Journal of Otolaryngology and Rhinology
Characterization of Variant Periodic Fever Phenotypes
Michael D Puricelli1, Christopher I Newberry2, Ryan M Neff3, Michael S Cooperstock4 and Eliav Gov-Ari1*
1Department of Otolaryngology - Head & Neck Surgery, University of Missouri, USA
2University of Missouri School of Medicine, USA
3Department of Otolaryngology - Head & Neck Surgery, University of Kentucky, USA
4Department of Child Health, Division Infectious Disease, Immunology & Rheumatology, University of Missouri, USA
*Corresponding author:
Eliav Gov-Ari, MD, Department of Otolaryngology - Head & Neck Surgery, University of Missouri, One Hospital Dr MA314 Columbia, MO 65212, Missouri, USA, Tel: (573) 882-8174, E-mail:
govarie@health.missouri.edu
J Otolaryngol Rhinol, JOR-2-020, (Volume 2, Issue 3), Research Article
Received: February 16, 2016: Accepted: May 20, 2016: Published: May 23, 2016
Citation: Puricelli MD, Newberry CI, Neff RM, Cooperstock MS, Gov-Ari E (2016) Characterization of Variant Periodic Fever Phenotypes. J Otolaryngol Rhinol 2:020.
Copyright: © 2016 Puricelli MD, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:Diagnostic criteria for periodic fever, aphthous stomatitis, pharyngitis, and cervical adenitis syndrome (PFAPA) have been challenged, resulting in calls for reclassification. We explore the natural history and treatment response among PFAPA and incomplete periodic fever variants to assess existing therapies among excluded individuals.
Methods:Periodic fever cases were retrospectively reviewed. A pediatric infectious disease specialist excluded alternative diagnoses and telephone follow-up confirmed data fidelity.
Results:469 charts were reviewed and 31 potential cases identified. 29 individuals were included with 20 successfully contacted. 11 cases met the classic definition (CD), 9 had intermittent cardinal symptom(s) (IS), and 9 had isolated periodic fever (IPF). Treatment included observation, steroid administration, and/or surgery (tonsillectomy or adenotonsillectomy). Steroids terminated each episode in all treated patients, 9/11 with CD and 11/18 with IS/IPF. When steroid treatment shortened the inter-episode interval, higher doses reversed this effect. With observation or steroid treatment, either spontaneous resolution occurred after a mean of 105, 54, and 36 months or episodes persisted after a mean of 18, 53, and 57 months for CD, IS, and IPF, respectively. Surgery resulted in a sustained cure for 4 patients in each group. Surgical treatment reduced total disease duration in the CD and IPF groups p < 0.01. No individual suffered long-term adverse effects.
Conclusions:Treatment of PFAPA and incomplete variants with steroids reliably terminates acute episodes. Surgical treatment alters the natural history of CD and IPF. PFAPA treatments should be further investigated for IS and IPF and further research is needed to definitively characterize these groups.
Keywords
Periodic fever, PFAPA, Tonsillectomy
Key point
PFAPA is the leading cause of childhood periodic fever. Treatment includes steroids and/or tonsillectomy. Recently, diagnostic criteria have been challenged. We report corticosteroid efficacy in PFAPA and incomplete variants. Tonsillectomy significantly alters the disease natural history in both groups.
Introduction
First reported by Marshall et al. in 1987, periodic fever, aphthous stomatitis, pharyngitis, and cervical adenitis syndrome (PFAPA) is the most common cause of periodic fever in children [1-3]. While PFAPA is a disorder of unknown etiology, local immune dysfunction or a microbial reservoir in the tonsils are proposed as possible mechanisms underlying episodes [2,4]. The incidence is reported as 2.3 per 10,000 children under age 5 years, with male predominance [5]. Diagnosis is made on clinical findings: (1) regularly recurring fevers beginning under age five; (2) constitutional symptoms without upper respiratory infection with at least one cardinal symptom (aphthous stomatitis, cervical adenitis, pharyngitis); (3) exclusion of cyclic neutropenia; (4) asymptomatic between episodes; and (5) normal growth and development [6]. Periodic fever is the hallmark of the syndrome, with temperature typically reaching 39°C or higher [4]. Each case may have its own predictable "clockwork" periodicity, usually 3-6 weeks, and duration, typically 3-7 days [4,5]. Cardinal symptoms, including aphthous stomatitis, pharyngitis, and cervical adenitis are present at variable rates, with reports ranging from 38-75%, 65-100%, and 61-100%, respectively [2,4,5,7]. Associated symptoms including headache, fatigue, vomiting, abdominal pain, and joint pain have been reported [4-6,8].
