioral Assessment Scale) [37].
In another review, Ozek, et al. (2010) performed a meta-analysis on all of the 7 above-mentioned studies assessing the effect of PET on long-term neurodevelopmental prognosis [30,44,48,53,54,56,57], and evaluated the effect of PET especially on mortality and neurodevelopmental prognosis at 2 years of age and school-age [36]. Secondary parameters evaluated were seizures, cerebral infarct, necrotizing enterocolitis (NEC), hypoglycemia, hyperbilirubinemia, and thrombocytopenia [36]. Although the meta-analysis had limited data (poor follow-up) and did not account for patients who were lost on follow-up, the authors concluded that PET had no significant benefits in asymptomatic patients or those who have mild symptoms [36].
In addition to no documented positive effects of PET on long-term neurodevelopmental prognosis, and after the observation of a significant increase in the incidence of NEC in newborns performed PET [30,36,37,54], PET is currently being preferred in only (symptomatic) newborns who have symptoms associated with hyperviscosity and not in asymptomatic polycythemic newborns [8,12,22,36,37].
Criteria to Write the Review
Our main aim was to help pediatricians and neonatal caregivers who care for newborns to identify and manage those with neonatal polycythemia. On a monthly basis over the last 43 years, we have searched MEDLINE for English-language articles using the terms "neonatal polycythemia", "hyperviscosity", "partial exchange transfusion" and "hematocrit". We also reviewed articles on these subjects in the Cochrane Database of Systematic Reviews, as well as all meta-analyses and recent textbooks, all published from 1982 to 2016.
References
-
Oski FA, Naiman JL (1982) Polycythemia and hyperviscosity in the neonatal period. In: Oski FA, Naiman JL, Hematologic Problems in the Newborn. (3rd edn), WB Saunders, New York, 87-96.
-
Wiswell TE, Cornish JD, Northam RS (1986) Neonatal polycythemia: frequency of clinical manifestations and other associated findings. Pediatrics 78: 26-30.
-
Stevens K, Wirth FH (1980) Incidence of neonatal hyperviscosity at sea level. J Pediatr 97: 118-119.
-
Ramamurthy RS, Brans YW (1981) Neonatal polycythemia: I. Criteria for diagnosis and treatment. Pediatrics 68: 168-174.
-
Brooks GI, Backes CR (1981) Hyperviscosity secondary to polycythemia in the appropriate for gestational age neonate. J Am Osteopath Assoc 80: 415-418.
-
Rawlings JS, Pettett G, Wiswell TE, Clapper J (1982) Estimated blood volumes in polycythemic neonates as a function of birth weight. J Pediatr 101: 594-599.
-
Mentzer WC, Glader BE (1998) Polycthemia. In: Taeusch HW, Ballard RA, Gleason CA, Avery's Diseases of the Newborn. WB Saunders Company, Philadelphia 1104-1106.
-
Remon JI, Raghavan A, Maheshwari A (2011) Polycythemia in the newborn. Neoreviews 12: e20-e28.
-
Rosenkrantz TS, Oh W (2005) Polycythemia and hyperviscosity in the newborn. In: De Alarcón P, Werner E, Neonatal Hematology. Cambridge University Press, New York, 171-186.
-
Upadhyay A, Aggarwal R, Deorari AK, Paul VK (2002) Polycythemia in the newborn. Indian J Pediatr 69: 79-82.
-
Jeevasankar M, Agarwal R, Chawla D, Paul VK, Deorari AK (2008) Polycythemia in the newborn. Indian J Pediatr 75: 68-72.
-
Mimouni FB, Merlob P, Dollberg S, Mandel D, Israeli Neonatal Association (2011) Neonatal polycythaemia: critical review and a consensus statement of the Israeli Neonatology Association. Acta Paediatr 100: 1290-1296.
-
Gross GP, Hathaway WE, McGaughey HR (1973) Hyperviscosity in the neonate. J Pediatr 82: 1004-1012.
-
Wirth FH, Goldberg KE, Lubchenco LO (1979) Neonatal hyperviscosity: I. Incidence. Pediatrics 63: 833-836.
-
Drew JH, Guaran RL, Grauer S, Hobbs JB (1991) Cord whole blood hyperviscosity: measurement, definition, incidence and clinical features. J Paediatr Child Health 27: 363-365.
