Journal of Rheumatic Diseases and Treatment
Glucocorticoid-Induced Leucine Zipper (Gilz) in Rheumatoid Arthritis: Relationship with Disease Activity and Steroid Use
Laura E Eades*, Angeline S Thiagarajah, Sarah A Jones, Huapeng Fan, Anastasios Nalpantidis, Eric F Morand and Michelle Leech
Centre for Inflammatory Diseases, School of Clinical Sciences at Monash Health, Monash University, Australia
*Corresponding author: Laura Eades, Centre for Inflammatory Diseases, School of Clinical Sciences at Monash Health, Monash Medical Centre, Monash University, 246 Clayton Road, Clayton, Victoria 3168, Australia, Tel: 0424589400, E-mail: laura.eades@monashhealth.org
J Rheum Dis Treat, JRDT-3-047, (Volume 3, Issue 1), Research Article; ISSN: 2469-5726
Received: November 07, 2016 | Accepted: February 18, 2017 | Published: February 21, 2017
Citation: Eades LE, Thiagarajah AS, Jones SA, Fan H, Nalpantidis A, et al. (2017) Glucocorticoid-Induced Leucine Zipper (Gilz) in Rheumatoid Arthritis: Relationship with Disease Activity and Steroid Use. J Rheum Dis Treat 3:047. 10.23937/2469-5726/1510047
Copyright: © 2017 Eades LE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aims: Approximately 50% of rheumatoid arthritis (RA) patients require treatment with glucocorticoids (GC) to control inflammation [1]. Although effective, GC cause numerous adverse effects. Glucocorticoid-induced leucine zipper (GILZ) is an anti-inflammatory protein, which is up-regulated by GC. Although GILZ mediates the anti-inflammatory effects of GC, it appears to have osteogenic effects on bone [2]. This has raised interest in GILZ as a potential therapeutic approach, which could replicate the anti-inflammatory effects of GC while minimising the adverse effects [1]. Prior to examining GILZ as a therapeutic avenue, a greater understanding of its role in RA is needed.
Methods: Clinical data was obtained from RA patients and medical records. Three control cohorts were also recruited, including 23 healthy controls, 25 patients with fibromyalgia and 44 patients with depression. Blood was taken from each participant. Following isolation of peripheral blood mononuclear cells (PBMC) from whole blood, RNA was extracted. Reverse transcription polymerase chain reaction (RT PCR) and quantitative real time PCR (qPCR) were performed to quantify GILZ expression.
Results: GILZ expression was increased in RA patients compared to healthy controls, patients with fibromyalgia and patients with depression. Prednisolone use was associated with increased GILZ expression, and there was a weak positive correlation between GILZ expression and prednisolone dose. There was a strong negative correlation between GILZ adjusted for prednisolone dose and disease activity in RA patients.
Conclusions: Our results suggest that patients who are better able to induce GILZ in response to GC may experience more significant improvement in disease activity with relatively lower doses of steroids.
Keywords
Rheumatoid arthritis, GILZ, Glucocorticoids, Anti-inflammatory
Introduction
Rheumatoid arthritis (RA) is a common autoimmune inflammatory condition, which affects approximately 1% of the population [3,4]. Despite dramatic improvements in the treatment of RA following the evolution of synthetic and biological disease modifying anti-rheumatic drugs (DMARDs), approximately 50% of patients remain on glucocorticoids (GC) in order to adequately control inflammation [1,5]. Although GC have potent anti-inflammatory effects, they are associated with numerous side effects, which are dose dependent and related to duration of use [6-9]. These side effects limit the use of GC in RA patients, and contribute significantly to morbidity and mortality in patients whose RA is unable to be controlled without them. Consequently, there is a need for further research into methods of delineating the beneficial anti-inflammatory effects of GC from their pathological side effects.