The natural history of PFAPA is a benign course without long-term complications [5,8]. Skipped episodes are observed occasionally [6-8]. Long-term follow-up has been reported in one series with spontaneous resolution in 50 of 59 patients at a mean of 6.3 years [8]. Nine individuals experienced persisting episodes at a mean follow-up duration of 18.1 years [8]. Others have reported shorter mean duration to spontaneous resolution; however a lower percentage of patients experienced resolution in these series [5,6].
Treatment is usually recommended since symptoms can be uncomfortable and disrupt family life. Cimetidine has been investigated but demonstrated limited efficacy [6-8]. Oral corticosteroids are highly effective, providing rapid termination of episodes in 84-97% of patients [4,7,8]. In up to 50% of cases, however, steroid treatment has been associated with shortening of disease-free interval, leading to frequent use in some individuals [4,7]. This can often be corrected by increasing the steroid dosage. Adverse effects have also been reported in up to one-third of children [9].
A Cochrane review of surgical therapy (tonsillectomy or adenotonsillectomy) identified two randomized controlled studies involving 67 children with a 4.38 pooled relative risk of symptom resolution [10-12]. Surgical candidacy in 2010 was proposed to require: (1) onset before age 5 years; (2) regular recurring abrupt episodes of fever lasting about five days with at least one characteristic feature (cardinal symptom) but without other signs of respiratory tract infection; (3) prompt remission of symptoms after corticosteroid administration and completely asymptomatic interval; and (4) normal growth and development [13]. A final criterion proposed to select the best candidates includes at least five prior episodes characterized by high fever and unknown origin [13]. The potential benefit of surgical therapy is reflected in the 2011 American Academy of Otolaryngology Head & Neck Surgery (AAO-HNS) Clinical Practice Guideline: Tonsillectomy in Children [14]. This guideline recommends that clinicians should assess children with recurrent throat infections for PFAPA, and that tonsillectomy may be considered in certain cases [14]. Adenoidectomy, while performed in conjunction with tonsillectomy in some series, is not curative independently and is felt by some to provide no additional value compared with tonsillectomy alone [15,16]. Since publication of the AAO-HNS guideline, results of a literature search from 1987-2010 for tonsillectomy or adenotonsillectomy for PFAPA syndrome identified 15 studies and 149 children [17]. Operation yielded a pooled complete resolution rate of 83% and a meta-analysis of two randomized controlled trials revealed an odds ratio for complete resolution of 13 [17]. In a separate series, Licameli et al. reported complete resolution in 99/102 patients following adenotonsillectomy for PFAPA [4].
As evidence for therapeutic options expanded, the need for consistent diagnostic criteria has increasingly become evident. Different sets of diagnostic criteria have been used in 10 cohorts with at least 25 PFAPA patients since first described by Marshall in 1987 [18]. Individuals with onset after 5 years of age are included in some series and an adult onset phenotype has been proposed [4,19]. Surgical therapy has been provided to individuals with periodic fever in the absence of cardinal symptoms [11]. A web-based multicenter cohort reviewed 301 cases, defining discrete phenotypic cohorts of PFAPA: a complete cluster and incomplete cluster [18]. Based on review of their data, Hofer et al. reported that the "diagnosis of PFAPA is generally based upon a number of variables…that are much more complex than the mere satisfaction of the current diagnostic criteria" [18].
We retrospectively reviewed cases of periodic fever at the University of Missouri to assess natural history and treatment response among patients with PFAPA and variants excluded by existing diagnostic criteria.