-
Rosenkrantz TS (2003) Polycythemia and hyperviscosity in the newborn. Semin Thromb Hemost 29: 515-527.
-
Rosenkrantz TS, Philipps AF, Skrzypczak PS, Raye JR (1988) Cerebral metabolism in the newborn lamb with polycythemia. Pediatr Res 23: 329-333.
-
Rosenkrantz TS, Philipps AF, Knox I, Zalneraitis EL, Porte PJ, et al. (1992) Regulation of cerebral glucose metabolism in normal and polycythemic newborn lambs. J Cereb Blood Flow Metab 12: 856-865.
-
Norman M, Fagrell B, Herin P (1992) Effects of neonatal polycythemia and hemodilution on capillary perfusion. J Pediatr 121: 103-108.
-
Drew JH, Guaran RL, Cichello M, Hobbs JB (1997) Neonatal whole blood hyperviscosity: the important factor influencing later neurologic function is the viscosity and not the polycythemia. Clin Hemorheol Microcirc 17: 67-72.
-
Arslanoğlu S (2001) Yenidogan doneminde polisitemi/hiperviskozite sendromu: yaklaşım ve tedavide yenilikler. T Klin Pediatri 10: 49-58.
-
http://www.uptodate.com/contents/neonatal-polycythemia.
-
Carpasso L, Raimondi F, Capasso A, Crivaro V, Capasso R, et al. (2003) Early cord clamping protects at-risk neonates from polycythemia. Biol Neonate 83: 197-200.
-
Jahazi A, Kordi M, Mirbehbahani NB, Mazloom SR (2008) The effect of early and late umbilical cord clamping on neonatal hematocrite. J Perinatol 23: 523-525.
-
Chalouhi GE, Stirnemann JJ, Salomon LJ, Essaoui M, Quibel T, et al. (2010) Specific complications of monochorionic twin pregnancies: twin-twin transfusion syndrome and twin reversed arterial perfusion sequence. Semin Fetal Neonatal Med 15: 349-356.
-
Kurlat I, Sola A (1992) Neonatal polycythemia in appropriately grown infants of hypertensive mothers. Acta Paediatr 81: 662-664.
-
Yeruchimovich M, Mimouni FB, Green DW, Dollberg S (2000) Nucleated red blood cells in healthy infants of women with gestational diabetes. Obstet Gynecol 95: 84-86.
-
Wiedmeier SE, Henry E, Christensen RD (2008) Hematological abnormalities during the first week of life among neonates with trisomy 18 and trisomy 13: data from multihospital healthcare system. Am Med Genet A 146A: 312-320.
-
Widness JA, Pueschel SM, PezzulloJC, Clemons GK (1994) Elevated erythropoietin levels in cord blood of newborns with Down’s syndrome. Biol Neonate 66: 50-55.
-
Black VD, Lubchenco LO, Koops BL, Poland RL, Powell DP (1985) Neonatal hyperviscosity: randomized study of effect of partial plasma exchange transfusion on long-term outcome. Pediatrics 75: 1048-1053.
-
Black VD, Lubchenco LO, Luckey DW, Koops BL, McGuinness GA, et al. (1982) Developmental and neurologic sequelae of neonatal hyperviscosity syndrome. Pediatrics 69: 426-431.
-
Mentzer WC, Glader BG (2005) Erythrocyte disorders in infancy. In: Taeusch HW, Ballard RA, Gleason CA, Avery ME, Avery’s diseases of the newborn. Elsevier Inc, Philadelphia, 1180-1214.
-
Dollberg S, Marom R, Mimouni FB, Littner Y (2007) Increased energy expenditure after dilutional Exchange transfusion for neonatal polycythemia. J Am Coll Nutr 26: 412-415.
-
Lambert DK, Christensen RD, Henry E, Besner GE, Baer VL, et al. (2007) Necrotizing enterocolitis in term neonates: data from a multihospital health-care system. J Perinatol 27: 437-443.
-
Martinez-Tallo E, Claure N, Bancalari E (1997) Necrotizing enterocolitis in full-term or near-term infants: risk factors. Biol Neonate 71: 292-298.
-
Ozek E, Soll R, Schimmel MS (2010) Partial exchange transfusion to prevent neurodevelopmental disability in infants with polycythemia. Cochrane Database Syst Rev 20: CD005089.