One of the ways in which GC act to reduce inflammation is through the up-regulation of glucocorticoid-induced leucine zipper (GILZ), a protein which mimics the anti-inflammatory effects of GC, but may not be associated with the pathological side effects. GILZ reduces inflammation by decreasing the production of pro-inflammatory cytokines by T cells, and directing them towards an anti-inflammatory T helper 2 type response [10,11]. In addition, GILZ prevents dendritic cell maturation and decreases the responses of macrophages and monocytes to inflammatory stimuli [12-14]. However, unlike GC, GILZ has been shown to increase osteogenic differentiation and enhance osteoblast gene expression, thus contributing to bone protection [2,15].
Our understanding of the role of GILZ in RA is early in its evolution. Our group has previously shown that GILZ over-expression decreases the severity of arthritis in animal models, both clinically and histologically [16]. Conversely, mice in which GILZ is silenced have significantly increased production of pro-inflammatory cytokines such as tumour necrosis factor and interleukin-1 in the synovium, as well as increased bone and joint destruction when examined histologically [17].
There is also a suggestion that GILZ may be up-regulated in human RA, with synovial tissue from RA patients having higher levels of GILZ compared to healthy controls [17]. A study by Quax, et al. examined GILZ in the context of GC in RA patients recently [18]. There was a weak, non-significant correlation between higher levels of GILZ expression in ex-vivo PBMC following DEX administration and associated improvement in disease activity following intramuscular GC injection in vivo [18]. This suggests that patients who are better able to induce GILZ in response to GC exposure may respond better to GC therapy [18]. The in vivo peripheral blood expression of GILZ in RA patients in the context of usual dose steroids and intercurrent disease activity has not previously been assessed. We therefore aimed to further our understanding of the expression of GILZ in RA in an observational standard care setting, with a particular focus on the relationship between GILZ and GC use, as well as disease activity. Given that GILZ expression is induced by hypothalamic pituitary adrenal (HPA) axis stress, we also sought to compare GILZ expression in RA with non-inflammatory conditions known to the influence and respond to the HPA axis, including depression and fibromyalgia.
Methods
Ethics
Institutional Human Research Ethics Committee approval was obtained prior to collection of data.
Participant recruitment and data collection
Eighty-six patients with RA defined by ACR/EULAR 2010 criteria, were recruited [19]. All consenting patients with a diagnosis of RA (ACR 2010) were eligible to participate. The tender joint count (TJC) and swollen joint count (SJC) for each participant was assessed on the day of recruitment by a rheumatologist. The TJC and SJC were combined with the most recent ESR measurement (within ten days of recruitment) to calculate the DAS28 score (disease activity). Further clinical information regarding patient demographics and current medications was retrieved from medical records.
Non-inflamed controls
Three control cohorts were recruited, including healthy controls, patients with fibromyalgia and patients with depression, to assess the difference in GILZ expression between RA patients and healthy controls, as well as RA patients and patients with non-inflammatory chronic diseases. None of the controls had a diagnosis of an inflammatory illness or were taking a GC. Demographic information was collected from each control participant. The healthy control cohort comprised of 23 healthy non age-matched volunteers. A cohort of 30 fibromyalgia patients (ACR Fibromyalgia criteria) was recruited from the fibromyalgia outpatient clinic. Finally, a cohort of 49 depression patients (DSM IV) was recruited from private outpatient consulting rooms and a general practice clinic.
Blood sample collection
Blood was collected from each participant in an ethylenediaminetetraacetic acid (EDTA) tube (Greiner Bio-one). For patients who had not had their ESR measured within the ten days prior to recruitment, additional blood was taken and sent for analysis.
Isolation of peripheral blood mononuclear cells and RNA extraction
PBMC were isolated from whole blood collected in EDTA tubes. Whole blood was diluted in phosphate buffered saline, and Histopaque (Sigma-Aldrich) was used to separate the PBMC from other blood cells. The extracted PBMC were purified using red cell lysis buffer. The purified PBMC pellet was resuspended in a 1:1,000 beta-mercaptoethanol in RLT buffer (Qiagen) solution.
RNA was extracted from lysed PBMC using the phenol-chloroform method. TRIsure (Bioline) was added to the RLT and beta-mercaptoethanol solution containing PBMC. Chloroform was added to the resultant solution, in order to separate the RNA from DNA and proteins. The RNA pellets were left to dry before being resuspended in nuclease free water.