Patients and Methods
Protocol and data collection
Periodic fever cases were identified via retrospective chart review. As no established diagnosis code for PFAPA was available during the chart review period, Institutional Review Board approval was obtained for review of patient encounters within child health, pediatric infectious disease, family medicine, and otolaryngology head and neck surgery with diagnosis codes 023.9 (Brucellosis NOS), 077.2 (Pharyngoconjunctival fever), 078.4 (Foot & mouth disease), 087.9 (Relapsing fever NOS), 277.31 (Familial Mediterranean fever), or 780.60 (Fever NOS) treated within the University of Missouri Healthcare System after the implementation of the electronic medical record, from January 2004 to September 2013. The returned records were independently reviewed by two authors (MDP, RMN) to identify the maximum number of relevant cases and ensure accuracy of recorded data. Among those with disagreement, an additional review was completed to reconcile differences.
Medical record review
The following data points were extracted from medical records: (1) Febrile episode onset, duration, interval and course; (2) Age of onset; (3) Presence and frequency of cardinal and accompanying symptoms, classified as always, sometimes, or never present; (4) Treatment received, outcome, and adverse effects of treatment; (5) Length of follow-up; (6) Age appropriate height and weight; (7) Pediatric infectious disease specialist exclusion of differential diagnoses; (8) Family history of periodic fever syndrome. Differential diagnoses were primarily excluded clinically using generally accepted criteria [6,7]. Supplemental laboratory testing, applied in selected cases, included immunoglobulin levels and genetic testing (Periodic Fever Panel, Gene Dx, Inc.) to exclude polymorphisms in the genes ELANE (ELA2) for Cyclic Neutropenia and Severe Congenital Neutropenia; LPIN2 for Majeed Syndrome; MEFV for Familial Mediterranean Fever; MVK for Hyper-IgD Syndrome, Mevalonate Kinase Deficiency, and Mevalonic Aciduria; NLRP3 (CIAS1)for Chronic Infantile Neurologic Cutaneous and Articular Syndrome, Familial Cold Auto inflammatory Syndrome, Muckle-Wells Syndrome, Neonatal Onset Multisystem Inflammatory Disease; PSTPIP1 for Pyogenic Sterile Arthritis, Pyoderma Gangrenosum, and Acne Syndrome; and TNFRSF1A for Familial Hibernian Fever and TNF Receptor-Associated Periodic Syndrome. When conflicting information about the presence of symptoms was documented, telephone follow-up data was used to reconcile differences. In the case of conflicting data without telephone follow-up data, the preoperative evaluation was accepted. Refer to table 1 for inclusion criteria.
![]()
Table 1: Inclusion criteria.
View Table 1
Classification of periodic fever
Patients were classified into one of four groups based on chart review: (1) Complete phenotype with all cardinal symptoms present at the time of each periodic fever episode; (2) Incomplete phenotype with at least one of the cardinal symptoms present at the time of each periodic fever episode; (3) Incomplete phenotype with intermittent cardinal symptom(s) (all cardinal symptoms absent during some periodic fever episodes) (IS); (4) Isolated periodic fever with no cardinal symptoms during periodic fever episodes (IPF). For the purposes of data analysis, groups 1 and 2, which meet the traditional definition of PFAPA, were combined into the classic definition (CD) group.
Telephone follow-up
To ensure the accuracy of data and extend follow-up, additional Institutional Review Board approval was obtained for telephone follow-up with families of identified cases. A standardized survey script was used, and data was gathered by a single author (CIN). Data was compiled and differences were reconciled with re-review of the medical record and additional discussion with family.
Treatment
Individuals received no treatment (observation), oral corticosteroid, or surgical therapy. At the start of the study period, individuals treated with steroids were given prednisolone or prednisone 1 mg/kg/dose twice daily for two days beginning at the onset of symptoms. Subsequently, dosing was changed to a single 1-2 mg/kg dose, with an option for a repeat dose after 12 hours. Extracapsular tonsillectomy or adenotonsillectomy was performed in each surgical case.
Outcome assessment
Resolution was defined as the cessation of all febrile episodes. Termination was defined as the abrupt end of an episode, regardless of subsequent occurrences. Recurrence was defined as the return of periodic fever episodes after a hiatus of one or more episodes. Regardless of past recurrences, individuals were classified in the resolution group if ongoing episodes cease.
