Journal of Musculoskeletal Disorders and Treatment
Serum Sclerostin and Muscle Strength in Alcoholics
Emilio González-Reimers1*, Lucía Romero-Acevedo1, Geraldine Quintero-Platt1, Candelaria Martín-González1, Patricia Cabrera-García1, M Angeles Gómez-Rodríguez2, Elisa Espelosín-Ortega3 and Francisco Santolaria-Fernández1
1Servicio de Medicina Interna, Hospital Universitario de Canarias, Universidad de La Laguna, Spain
2Servicio de Medicina Nuclear, Hospital Universitario de Canarias, Universidad de La Laguna, Spain
3Laboratorio Central Hospital Universitario de Canarias. Universidad de La Laguna, Tenerife, Spain
*Corresponding author:
Emilio González-Reimers, Servicio de Medicina Interna, Hospital Universitario de Canarias, Universidad de La Laguna, Tenerife, Canary Islands, Spain, Tel: 0034-9226-78600, E-mail: egonrey@ull.es
J Musculoskelet Disord Treat, JMDT-3-030, (Volume 3, Issue 1), Research Article
Received: July 19, 2016 | Accepted: January 24, 2017 | Published: January 27, 2017
Citation: González-Reimers E, Romero-Acevedo L, Quintero-Platt G, Martín-González C, Cabrera-García P, et al. (2017) Serum Sclerostin and Muscle Strength in Alcoholics. J Musculoskelet Disord Treat 3:030.
Copyright: © 2017 González-Reimers E, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Chronic alcoholic myopathy is a common complication of alcoholism, leading to muscle atrophy and reduced muscle strength. Sclerostin inhibits bone synthesis in situations of reduced load, such as prolonged bed rest or spinal cord injury. The aim pf this study is to analyze the behavior of serum sclerostin in chronic alcoholic myopathy.
Methods: Seventy alcoholic male patients were included, 33 of them cirrhotics, drinkers of about 180 g ethanol daily during more than 30 years. After informed consent patients underwent a whole body densitometric analysis (Lunar Prodigy bone densitometer; General Electric Medical systems, Madison, Wisconsin USA), assessing lean mass and T score at femoral hip and lumbar spine. Dominant handgrip strength was assessed by a Collins dynamometer. Serum sclerostin was measured by one step enzyme-linked immunosorbent assay to the 70 patients and 9 age-matched controls.
Results: Serum sclerostin levels were significantly higher among cirrhotics than non cirrhotics and controls (F = 4.64, p = 0.012), and were related with Pugh score (ρ = 0.36, p = 0.002), prothrombin activity (ρ = -0.24, p = 0.05), albumin (ρ = -0.35, p = 0.003), and the duration of ethanol consumption (ρ = 0.33, p = 0.008). Cirrhotics showed lower lean mass than non cirrhotics, both at right arm (t = 2.44, p = 0.018) and left arm (t = 3.52, p < 0.001), and a trend to a lower handgrip. There was an inverse correlation between handgrip strength and serum sclerostin (ρ = -0.38, p = 0.03), that was independent of liver function and age by multivariate analysis, and a significant association among the lowest left arm lean mass tercile and sclerostin over the median (χ2 = 4.69; p = 0.03). No relation was observed between sclerostin and T-score or Z-score at the hip or lumbar spine.
Conclusion: Sclerostin is raised among alcoholics, keeping a relationship with liver function derangement and with reduced handgrip strength. Handgrip strength is independently related to sclerostin and age.
Keywords
Sclerostin, Alcoholism, Alcoholic myopathy, Liver cirrhosis, Muscle strength, Body lean mass
Introduction
Since several years ago it is well known that osteocytes form a tridimensional net within the bone cortex, able to detect changes in load. Prolongation of osteocytes contact with those of neighbouring cells, so that true "synapsis" becomes established, connecting the osteocytes among each other. An increase in bone load is immediately transformed into a biochemical signal that activates the canonical Wnt- β catenin pathway, leading to an increase in bone mass [1]. Osteocytes also secrete a biochemical mediator that exerts an opposite action, leading to increased bone resorption, namely sclerostin. Load changes on bone are heavily dependent on muscle contraction. In this sense there are several reports underscoring the role of sclerostin in situations of muscle disuse, such as spinal cord injury [2], or situations characterized by muscle unloading, such as prolonged bed rest [3], or, possibly, living in a low gravity environment.