GILZ mRNA measurement by polymerase chain reaction (PCR)
Reverse transcription polymerase chain reaction (RT-PCR) was performed using random primers and Superscript III (Invitrogen) to produce complementary DNA (cDNA). The concentration of cDNA was measured using a Nanodrop spectrophotometer. cDNA dilutions were performed based on Nanodrop measurements. GILZ mRNA expression was quantified using qPCR, with 18S mRNA used as a control. GILZ primers with the following sequences were used: forward 5'-GCACAATTTCTCCATCTCCTTCTT-3' and reverse 5'-TCAGATGATTCTTCACCAGATCCA-3'. A sample of human microvascular endothelial cells (HMEC) in which GILZ mRNA was silenced, and a sample of HMEC overexpressing GILZ were also included on each PCR plate to act as high and low controls for GILZ expression. mRNA was quantified in duplicate samples using SYBR Green (Sigma). GILZ expression was compared to 18S expression using the delta Ct (dCt) method. The resulting dCt values were then normalised to the low GILZ expressing HMEC using the delta delta Ct (ddCt) method, providing a relative numerical measurement of mRNA expression. Of the recruited patients, GILZ expression measurements were obtained for 79 patients.
Statistical Analysis
Results were analysed using IBM SPSS Statistics 20. The normality of continuous variables was assessed using the Kolmogorov-Smirnov and Shapiro-Wilk tests. For normally distributed variables, parametric tests including Pearson correlation and t-test were used. For variables which were not normally distributed, non-parametric tests, including Spearman correlation and Mann-Whitney U test were used. For all statistical analyses, results with a p value less than 0.05 were considered statistically significant.
Results
Demographic information
In total, 178 patients were recruited, including 86 patients with RA, 23 healthy controls, 25 patients with fibromyalgia and 44 patients with depression. Final patient numbers with complete data available for each analysis are displayed graphically. Of note, RA patients were significantly older than controls, but gender distribution did not differ significantly between the two groups. Demographic information regarding RA patients and healthy controls is summarised in table 1.
![]()
Table 1: Demographic information regarding RA patients and healthy controls. The mean age of RA patients was significantly higher than that of controls (independent samples t-test). There was no significant difference in gender distribution between RA patients and controls (Chi-Square test). Abbreviations: SD: Standard Deviation; NS: Not Significant, ** = p < 0.01.
View Table 1
In the RA patient cohort, the mean number of years since RA diagnosis was 13.3. 81.7% of patients were seropositive, with 71.6% positive for rheumatoid factor, and 66.1% positive for anti-CCP antibodies. Additional demographic information regarding the RA patient group is presented in table 2.
![]()
Table 2: Additional demographic information regarding RA patients.
View Table 2
Disease activity in the RA patient cohort was assessed using clinical, laboratory and composite measures. The median CRP level was 4.8 mg/L. 47% of patients had an elevated CRP (greater than 5 mg/L). The median ESR was 16 mm/hr, with 36% of patients having an elevated ESR. The DAS28 score was used as a composite measure of disease activity. The mean DAS28 score was 3.35, which denotes a moderate level of disease activity.
A summary of patient medications is provided in table 3. Of note, 43.5% of patients were taking prednisolone; of patients taking prednisolone, the dose ranged from 1mg to 25 mg daily. 82.6% of patients were taking at least one synthetic DMARD and 37.6% were taking a bDMARD. The most commonly used synthetic DMARD was methotrexate, and the most commonly used bDMARDs were etanercept and adalimumab.
![]()
Table 3: Percentages of the RA patient cohort using prednisolone, DMARDs and bDMARDs.
View Table 3
GILZ expression is higher in RA patients compared to controls
Prior to examining the relationship of GILZ expression with RA, steroid use and disease activity, we first examined the effect of age, gender, smoking, seropositivity and duration of disease on GILZ expression to determine whether these factors could act as confounders. None of these factors correlated significantly with GILZ expression (data not sown). In addition, there was no correlation between GILZ and the individual parameters of CRP or ESR (data not shown).
The RA patient cohort had significantly increased GILZ expression compared to the healthy control cohort, as shown in figure 1A. Moreover RA patients had significantly increased GILZ expression compared to both depression and fibromyalgia patients, as shown in figure 1B.