The CD, IS, and IPF groups were assessed separately. The febrile episode characteristics, clinical findings, treatment response, and natural history were documented. The impact of tonsillectomy or adenotonsillectomy on total symptom duration was compared to non-surgical treatments within each diagnostic group.
Statistical analysis
Descriptive statistics are used to summarize data and stratify response to treatment. ANOVA testing with Bonferroni correction (SigmaPlot) compared multiple groups and two sided t-test (Excel) compared two groups. Statistical significance was defined by p < 0.05.
Results
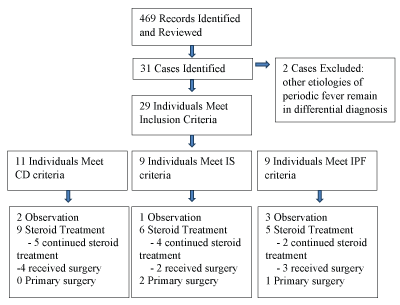
Four hundred sixty nine records were returned and reviewed. Thirty one possible cases were identified with two excluded because other etiologies of periodic fever had not been fully ruled out (Figure 1). Genetic testing was negative in all cases in which it was performed. All patient families were telephoned and twenty of twenty nine (69%) were successfully reached including 7/11 in the CD group, 6/9 in the IS group and 7/9 in the IPF group. One patient was reclassified from the IPF group to the IS group. In the 19 other cases (95%), appropriate classification was confirmed.
.
Figure 1: Patient flow diagram. CD: Classic definition; IS: Incomplete sometimes cardinal symptom; IPF: Isolated periodic fever
View Figure 1
Patient demographics
The mean age of onset was 2.2 years. Twenty three of twenty nine patients (79%) were male. The mean duration of febrile episodes was 4.8 days and interval between episodes 3.4 weeks. All febrile episodes were predictably spaced. Individuals were well between each episode, and every patient showed normal growth and development. Other causes of periodic fever were excluded by one pediatric infectious disease specialist (MSC). Refer to table 2 for febrile episode characteristics.
![]()
Table 2: Febrile episode characteristics. CD: Classic definition; IS: Incomplete sometimes cardinal symptom; IPF: Isolated periodic fever
View Table 2
Clinical findings
Eleven patients were identified in the CD group. One demonstrated the complete phenotype, while ten had an incomplete phenotype with at least one cardinal symptom present at each episode. Nine patients were categorized in the IS group and nine in the IPF group. Refer to table 3 for the rate of associated symptoms.
![]()
Table 3: Clinical findings. CD: Classic definition; IS: Incomplete sometimes cardinal symptom; IPF: Isolated periodic fever; A: always; S: sometimes
View Table 3
Family history
Five patients reported family history of apparently similar episodes: two meeting criteria for CD and three for the IPF group. The episodes were noted in a parent in three cases and in maternal/paternal cousins in two cases. Family history was negative for diagnosed syndromes of periodic fever.
Treatment response
Among the eleven patients in the CD group, two were observed and nine received steroids. Steroid treatment immediately terminated episodes in 100% of individuals. Frequency of episodes increased in two individuals and decreased in one patient. In the two patients with increased frequency of episodes, the prednisolone dose was increased from 1 mg/kg/dose twice daily for two days to a single 2 mg/kg/dose with the option for a second dose 12 hours later. In both cases, the original interval between episodes was restored. No adverse effects of steroid treatment were reported. Three patients initially treated with steroids went on to undergo adenotonsillectomy and one underwent tonsillectomy. Surgical therapy was immediately curative in all four patients. Mean follow-up duration after surgical treatment was 92 months.
Among nine patients in the IS group, one was observed and six treated with steroids. Steroids were abortive in 100% of individuals treated. Only one individual treated with steroids experienced change in frequency of episodes, which was an increased interval between occurrences. There were no reported adverse effects from steroid treatment. Adenotonsillectomy was the primary treatment modality in two individuals. Two others, initially treated with steroids, later received adenotonsillectomy. Surgery was immediately curative in all four individuals. Mean follow-up after surgery was 41 months.
