Journal of Geriatric Medicine and Gerontology
Diagnosis of Dementia-Circadian Dependency?
Wolfgang Trapp1,2*, Robert Meyrer1, Göran Hajak1 and Stefan Lautenbacher2
1Department of Psychiatry, Psychosomatic Medicine and Psychotherapy, Social Foundation Bamberg, Germany
2Department of Physiological Psychology, Otto-Friedrich University Bamberg, Germany
*Corresponding author: Dr. Wolfgang Trapp, Department of Psychiatry, Psychosomatic Medicine and Psychotherapy, Social Foundation Bamberg, St-.Getreu-Straße 18, 96049 Bamberg, Germany, Tel: +49-951-5032-6123, Fax: +49-951-50321009, E-mail: wolfgang.trapp@sozialstiftung-bamberg.de
J Geriatr Med Gerontol, JGMG-3-022, (Volume 3, Issue 1), Short Communication; ISSN: 2469-5858
Received: July 26, 2016 | Accepted: February 13, 2017 | Published: February 16, 2017
Citation: Trapp W, Meyrer R, Hajak G, Lautenbacher S (2017) Diagnosis of Dementia-Circadian Dependency? J Geriatr Med Gerontol 3:022. 10.23937/2469-5858/1510022
Copyright: © 2017 Trapp W, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Little is known whether tests used for the diagnosis of dementia syndromes are sensitive to time of day effects. In a sample of 145 participants, no such effect (test administration either in the morning from 9.30 to 11.30 a.m. or in the afternoon from 2.30 to 4.30 p.m.) could be found for most subtests of a neurocognitive test battery (CERAD-Plus) used for the detection of dementia syndromes. However, for subjects suffering from mild cognitive impairment, completion time for the Trail-Making Test B, a brief cognitive test for the assessment of cognitive flexibility, was longer in the afternoon compared to the morning.
Keywords
Circadian rhythms, Dementia, Mild cognitive impairment, Neurocognitive performance
Introduction
In healthy ageing persons, from the age of about 60 years, structural and functional changes in the prefrontal cortex and the medial temporal lobe lead to a general cognitive slowing as well as to a decrease of cognitive performance across several cognitive domains [1,2]. This is reflected in the fact that normative data of nearby all cognitive tests usually is stratified by age groups.
The sleep wake cycle is regulated by the interaction of a homeostatic (sleep pressure accumulates during time spent awake) and an oscillating circadian process that is controlled by the suprachiasmatic nucleus (SCN), the central circadian pacemaker located in the anterior hypothalamus which projects to major arousal promoting cell groups like the locus coeruleus. These cell groups mediate alertness, vigilance and attention as well as thalamocortical oscillations which modulate the throughput of sensory information to the cortex (see e.g. [3] for an overview). On the other hand the hippocampus sends input to the SCN which might reflect memory contribution to circadian regulation [4].
During ageing, along with a lower proportion of slow wave sleep, the consolidation of nocturnal sleep decreases, which among others leads to increased daytime napping [5,6]. On the circadian level, a reduced amplitude of circadian rhythmicity [7] as well as an advance of the circadian phase toward morningness [8,9] are found so that the majority of the elderly people are morning types [10].
These age-related changes lead to changes in daytime cognitive performance level. Commonly, cognitive performance worsens in older adults over the day in contrast to younger people, where often an improvement over the day can be found [11]. This so called synchrony effect denoting a beneficial impact on test performance when tests are administered at the preferred time of day (TOD) for diurnal activities could be demonstrated for several cognitive domains including vigilance and reaction time, verbal working memory, episodic memory and measures of executive functioning (EF) [9,12,13].
In Alzheimer's dementia the neural loss exhibited in the SCN [14] during ageing is more pronounced and in advanced dementia, there is an excessive increase of daytime sleepiness as a consequence of a breakdown of the normal sleep/wake-rhythm with wakefulness during the night [15]. However, increased daytime sleepiness compared to non-demented persons can already be found in mild dementia [16,17], activity rhythm fragmentation seems to be correlated with cognitive decline [18] and decreased activity rhythms have even be shown to be an early risk factor for the later development of mild cognitive impairment (MCI) or dementia [19,20].