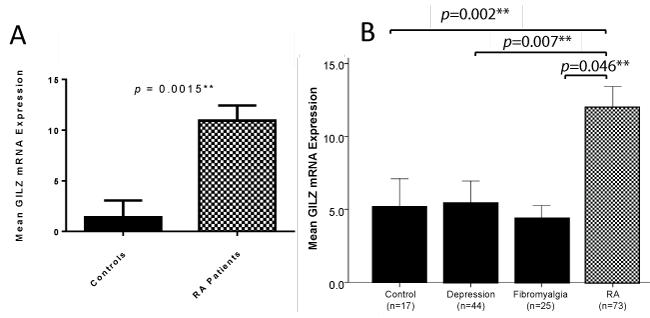
.
Figure 1: A) GILZ mRNA expression was significantly higher in RA patients compared to controls (Mann-Whitney U test). Throughout the results section, GILZ measurements are referred to as GILZ mRNA expression. Values of GILZ mRNA expression were derived using the ddCt method, by which GILZ mRNA expression was normalised to the control gene 18S, and calculated as a measurement relative to a low GILZ expressing HMEC. Error bars represent standard error of the mean, * = p < 0.05, ** = p < 0.01; B) GILZ expression is significantly higher in RA patients than in depression and fibromyalgia patients (Mann-Whitney U test). GILZ expression was unable to be measured in five depression patients and five fibromyalgia patients. The remaining n values for these groups are shown on the graph. Error bars represent standard error of the mean, * = p < 0.05, ** = p < 0.01.
View Figure 1
As GILZ is also known to be induced by glucocorticoids, analysis of GILZ expression in the RA patients in this cohort who were not taking prednisolone was undertaken. GILZ expression was therefore examined with prednisolone users excluded from the analysis. The association between RA and increased GILZ expression persisted although the difference between the two cohorts was less pronounced, as shown in figure 2.
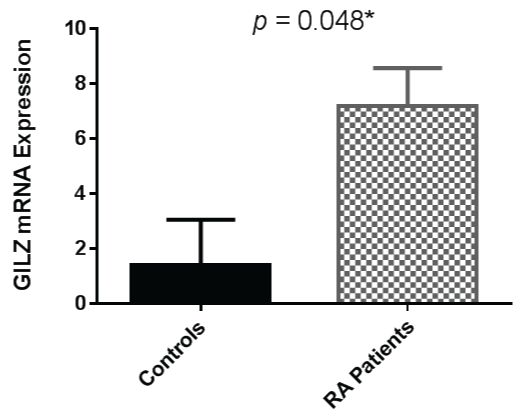
.
Figure 2: GILZ mRNA expression remains significantly higher in RA patients compared to controls when patients taking prednisolone are excluded (Mann-Whitney U Test). Error bars represent standard error of the mean, * = p < 0.05.
View Figure 2
GILZ expression is increased in patients taking prednisolone
We next examined the relationship between GILZ expression and prednisolone use. Prednisolone users had significantly increased GILZ expression compared with those not taking prednisolone, as illustrated in figure 3A. The relationship between prednisolone dose and GILZ expression was next assessed. There was a weak positive but significant correlation between GILZ expression level and prednisolone dose as shown in figure 3B.
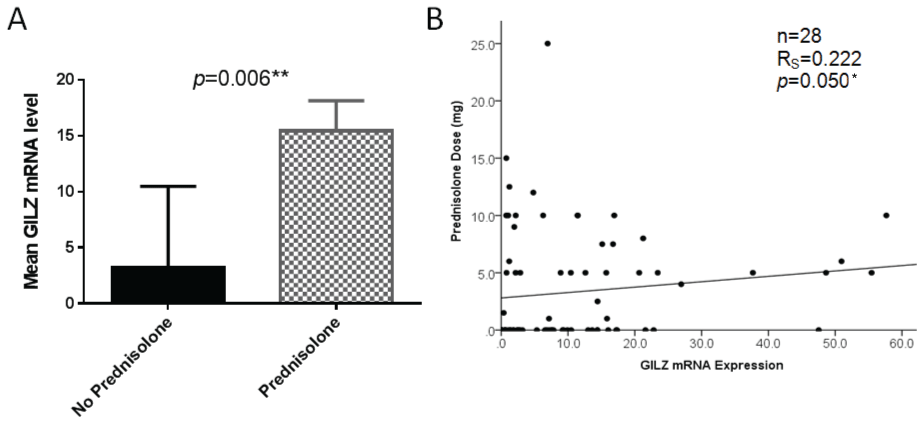
.