In the IPF group, three patients were observed and five treated with steroids. Steroids were immediately effective in all five treated patients. There was no reported change in frequency of events with steroid treatment. One patient discontinued prednisolone after experiencing hallucinations with steroid treatment; another discontinued it due to parental concern for adverse effects from long-term use. Among the five initially treated with steroids, two later received adenotonsillectomy and one received tonsillectomy. One additional patient proceeded directly to adenotonsillectomy without steroid trial. Immediate cure was observed in 100% of operated patients. Mean post-surgical follow-up was 52 months. One patient who underwent observation received adenoidectomy for an unrelated reason and no impact was noted in the periodic fever episodes.
There were no complications from surgical treatment in any group. Refer to table 4 for treatment response data.
![]()
Table 4: Treatment response. CD: Classic definition; IS: Incomplete sometimes cardinal symptom; IPF: Isolated periodic fever
View Table 4
Natural history
Seven patients in the CD group were managed solely with steroids or observed. Four of the seven experienced spontaneous resolution of symptoms during the study period at a mean duration of 105 months. Three continued to experience episodes with mean follow-up of 18 months. Relapse occurred in four of seven individuals, including three who later experienced spontaneous resolution. The surgically treated patients, who had experienced mean symptom duration of 37 months prior to operation, all were immediately cured by the procedure, p < 0.006, compared to those with spontaneous resolution.
Five patients in the IS group were managed solely with steroids or observed. Three experienced spontaneous resolution at a mean duration of 54 months. The remaining two individuals continued to have episodes at a mean follow-up duration of 53 months. Relapse occurred in one of five individuals, with subsequent resolution. The surgically treated patients experienced mean symptom duration of 31 months prior to immediate post-procedure cure.
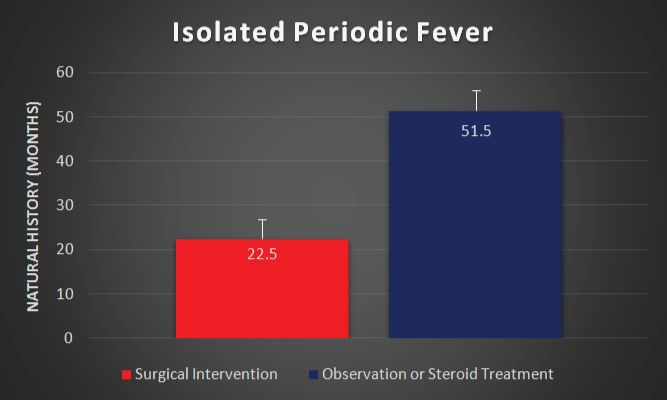
Five patients in the IPF group received steroids or were observed without surgical intervention. Spontaneous resolution was seen in one individual after 36 months. Four continued to experience episodes at a mean duration of 57 months. Relapse occurred in all four individuals with ongoing episodes. The surgically treated individuals experienced mean symptom duration of 23 months prior to immediate post-operative cure, p < 0.003 compared to individuals with spontaneous resolution or ongoing episodes (Figure 2).
.
Figure 2: Duration of ongoing episodes, representing the natural history of isolated periodic fever, among individuals who received steroid treatment or observation (with ongoing episodes or resolution) is compared to surgical treatment. Surgical treatment alters the natural history of isolated periodic fever (p < 0.003). Error bars demonstrate the standard error of the mean.
View Figure 2
Discussion
We report eleven cases meeting suggested PFAPA diagnostic criteria and 18 otherwise identical cases of periodic fever with partial or no expression of the required cardinal symptoms [6,20]. Interestingly, the clinical presentation, natural history and, most importantly, response to treatment of the variant periodic fever cases resembled those of PFAPA.
The frequency of cardinal symptoms in our CD patients was similar to previously reported rates [2,4,5,7]. Aphthous stomatitis, pharyngitis and cervical adenitis were present always/sometimes in 27/18%, 100/0%, and 55/27% of patients, respectively. Only one of the eleven patients had the complete cluster with all symptoms present with every episode. In contrast, 40.8% of patients in the Hofer et al series had the complete phenotype [18]. As expected, the IS and IPF groups had lower rates of associated symptoms. Because symptoms in our series are classified as always, sometimes, or never as compared to less precise classification schemes, direct comparison of frequency between studies is difficult. Further, the imprecision of existing criteria might have led some patients with IS phenotype to have been reported in other series as part of PFAPA while others were excluded for not meeting strict diagnostic criteria. There are few other cases of IS and IPF reported in the literature [11,21,22].