-
Dempsey EM, Barrington K (2006) Short and long term outcomes following partial exchange transfusion in the polycythaemic newborn: a systematic review. Arch Dis Child Fetal Neonatal Ed 91: F2-F6.
-
Maayan-Metzger A, Itzchak A, Mazkereth R, Kuint J (2004) Necrotizing enterocolitis in full-term infants: case-control study and review of the literature. J Perinatol 24: 494-499.
-
Sarkar S, Rosenkrantz TS (2008) Neonatal polycythemia and hyperviscosity. Semin Fetal Neonatal Med 13: 248-255.
-
Saggese G, Bertelloni S, Baroncelli GI, Cipolloni C (1992) Elevated calcitonin-gene related peptide in polcythemic newborn infants. Acta Paediatr 81: 966-968.
-
Schimmel MS, Bromiker R, Soll RF (2004) Neonatal polycythemia: is partial exchange transfusion justified? Clin Perinatol 31: 545-553.
-
Sankar MJ, Agarwal R, Deorari A, Paul VK (2010) Management of polycythemia in neonates. Indian J Pediatr 77: 1117-1121.
-
Acunas B, Celtik C, Vatansever U, Karasalihoglu S (2000) Thrombocytopenia: an important indicator for the application of partial exchange transfusion in polycythemic newborn infants? Pediatr Int 42: 343-347.
-
Kumar A, Ramji S (2004) Effect of partial exchange transfusion in asymptomatic polycythemic LBW babies. Indian Pediatr 41: 366-372.
-
Swetnam SM, Yabek SM, Alverson DC (1987) Hemodynamic consequences of neonatal polycythemia. J Pediatr 110: 443-447.
-
Bada HS, Korones SB, Pourcyrous M, Wong SP, Wilson WM 3rd, et al. (1992) Asymptomatic syndrome of polycythemic hyperviscosity: effect of partial plasma exchange transfusion. J Pediatr 120: 579-585.
-
Rosenkrantz TS, Oh W (1982) Cerebral blood flow velocity in infants with polycythemia and hyperviscosity: effects of partial exchange transfusion with Plasmanate. J Pediatr 101: 94-98.
-
Bada HS, Korones SB, Kolni HW, Fitch CW, Ford DL, et al. (1986) Partial plasma exchange transfusion improves cerebral hemodynamics in symptomatic neonatal polycythemia. Am J Med Sci 291: 157-163.
-
Maertzdorf WJ, Tangelder GJ, Slaaf DW, Blanco CE (1989) Effects of partial plasma exchange transfusion on cerebral blood flow velocity in polycythaemic preterm, term and small for date newborn infants. Eur J Pediatr 148: 774-778.
-
Ergenekon E, Hirfanoglu IM, Turan O, Beken S, Gucuyener K, et al. (2011) Partial exchange transfusion results in increased cerebral oxygenation and faster peripheral microcirculation in newborns with polycythemia. Acta Paediatr 100: 1432-1436.
-
Murphy DJ Jr, Reller MD, Meyer RA, Kaplan S (1985) Effects of neonatal polycythemia and partial exchange transfusion on cardiac function: an echocardiographic study. Pediatrics 76: 909-913.
-
de Waal KA, Baerts W, Offringa M (2006) Systematic review of the optimal fluid for dilutional exchange transfusion in neonatal polycythaemia. Arch Dis Child Fetal Neonatal Ed 91: F7-F10.
-
Goldberg K, Wirth FH, Hathaway WE, Guggenheim MA, Murphy JR, et al. (1982) Neonatal hyperviscosity. II. Effect of partial plasma exchange transfusion. Pediatrics 69: 419-425.
-
Malan AF, de V Heese H (1980) The management of polycythaemia in the newborn infant. Early Hum Dev 4: 393-403.
-
Delaney-Black V, Camp BW, Lubchenco LO, Swanson C, Roberts L, et al. (1989) Neonatal hyperviscosity association with lower achievement and IQ scores at school age. Pediatrics 83: 662-667.
-
Ratrisawadi V, Plubrukarn R, Trakulchang K, Puapondh Y (1994) Developmental outcome of infants with neonatal polycythemia. J Med Assoc Thai 77: 76-80.
-
Hakanson DO (1981) Neonatal hyperviscosity syndrome: long-term benefit of partial plasma exchange transfusion (Abstract). Pediatr Res 15: 449A.