Considering all this, it is surprising how little attention has been devoted to the question, whether the performance in neurocognitive tests used for the diagnosis of dementia syndromes is sensitive to synchrony effects. To date, TOD effects have been examined only for the Mini Mental State Examination (MMSE) [21], a brief screening tool for dementia syndromes and no effects of TOD on test performance could be found [22,23].
Thus, the aim of this study was to investigate, whether test performance in more complex neuropsychological test batteries used for thorough assessment of cognitive functions in elderly patients is influenced by TOD. As nothing much is known about the sensitivity of neurocognitive test batteries for TOD/synchrony effects, we sought to include participants suffering from a broad range of cognitive achievement reaching from moderate dementia to unimpaired cognitive functioning, which resembles the situation found in everyday clinical practice, when potential dementia has to be diagnosed.
Materials and Methods
Subjects
Between April 2014 and September 2015, 145 (78 female, 67 male, mean age 74.9 years, SD 7.7 years) participants were recruited from the departments of geriatric internal medicine and geriatric psychiatry of a general hospital in Bamberg, Germany, including 121 patients suffering from either mild cognitive impairment (MCI, n = 14) or mild (DMI, n = 81) to moderate (DMO, n = 26) dementia and a control sample (CNT) of 24 subjects recruited from the department of internal medicine's geriatric rehabilitation ward of the same hospital. All subjects were visited for routine dementia screening.
Exclusion criteria were prior or concurrent diagnosis of psychotic or depressive symptoms or a bipolar disorder, alcohol/substance related disorders, significant cardiopulmonary or neurological impairments and uncorrectable deficits in vision or hearing that would interfere with performing the neurocognitive tests.
39 patients could not be included because of past or present marked depressive (n = 20) or psychotic symptoms (n = 4), insufficient vision and hearing (n = 5), alcohol abuse (n = 8), past stroke (n = 2) of the 147 patients, who resulted eligible, 2 refused to participate in the study.
Written informed consent was obtained either from participants themselves or from their legal guardians. The study protocol was approved by the Ethical Review Board of the University of Bamberg.
Testing
The German version of the Consortium to Establish a Rationale in Alzheimer's Diagnostic neuropsychological battery (CERAD-Plus) [24], which includes the Mini Mental State Examination (MMSE), was administered to all participants. In its current version, the CERAD-Plus battery includes-beside the MMSE-10 tasks covering phonemic fluency, semantic fluency, naming (Boston Naming Test), word list learning, delayed free recall and recognition of word list, figure drawing (copying geometric shapes), delayed figure recall as well as the Trail-Making Tests form A (connect figures from 1 to 25 in ascending order) and B (alternately connect figures from 1 to 13 and letters from A to L in ascending order).
To investigate TOD effects on neurocognition, administration time of the tests was recorded. Two groups of patients were formed depending on whether they were tested in the morning from 9.30 to 11.30 a.m. (n = 93) or in the afternoon from 2.30 to 4.30 p.m. (n = 52).
Statistical analysis
T-tests for independent samples were used to compare both groups with respect to age and years of education. To test for differences in the male/female ratio in both TOD groups and for differences in TOD a.m. to p.m. ratio across the four diagnostic groups χ2-tests were performed.
To avoid repetitive statistical testing, multivariate analyses of variance using "diagnostic group" (CNT, MCI, DMI or DMO) as well as TOD (testing in the morning vs. testing in the afternoon) as between subject factors and the groups of cognitive measures displayed in table 1 as dependent measures were conducted first. For those groups of dependent variables yielding significant TOD main effects or TOD x "diagnostic group" interaction effects, analyses of variance using the same design were performed separately for each dependent variable. In case of significant main or interaction effects for single CERAD-Plus subtests, t-tests for independent samples were performed separately for each diagnostic group to compare a.m. vs. p.m. test performance.
![]()
Table 1: Results of multivariate and univariate analyses of variance and a posteriori comparisons.
CNT: control subjects; MCI: mild cognitive impairment; DMI: mild dementia; DMO: moderate dementia.
View Table 1
Statistical significance level was set to 0.05 for all analyses.