In alcoholic patients muscle atrophy has been described. A direct effect of ethanol seems to play a major role in muscle atrophy in these patients, although undernutrition, vitamin D deficiency, ethanol mediated neuropathy and the peculiar lifestyle of the alcoholics may undoubtedly play significant contributory roles [4]. In parallel with muscle affectation, more or less severe osteoporosis constitutes also a common finding among these patients [5]. As in muscle, a direct effect of ethanol on osteocyte/osteoblast function seems to play a major role [6], together with a probably effect on bone resorption [7].
In a preliminary report we described raised sclerostin levels among 31 alcoholics (11 with coexisting hepatitis C virus (HCV) infection). Raised sclerostin levels were directly related with serum telopeptide and inversely with osteocalcin, fully in accordance with expected role of sclerostin on bone [8]. Other authors have also shown raised sclerostin levels in cirrhotics, especially in Child´s B or C patients [9]. However in these studies the relation between sclerostin and muscle was not analyzed. This relation does exist and has been pointed out in studies in which plasma sclerostin has been measured in individuals participating in a cycling race [10]. Given the dependence of the Wnt- β catenin system on bone load, the bone-forming effect of muscle contraction and the muscle atrophy described in alcoholics, the aim of the present study is to analyze the relation of sclerostin with lean mass in arms, legs and trunk, and handgrip strength in a cohort of 70 alcoholic men. Women were not included due to differences in body composition, muscle strength and muscle mass with respect to men.
Patients and Methods
We included 70 alcoholic men aged 59.01 +/- 11.13 years drinkers of 183 +/- 104 g ethanol daily during 34 +/- 12 years, consecutively admitted to the Internal Medicine Unit of our hospital due to organic problems related with excessive ethanol consumption (withdrawal syndrome, complicated with pneumonia or urinary infection, decompensation of liver cirrhosis). Given the different body composition and muscle strength of men and women, only men were included. The sample comprised 33 cirrhotics and 37 non cirrhotics. Diagnosis of liver cirrhosis was based on liver ultrasonography (heterogeneous liver together with dilated portal vein and splenomegaly), and clinical data, such as ascites, encephalopathy and or variceal bleeding. Some clinical and biological data of these patients are shown in table 1. For cirrhotics, the Child-Pugh score was calculated; in order to globally estimate liver function in the totality of the sample, we also calculated a similar score for non-cirrhotics. Seven patients showed positive antibodies against hepatitis C virus (HCV) infection. Hepatitis B was absent in the patients included. Besides ethanol and tobacco (51 patients), no other drug was used.
![]()
Table 1: Some relevant clinical features of the patients included (33 cirrhotics and 37 non-cirrhotics). In addition to mean ± standard deviation, median value and interquartile range are given for those variables with a non-parametric distribution.
View Table 1
After clinical stabilization in relation with the problem that caused hospital admission, the patients underwent whole body densitometry (Lunar Prodigy bone densitometer; General Electric Medical Systems, Madison, Wisconsin USA), determining lean mass in both arms, trunk and both legs. Bone Z-scores were also recorded at the hips and the lumbar spine. Muscle function was assessed determining dominant arm hand grip strength with a Collins hand dynamometer. Blood was extracted in fasting conditions, and serum was separated and frozen at -80 °C. Serum sclerostin was measured by one step enzyme-linked immunosorbent assay (ELISA, Biomedica gruppe; Wien, Austria, inter-assay variation coefficient 4-6%; intra-assay variation coefficient 5%). Sclerostin was also determined to 13 age-matched male controls, workers at our hospital, who did not drink alcoholic beverages besides 1-2 drinks/week, aged 55.75 ± 9.32 years, i.e., similar to that of our controls (t = 0.96; p = 0.34).
In addition, complete routine biochemical laboratory evaluation was also performed. Child-Pugh's score was calculated for all the patients, including cirrhotics and non cirrhotics (based on prothrombin, albumin, bilirubin, ascitis and encephalopathy).
Statistics
The Kolmogorov- Smirnov test was used to analyze if sclerostin and the remaining variables followed a normal distribution or not. In those cases in which variables did not follow a normal distribution, we used non parametric tests, such as Mann-Whitney's and Kruskal -Wallis tests to assess differences between groups, and Spearman's correlation (ρ). On the other hand, Student's t test, variance analysis with Student-Newman-Keuls post hoc analysis, and Pearson's correlation were also used to analyse parametric variables, and χ2 was used to study the association between two qualitative variables. We performed stepwise logistic regression and/or multiple correlation analyses, in order to disclose which of the analyzed variables were independently related to sclerostin or to handgrip strength. Statistically significance was established if p value was < 0.05 (two-tailed). Statistical analyses were performed using SPSS software (Chicago, IL, USA).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. This study forms part of a larger protocol, destined to assess vascular risk among alcoholics that was approved by the ethical committee of our hospital.