Figure 3: A) Significantly higher GILZ mRNA expression was observed in prednisolone users compared with non-users (Mann-Whitney U test). Error bars represent standard error of the mean; B) There is a weak but significant positive correlation between GILZ mRNA expression level and prednisolone dose in all patients taking prednisolone (Spearman's Rho test), * = p < 0.05, ** = p < 0.01.
View Figure 3
GILZ expression correlates negatively with disease activity when adjusted for prednisolone dose
Prior to examining the relationship between GILZ expression and disease activity, we tested the correlation between DMARD and bDMARD use and disease activity, to determine whether these medications could act as confounders. Neither DMRARD nor bDMARD use correlated significantly with disease activity (data not shown). Given that GILZ was shown to be higher in patients taking prednisolone, we adjusted GILZ expression for prednisolone dose prior to examining the correlation with DAS28. There was a strong negative correlation between GILZ expression when adjusted for prednisolone dose and disease activity in RA patients, as shown in figure 4. Interestingly, prednisolone dose itself did not correlate negatively with DAS28 in this study. This may indicate that patients who are better able to up-regulate GILZ in response to prednisolone have decreased disease activity, compared to patients who are taking higher doses of prednisolone without the same GILZ inducibility.
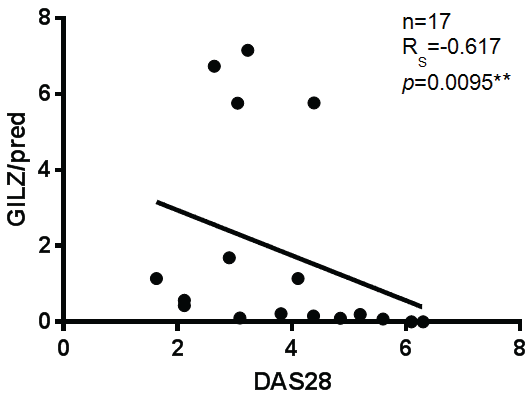
.
Figure 4: There is a strong negative correlation between GILZ adjusted for prednisolone dose and disease activity. (Spearman's Rho Test), ** = p < 0.01.
View Figure 4
Discussion
The demographics of our patients were similar to RA patient cohorts described in the literature. Rates of seropositivity in our cohort were consistent with meta-analyses described in the literature [20]. The mean age and gender distribution in our cohort were similar to the cohorts with which we compared our results [18,21-23]. Importantly, our controls were not age matched to RA patients. Little is known about the effect of age on GILZ expression, however, it is unlikely that older age is responsible for the higher GILZ expression in the RA cohort, given that we found no correlation between GILZ expression and age in either cohort.
Generally, our patients represented chronic, rather than recent onset RA, with a mean of 13.3 years since diagnosis. This was longer than a number of study cohorts with which we compared our results ([18,21-23]. However, given that we found no correlation between chronicity of disease and GILZ expression, this is unlikely to have contributed to differences between our results and those described in the literature.
The mean DAS28 score of 3.34 (moderate disease activity) in our cohort was similar to that in the cohort studied by Quax, et al., who also examined GILZ expression ex vivo in the PBMC of RA patients [18]. The proportion of patients taking prednisolone in our cohort was 43.5%, which is slightly lower than the 50% described in the literature [1,5,8].