CD, IS, and IPF in our series each showed persistent symptoms for years, without long-term complications. Among those followed until resolution, in CD, symptoms persisted for approximately 8.8 years from initial onset, similar to the reported duration of 6.3 years in the literature [8]. IS was 54 months and IPF was 36 months. It is noteworthy that patients with ongoing symptoms have experienced episodes for a mean of 57 months in the IPF group suggesting a longer total duration of illness among these patients.
Steroid treatment was remarkably effective, with 100% of patients experiencing immediate termination, whether CD, IS, or IPF. Within the study period, our practice pattern shifted from four steroid doses administered over two days to a single 2 mg/kg dose of prednisolone or prednisone, with an option for a repeat dose 12 hours later. Both prescriptions were fully effective for termination of episodes. Asymptomatic interval shortening was reversed using the higher dose regimen. Adverse effects were reported in only 10% of patients, a rate lower than reported by others [9].
Tonsillectomy, in two patients, or adenotonsillectomy, in ten patients, led to cure in 100% of individuals in each clinical group. A single patient underwent adenoidectomy alone for an unrelated reason with no impact on the course of periodic fever. These findings are consistent with the theory that tonsils and not adenoids are in some way critical to the pathophysiology of the PFAPA syndrome [2,4]. Importantly, it also suggests that the tonsils could be equally involved in the pathophysiology of IS and IPF. This appears to be the first study to demonstrate a statistically significant cure for IPF in response to tonsillectomy or adenotonsillectomy. This is consistent with the findings from the only other series to include patients with isolated periodic fever, where cure was achieved in 7/7 patients with tonsillectomy [11].
While the pathophysiology of CD, IS, and IPF remains to be delineated, we believe the salient features to identify patients who might benefit from treatment should include characteristic febrile episodes, predictable periodicity, and exclusion of other etiologies of periodic fever by a provider with experience in the diagnosis of such conditions. Oral steroid treatment is highly effective and carries a low risk profile. Tonsillectomy as an alternative provides an opportunity for prompt cure of the disorder. If tonsillectomy is to be considered, it is our practice to recommend that a therapeutic trial of steroids be undertaken first. A positive response in the appropriate clinical setting strongly supports the diagnosis of periodic fever and may predict surgical success.
There are several limitations to our data. First, the retrospective nature of our study resulted in omissions of data in some cases and introduced a risk for recall bias. We attempted to mitigate chart review errors by using a two reviewer protocol and by cross-referencing clinical findings from multiple providers treating each patient. Further, telephone follow-up was used to confirm the accuracy of findings and extend follow-up duration. Conclusions are limited because of relatively small sample size. Nevertheless, the data suggest a need to further investigate PFAPA therapeutic modalities among incomplete variants and consider whether IS and IPF represent a spectrum of PFAPA or unique entities.
Conclusion
We report two incomplete periodic fever variants that otherwise closely resemble PFAPA, with similar clinical presentation, natural history and response to medical and surgical therapy. Further diagnostic and therapeutic research should apply proven PFAPA treatments to IS and IPF and they should be considered as candidate variants of PFAPA as new classification schemes are defined. For such studies, prospective collaborative efforts should be considered.
Acknowledgments
Kurt Marshall, MS for statistical assistance.
References
-
Marshall GS, Edwards KM, Butler J, Lawton AR (1987) Syndrome of periodic fever, pharyngitis, and aphthous stomatitis. J Pediatr 110: 43-46.
-
Valenzuela PM, Araya A, Pérez CI, Maul X, Serrano C, et al. (2013) Profile of inflammatory mediators in tonsils of patients with periodic fever, aphthous stomatitis, pharyngitis, and cervical adenitis (PFAPA) syndrome. Clin Rheumatol 32: 1743-1749.
-
Marshall GS (2014) Prolonged and recurrent fevers in children. J Infect 68 Suppl 1: S83-93.
-
Licameli G, Lawton M, Kenna M, Dedeoglu F (2012) Long-term surgical outcomes of adenotonsillectomy for PFAPA syndrome. Arch Otolaryngol Head Neck Surg 138: 902-906.