Results
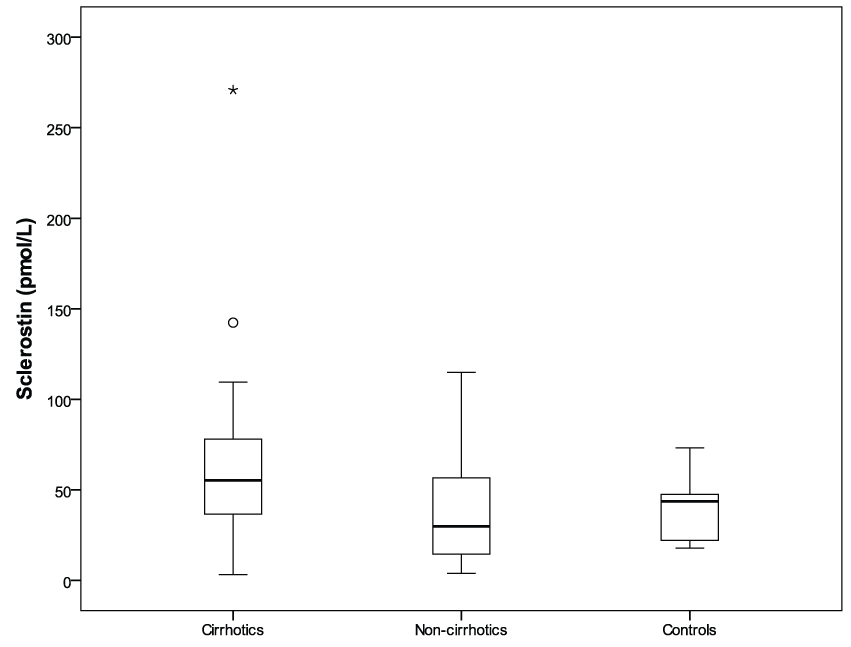
Serum sclerostin levels were significantly different when cirrhotics, non cirrhotics and controls were compared (F = 4.64, p = 0.012; Kruskall-Wallis test = 10.43; p = -0.005; Figure 1). Highest values were observed among cirrhotics (Table 2). Serum sclerostin was directly related with Pugh score, among cirrhotics only (ρ = 0.34, p = 0.05), but also when a similar score was calculated for all the patients, both cirrhotics and non-cirrhotics (ρ = 0.36, p = 0.002), and inverse correlations were observed with prothrombin activity (ρ = -0.24, p = 0.05) and albumin (ρ = -0.35, p = 0.003). Serum sclerostin also showed a direct correlation with the duration of ethanol consumption (ρ = 0.33, p = 0.008), but not with the amount of ethanol consumed daily Serum sclerostin was also directly related with serum creatinine (ρ = 0.29, p = 0.016).
.
Figure 1: Sclerostin levels in cirrhotics, non cirrhotics and controls (Kruskall-Wallis test = 10.43; p = -0.005).
Circles = outliers; asterisks = extreme values.
View Figure 1
![]()
Table 2: Lean and fat mass and T-score at femoral neck and lumbar spine in cirrhotics and non-cirrhotics.
View Table 2
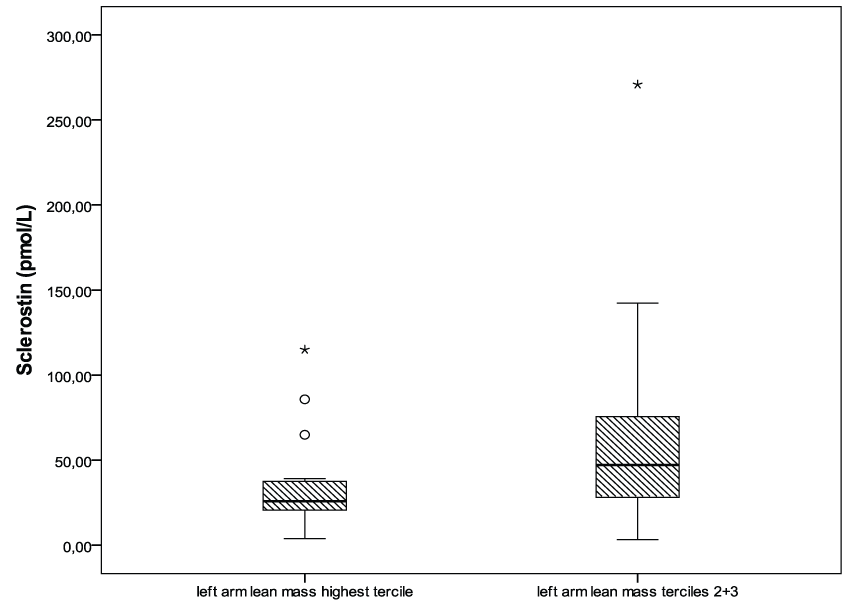
Cirrhotics showed lower lean mass than non cirrhotics, both at right arm (t = 2.44, p = 0.018) and left arm (t = 3.52, p < 0.001). There was also a trend to a lower handgrip among cirrhotic patients that did not reach statistical significance. There was an inverse correlation between serum sclerostin and handgrip strength (ρ = -0.38, p = 0.03). There was a trend to inverse correlations between sclerostin and lean mass, that did not reach statistical signification (the closest one, with left arm lean mass: ρ = -0.25, p = 0.066). However, when we classified left arm lean mass in terciles and compared sclerostin over the median or below the median with lean mass terciles (the highest one versus the second and third terciles) we found an association between the lowest lean mass and the highest sclerostin levels (χ2 = 4.69; p = 0.03; Table 3). In the same sense, sclerostin levels were lower among patients with the higher left arm lean mass tercile (Z = 2.29; p = 0.027, Figure 2). No relation was observed between sclerostin and T-score or Z-score at the hip or lumbar spine.