GILZ expression in increased in RA patients
Our study showed for the first time that GILZ is higher the PBMC of RA patients compared to controls, independent of prednisolone use. This finding is supported by evidence from previous ex vivo studies. Beaulieu, et al. found that GILZ expression is increased in the synovial sublining and endothelium of RA patients compared to healthy controls, suggesting that increased production of GILZ occurs in response to synovial inflammation in RA [17]. This finding appears to be mirrored in the PBMC of RA patients as demonstrated in our study, suggesting that systemic inflammation also results in up-regulation of GILZ production in the peripheral blood. A possible explanation for this is that the inflammatory stress of RA may activate the HPA axis, culminating in the release of endogenous GC, which induce GILZ production.
However, this finding is not in keeping with the results of several studies which conclude that HPA axis hypofunction occurs in RA. A study by Straub, et al. demonstrated that cortisol levels in RA patients are significantly lower in relation to pro-inflammatory cytokine levels compared to healthy controls [23]. This finding was supported by two studies which demonstrated that RA patients have lower diurnal cortisol levels, and a decreased cortisol response to exercise and surgical stress compared to controls [24,25].
The results of these studies suggest that, in RA, the magnitude of the response of the HPA axis to inflammation is lower than would be expected if it were functioning normally. In contrast, our findings suggest that the HPA axis may be appropriately up-regulated by inflammation in our patient cohort, resulting in increased GILZ production. Another potential explanation for this discrepancy could be that discrete aspects of the HPA axis, for example, responsiveness of GILZ to GC, may be up-regulated in RA to compensate for hypofunction in other parts of the HPA axis, such as cortisol production.
Interestingly, both depression and fibromyalgia are non-inflammatory conditions which are known to activate HPA stress response. The relatively lower GILZ expression in fibromyalgia and depression groups similar to healthy controls found in our study may indicate either those HPA responses are blunted in these conditions or that inflammatory stress constitutes a more powerful stimulus to GILZ via HPA and non-HPA mediated mechanisms. These findings are supported by a study by Macedo, et al., which found lower levels of GILZ expression in fibromyalgia patients compared to controls, and a study by Frodl, et al., which found no difference in GILZ expression between depression patients and controls [26,27]. Conversely, two meta-analyses have demonstrated increased GILZ expression in patients with depression [28,29]. However, these studies mainly recruited from inpatient settings, therefore patients were likely to have more severe disease, which may have resulted in greater activation of the stress response and up-regulation of the HPA axis.
GILZ expression is increased in patients taking prednisolone
GILZ expression is known to be exquisitely sensitive to upregulation by GC [17]. In our cohort, prednisolone use correlated with higher GILZ expression, and there was a weak positive correlation between GILZ expression and prednisolone dose. Although there are no previous studies which have directly examined the effect of exogenous GC administration on GILZ expression in humans with RA in vivo, this finding is well supported by the current literature. Beaulieu, et al. demonstrated that administration of dexamethasone (DEX) to cultured human RA fibroblast-like synoviocytes resulted in a dramatic, dose-dependent increase in GILZ expression [17]. In addition, they found that mice with inflammatory arthritis who were treated with DEX had higher GILZ expression in their spleen and joints than controls [17].
Furthermore, GILZ expression has been found to be up-regulated by ex vivo DEX administration to PBMC isolated from both RA patients and healthy controls [18]. Therefore, it would be logical to suggest that exposure of peripheral blood cells to exogenous GC in patients taking prednisolone would result in increased GILZ production, as was demonstrated in our study.
A study by Hamdi, et al. measured GILZ expression in response to prednisolone treatment in humans in vivo for the first time [13]. GILZ expression in the monocytes of patients with alcoholic hepatitis was measured before and after 48 hours of prednisolone treatment [13]. GILZ expression was significantly increased by prednisolone treatment in these patients [13]. Although patients in this study did not have RA, and were treated with prednisolone for a far shorter period of time than the patients in our cohort, the results do support our findings that GILZ expression in peripheral blood is increased by prednisolone treatment in clinical settings.
GILZ expression correlates negatively with disease activity when adjusted for prednisolone dose
Prednisolone dose itself did not correlate with disease activity in our cohort. This may be explained by high concurrent use of synthetic and bDMARDs, which themselves would modify disease activity, as well as the low to moderate disease activity in the cohort overall. When GILZ expression was adjusted for prednisolone dose, however, a strong negative correlation with disease activity was observed. This is the first time that GILZ has been measured in vivo in an RA cohort in relation to prednisolone use and disease activity. This finding suggests that the inducibilty of GILZ by prednisolone rather than prednisolone dose per se may be a better indicator of treatment responsiveness.