-
Forsvoll J, Kristoffersen EK, Oymar K (2013) Incidence, clinical characteristics and outcome in Norwegian children with periodic fever, aphthous stomatitis, pharyngitis and cervical adenitis syndrome; a population-based study. Acta Paediatr 102: 187-192.
-
Thomas KT, Feder HM Jr, Lawton AR, Edwards KM (1999) Periodic fever syndrome in children. J Pediatr 135: 15-21.
-
Feder HM, Salazar JC (2010) A clinical review of 105 patients with PFAPA (a periodic fever syndrome). Acta Paediatr 99: 178-184.
-
Wurster VM, Carlucci JG, Feder HM Jr, Edwards KM (2011) Long-term follow-up of children with periodic fever, aphthous stomatitis, pharyngitis, and cervical adenitis syndrome. J Pediatr 159: 958-964.
-
Tasher D, Somekh E, Dalal I (2006) PFAPA syndrome: new clinical aspects disclosed. Arch Dis Child 91: 981-984.
-
Burton MJ, Pollard AJ, Ramsden JD, Chong LY, Venekamp RP (2014) Tonsillectomy for periodic fever, aphthous stomatitis, pharyngitis and cervical adenitis syndrome (PFAPA). Cochrane Database Syst Rev 9: CD008669.
-
Renko M, Salo E, Putto-Laurila A, Saxen H, Mattila PS, et al. (2007) A randomized, controlled trial of tonsillectomy in periodic fever, aphthous stomatitis, pharyngitis, and adenitis syndrome. J Pediatr 151: 289-292.
-
Garavello W, Romagnoli M, Gaini RM (2009) Effectiveness of adenotonsillectomy in PFAPA syndrome: a randomized study. J Pediatr 155: 250-253.
-
Kuppersmith RB, Schams ST, Rosenfeld RM (2010). Extracts from The Cochrane Library: Tonsillectomy for periodic fever, aphthous stomatitis, pharyngitis and cervical adenitis syndrome (PFAPA). Otolaryngol Head Neck Surg 143: 473-475.
-
Baugh RF, Archer SM, Mitchell RB, Rosenfeld RM, Amin R, et al. (2011) Clinical practice guideline: tonsillectomy in children. Otolaryngol Head Neck Surg 144: S1-30.
-
Peridis S, Koudoumnakis E, Theodoridis A, Stefanaki K, Helmis G, et al. (2010) Surgical outcomes and histology findings after tonsillectomy in children with periodic fever, aphthous stomatitis, pharyngitis, and cervical adenitis syndrome. Am J Otolaryngol 31: 472-475.
-
Wong KK, Finlay JC, Moxham JP (2008) Role of Tonsillectomy in PFAPA Syndrome. Arch Otolaryngol Head Neck Surg 134: 16-19.
-
Garavello W, Pignataro L, Gaini L, Torretta S, Somigliana E, et al. (2011) Tonsillectomy in children with periodic fever with aphthous stomatitis, pharyngitis, and adenitis syndrome. J Pediatr 159: 138-142.
-
Hofer M, Pillet P, Cochard MM, Berg S, Krol P, et al. (2014) International periodic fever, aphthous stomatitis, pharyngitis, cervical adenitis syndrome cohort: description of distinct phenotypes in 301 patients. Rheumatology (Oxford) 53: 1125-1129.
-
Padeh S, Stoffman N, Berkun Y (2008) Periodic fever accompanied by aphthous stomatitis, pharyngitis and cervical adenitis syndrome (PFAPA syndrome) in adults. Isr Med Assoc J 10: 358-360.
-
Marshall GS, Edwards KM, Lawton AR (1989) PFAPA syndrome. Pediatr Infect Dis J 8: 658-659.
-
Abramson JS, Givner LB, Thompson JN (1989) Possible role of tonsillectomy and adenoidectomy in children with recurrent fever and tonsillopharyngitis. Pediatr Infect Dis J 8: 119-120.
-
Miller LC, Sisson BA, Tucker LB, Schaller JG (1996) Prolonged fevers of unknown origin in children: patterns of presentation and outcome. J Pediatr 129: 419-423.