.
Figure 2: Sclerostin levels and left arm lean mass classified in terciles (Z = 2.29; p = 0.027).
Circles = outliers; asterisks = extreme values.
View Figure 2
![]()
Table 3: Association between highest left arm lean mass and lowest Sclerostin levels an mass (p = 0.03).
View Table 3
In order to disclose whether the relation between handgrip strength and sclerostin was due to a relation of sclerostin with liver cirrhosis we performed a multivariate analysis between the relation of handgrip strength and the following variables (stepwisely introduced): Child-Pugh's score, prothrombin, bilirubin, albumin and sclerostin. We found that the only variable selected was serum sclerostin (beta value = -0.30, p = 0.021) (Table 4A and Table 4B). Handgrip strength maintained its inverse relation with sclerostin also when the variable age was introduced in the multivariate analysis (Table 5A and Table 5B).
![]()
Table 4A and Table 4B: Stepwise multiple regression analysis destined to disclose which variables were independently related to handgrip strength.
View Table 4A and Table 4B
![]()
Table 5A and Table 5B: Stepwise multiple regression analysis destined to disclose which variables were independently related to handgrip strength.
View Table 5A and Table 5B
In order to establish which variables sclerostin levels depend on we classified the patients in those with sclerostin above or below the median, and performed a logistic regression analysis. We observed that prothrombin activity was the first variable selected, followed by years of addiction. The remaining variables included in the analysis (age, albumin, bilirubin, daily ethanol consumption, left arm lean mass, handgrip strength and creatinine) were not selected (Table 6A and table 6B).
![]()
Table 6A and Table 6B: Logistic regression analysis performed in order to assess which variables were independently related to raised sclerostin values (over the median).
View Table 6A and Table 6B
Discussion
In this study we found that serum sclerostin levels were raised in cirrhotics, keeping a relationship with liver function. These findings support a role of liver function on sclerostin levels, and are in accordance with the study of Rhee, et al. [9]. These authors also found an inverse correlation with more severe stages of cirrhosis (Child's B and C groups) among 47 patients, and a direct one with creatinine, as in the present study. As said before sclerostin may be viewed as a marker of bone breakdown in opposition to the bone forming effect of the canonical Wnt β catenin system. It is well known that alcoholism is a risk factor for osteoporosis that increases when liver cirrhosis ensues [6]. It is therefore logical that sclerostin -which raised levels are associated with low bone mass- are also increased in more advanced stages of liver cirrhosis, although in our study no relation was observed with T-score values at hip and lumbar spine, possible because in this group of patients bone affectation was not very intense.
Several studies have shown that a relation between sclerostin and muscle mass also exists, as commented earlier [10]. Although the interpretation may be not "a direct one", changes in sclerostin probably reflect homeostatic adaptation of the bone to the changes in load exerted by the muscle activity, so that in situations of muscle atrophy and/or disuse, sclerostin levels may raise. Chronic alcoholic myopathy is a well known complication of alcoholics, already described several decades ago [11-14]. As shown in this study, decreased handgrip and reduced lean mass are more intense among cirrhotic patients, and these two variables were related with raised sclerostin levels. Since, as commented, other factors such as notably poor nutrition and vitamin D deficiency also play contributory roles on alcoholic myopathy, and are more frequently observed among cirrhotics [15], it is logical that cirrhotics are more intensely affected than non cirrhotic.