Our findings are supported by a study by Quax, et al., which aimed to correlate the response to prednisolone administration in vivo (measured by improvement in DAS28 score) in RA patients, with the amount of DEX needed to induce a particular level of GILZ expression in vitro in PBMC extracted from the patients [18]. They found that patients with a greater improvement in DAS28 score after prednisolone treatment required a lower dose of DEX to induce the target level of GILZ expression, although this result was not statistically significant [18]. Therefore, patients whose PBMC were able to induce GILZ expression more effectively in response to DEX in vitro were more likely to respond to GC treatment in vivo.
In conclusion, we have shown that GILZ is increased in the peripheral blood of RA patients, and even more so in patients taking prednisolone. We have also demonstrated that GILZ expression correlates negatively with disease activity when adjusted for prednisolone dose. Our results suggest that measuring GILZ inducibility in response to prednisolone may be a useful way to determine which patients are likely to experience significant improvements in disease activity with relatively low doses of corticosteroids.
Ethical Statement
Institutional Human Research Ethics Committee approval was obtained prior to collection of data.
References
-
Beaulieu E, Morand EF (2011) Role of GILZ in immune regulation, glucocorticoid actions and rheumatoid arthritis. Nat Rev Rheumatol 7: 340-348.
-
Lekva T, Bollerslev J, Kristo C, Olstad OK, Ueland T, et al. (2010) The glucocorticoid-induced leucine zipper gene (GILZ) expression decreases after successful treatment of patients with endogenous Cushing's syndrome and may play a role in glucocorticoid-induced osteoporosis. J Clin Endocrinol Metab 95: 246-255.
-
Lee DM, Weinblatt ME (2001) Rheumatoid arthritis. Lancet 358: 903-911.
-
(2012) Australian Bureau of Statistics. Profiles of Health, Australia.
-
Fan H, Morand EF (2012) Targeting the side effects of steroid therapy in autoimmune diseases: the role of GILZ. Discov Med 13: 123-133.
-
Criswell LA, Saag KG, Sems KM, Welch V, Shea B, et al. (2000) Moderate-term, low-dose corticosteroids for rheumatoid arthritis. Cochrane Database Syst Rev 2000: CD001158.
-
Pisu M, James N, Sampsel S, Saag KG (2005) The cost of glucocorticoid-associated adverse events in rheumatoid arthritis. Rheumatology (Oxford) 44: 781-788.
-
Huscher D, Thiele K, Gromnica-Ihle E, Hein G, Demary W, et al. (2009) Dose-related patterns of glucocorticoid-induced side effects. Ann Rheum Dis 68: 1119-1124.
-
Saag KG, Koehnke R, Caldwell JR, Brasington R, Burmeister LF, et al. (1994) Low dose long-term corticosteroid therapy in rheumatoid arthritis: an analysis of serious adverse events. Am J Med 96: 115-123.
-
Cannarile L, Fallarino F, Agostini M, Cuzzocrea S, Mazzon E, et al. (2006) Increased GILZ expression in transgenic mice up-regulates Th-2 lymphokines. Blood 107: 1039-1047.
-
Cannarile L, Cuzzocrea S, Santucci L, Agostini M, Mazzon E, et al. (2009) Glucocorticoid-induced leucine zipper is protective in Th1-mediated models of colitis. Gastroenterology 136: 530-541.
-
Cohen N, Mouly E, Hamdi H, Maillot MC, Pallardy M, et al. (2006) GILZ expression in human dendritic cells redirects their maturation and prevents antigen-specific T lymphocyte response. Blood 107: 2037-2044.
-
Hamdi H, Bigorgne A, Naveau S, Balian A, Bouchet-Delbos L, et al. (2007) Glucocorticoid-induced leucine zipper: A key protein in the sensitization of monocytes to lipopolysaccharide in alcoholic hepatitis. Hepatology 46: 1986-1992.