A remarkably finding of this study is the fact that handgrip strength was inversely related with sclerostin levels, independently on liver function derangement and on age. It is also worth of note the lack of relation between sclerostin and bone mass both at lumbar spine and femur, suggesting that perhaps the altered behavior of sclerostin is partly independent on bone changes, or, at least, on clinically relevant bone changes.
A shortage of this study is the lack of histological analysis, both of muscle and liver. Muscle biopsy is rarely indicated in alcoholics, and chronic alcoholic myopathy is seldom accompanied by biochemical alterations [16], so reduced muscle mass and/or function remain as the main diagnostic criteria. On the other hand, the finding of the relation of newly described molecules with muscle alteration in these patients, as is the case of sclerostin, may aid in the diagnostic evaluation of this common complication of alcoholism.
Therefore, we conclude that serum sclerostin is raised among alcoholics, keeping a relationship with liver function derangement. Among cirrhotic, lean mass and handgrip strength are reduced, and muscle strength keeps and inverse relationship with sclerostin, that is independent on liver function or age. However, these results deserve confirmation, given the relatively short sample included in this study.
Conflicts of Interest
The authors declare that there are no conflicts of interest. No fundings were received for this research.
References
-
Baron R, Rawadi G (2007) Targeting the Wnt/beta-catenin pathway to regulate bone formation in the adult skeleton. Endocrinology 148: 2635-2643.
-
Battaglino RA, Lazzari AA, Garshick E, Morse LR (2012) Spinal cord injury-induced osteoporosis: pathogenesis and emerging therapies. Curr Osteoporos Rep 10: 278-285.
-
Spatz JM, Wein MN, Gooi JH, Qu Y, Garr JL, et al. (2015) The Wnt inhibitor Sclerostin is up-regulated by mechanical unloading in osteocytes in vitro. J Biol Chem 290: 16744-16758.
-
Preedy VR, Paice A, Mantle D, Dhillon AS, Palmer TN, et al. (2001) Alcoholic myopathy: biochemical mechanisms. Drug Alcohol Depend 63: 199-205.
-
Alvisa-Negrín J, González-Reimers E, Santolaria-Fernández F, García-Valdecasas-Campelo E, Alemán-Valls MR, et al. (2009) Osteopenia in alcoholics: effect of alcohol abstinence. Alcohol Alcohol 44: 468-475.
-
Diamond T, Stiel D, Lunzer M, Wilkinson M, Posen S (1989) Ethanol reduces bone formation and may cause osteoporosis. Am J Med 86: 282-288.
-
Dai J, Lin D, Zhang J, Habib P, Smith P, et al. (2000) Chronic alcohol ingestion induces osteoclastogenesis and bone loss through IL-6 in mice. J Clin Invest 106: 887-895.
-
González-Reimers E, Martín-González C, de la Vega-Prieto MJ, Pelazas-González R, Fernández-Rodríguez C, et al. (2013) Serum sclerostin in alcoholics: a pilot study. Alcohol Alcohol 48: 278-282.
-
Rhee Y, Kim WJ, Han KJ, Lim SK, Kim SH (2014) Effect of liver dysfunction on circulating sclerostin. J Bone Miner Metab 32: 545-549.
-
Grasso D, Corsetti R, Lanteri P, Di Bernardo C, Colombini A, et al. (2015) Bone-muscle unit activity, salivary steroid hormones profile, and physical effort over a 3-week stage race. Scand J Med Sci Sports 25: 70-80.
-
Urbano-Marquez A, Estruch R, Navarro-Lopez F, Grau JM, Mont L, et al. (1989) The effects of alcoholism on skeletal and cardiac muscle. N Engl J Med 320: 409-415.
-
Preedy VR, Peters TJ (1990) Alcohol and skeletal muscle disease. Alcohol Alcohol 25: 177-187.
-
Preedy VR, Salisbury JR, Peters TJ (1994) Alcoholic muscle disease: features and mechanisms. J Pathol 173: 309-315.
-
Romero JC, Santolaria F, González-Reimers E, Dìaz-Flores L, Conde A, et al. (1994) Chronic alcoholic myopathy and nutritional status. Alcohol 11: 549-555.
-
Arteh J, Narra S, Nair S (2010). Prevalence of vitamin D deficiency in chronic liver disease. Dig Dis Sci 55: 2624-2628.
-
Fernandez-Solà J, Preedy VR, Lang CH, Gonzalez-Reimers E, Arno M, et al. (2007) Molecular and cellular events in alcohol-induced muscle disease. Alcohol Clin Exp Res 31: 1953-1962.