-
Wang Y, Ma YY, Song XL, Cai HY, Chen JC, et al. (2012) Upregulations of glucocorticoid-induced leucine zipper by hypoxia and glucocorticoid inhibit proinflammatory cytokines under hypoxic conditions in macrophages. J Immunol 188: 222-229.
-
Zhang W, Yang N, Shi XM (2008) Regulation of mesenchymal stem cell osteogenic differentiation by glucocorticoid-induced leucine zipper (GILZ). J Biol Chem 283: 4723-4729.
-
Ngo D, Beaulieu E, Gu R, Leaney A, Santos L, et al. (2013) Divergent Effects of Endogenous and Exogenous Glucocorticoid-Induced Leucine Zipper in Animal Models of Inflammation and Arthritis. Arthritis Rheum 65: 1203-1212.
-
Beaulieu E, Ngo D, Santos L, Yang YH, Smith M, et al. (2010) Glucocorticoid-induced leucine zipper is an endogenous antiinflammatory mediator in arthritis. Arthritis Rheum 62: 2651-2661.
-
Quax R, Koper J, de Jong P, van Heerebeek R, Weel A, et al. (2012) In vitro glucocorticoid sensitivity is associated with clinical glucocorticoid therapy outcome in rheumatoid arthritis. Arthritis Res Ther 14: R195.
-
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, et al. (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62: 2569-2581.
-
Nishimura K, Sugiyama D, Kogata Y, Tsuji G, Nakazawa T, et al. (2007) Meta-analysis: diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis. Ann Intern Med 146: 797-808.
-
Gottenberg JE, Dayer JM, Lukas C, Ducot B, Chiocchia G, et al. (2012) Serum IL-6 and IL-21 are associated with markers of B cell activation and structural progression in early rheumatoid arthritis: results from the ESPOIR cohort. Ann Rheum Dis 71: 1243-1248.
-
Huang B, Wang QT, Song SS, Wu YJ, Ma YK, et al. (2012) Combined use of etanercept and MTX restores CD4+/CD8+ ratio and Tregs in spleen and thymus in collagen-induced arthritis. Inflamm Res 61: 1229-1239.
-
Straub RH, Paimela L, Peltomaa R, Scholmerich J, Leirisalo-Repo M (2002) Inadequately low serum levels of steroid hormones in relation to interleukin-6 and tumor necrosis factor in untreated patients with early rheumatoid arthritis and reactive arthritis. Arthritis Rheum 46: 654-662.
-
Chikanza IC, Petrou P, Kingsley G, Chrousos G, Panayi GS (1992) Defective hypothalamic response to immune and inflammatory stimuli in patients with rheumatoid arthritis. Arthritis Rheum 35: 1281-1288.
-
Pool AJ, Whipp BJ, Skasick AJ, Alavi A, Bland JM, et al. (2004). Serum cortisol reduction and abnormal prolactin and CD4+/CD8+ T-cell response as a result of controlled exercise in patients with rheumatoid arthritis and systemic lupus erythematosus despite unaltered muscle energetics. Rheumatology (Oxford). 43: 43-48.
-
Frodl T, Carballedo A, Hughes MM, Saleh K, Fagan A, et al. (2012) Reduced expression of glucocorticoid-inducible genes GILZ and SGK-1: high IL-6 levels are associated with reduced hippocampal volumes in major depressive disorder. Transl Psychiatry 2: e88.
-
Macedo JA, Hesse J, Turner JD, Meyer J, Hellhammer DH, et al. (2008) Glucocorticoid sensitivity in fibromyalgia patients: decreased expression of corticosteroid receptors and glucocorticoid-induced leucine zipper. Psychoneuroendocrinology 33: 799-809.
-
Knorr U, Vinberg M, Kessing LV, Wetterslev J (2010) Salivary cortisol in depressed patients versus control persons: a systematic review and meta-analysis. Psychoneuroendocrinology 35: 1275-1286.
-
Stetler C, Miller GE (2011) Depression and hypothalamic-pituitary-adrenal activation: a quantitative summary of four decades of research. Psychosom Med 73: 114-126.