Results
No difference was found for both TOD groups with respect to age (mean (std) yrs. for a.m. and p.m. group 74.5 (8.4) and 75.4 (6.2), t(143) = -0.791, p = 0.430), years of education (mean (std) for a.m. and p.m. group 12.0 (1.9) and 12.6 (1.2), t(143) = 1.299, p = 0.196) and male to female ratio (χ2(1) = 1.904, p = 0.168). Furthermore, TOD a.m. to p.m. ratio groups did not differ across the four diagnostic groups (χ2(3) = 1.724, p = 0.632).
Table 1 shows the results of all multivariate and univariate analyses of variance and t-tests. Trivially, group effects for diagnostic group (CNT, MCI, DMI, DMO) in the expected direction of neurocognitive functioning could be found in all analyses of variance.
Significant TOD main effects as well as "TOD x diagnostic group" interaction effects could only be found for the Trail-Making tests. Univariate analyses of variance for each TMT measure yielded significant main effects for TMT B processing time as well as the ratio of TMT B/TMT A processing time but not for TMT A processing time. Additionally the TMT B "TOD x diagnostic group" interaction effect also reached statistical significance.
A posteriori t-tests performed separately for each diagnostic group showed, that the differences in TMT B and TMT B/TMT A are limited to the mild cognitive impairment group: Absolute TMT B completion times (a.m. : mean (std) 218.7 (58.1) and p.m. : 298.4 (41.2)) of MCI patients as well as relative completion times compared to TMT A (a.m. : mean (std) 3.1 (1.2) and p.m. : 5.1 (1.9)) are longer in the afternoon compared to the morning (Figure 1).
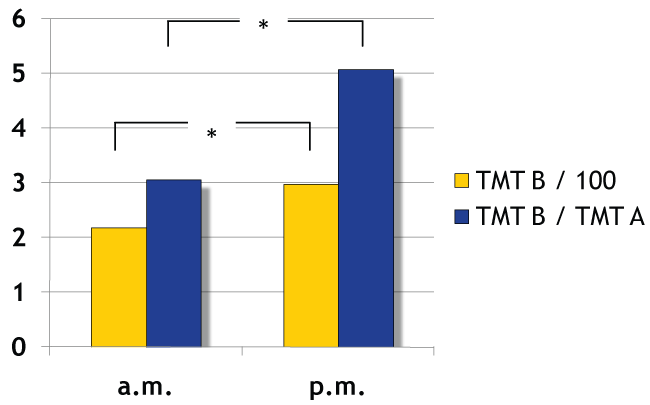
.
Figure 1: TMT B performance (processing time in seconds/100) and TMT A/TMT B processing time ratio of the MCI group (*: = t-tests, p < 0.05).
View Figure 1
Discussion
Robustness of dementia tests against TOD effects
Most of the neurocognitive tests analyzed appeared to be robust against TOD effects in our sample. This means that the results of former studies obtained for the MMSE screening could be replicated for the majority of cognitive tests used for in-depth analysis of dementia syndromes. At a first glance, this result seems surprising, as these tests cover a wider range of cognitive domains and are cognitively more demanding. On the other hand, they still differ from true experimenter-paced tests (occurrence of stimuli is determined by the experimenter) like the Psychomotor Vigilance Test, a simple reaction time task most widely used for the assessment of circadian rhythms in human cognition. Even the speed tests used in our sample were self-paced and did not include assessment of reaction times. Our results, however, imply that tests developed to be sensitive to cognitive deficits typical for beginning dementia syndromes, seem to be robust against TOD effects, which certainly is good news.
Executive functioning, ageing, circadian rhythms and dementia
Nevertheless, for mild cognitive impairment, often considered as an intermediate state between a cognitively unimpaired status and a beginning dementia, preliminary hints could be found that TMT B performance in the afternoon is worse than in the morning. That a synchrony effect was found just for a measure of executive functioning is indeed in accordance with current knowledge about the impact of ageing on neurocognitive functioning and circadian rhythms. In older age, lower blood flow and reduced resting metabolism have been found predominantly in the prefrontal cortex [25], where waking function and delta EEG activity in sleep are linked [26]. Also, cognitive functioning related to the prefrontal cortex has been shown to be particularly vulnerable to sleep loss [3]. Our findings match the results of a study that found poorer performance in executive function tasks in the afternoon compared to the morning only in participants suffering from cognitive impairment and not in cognitively unimpaired subjects of a middle-aged (mean age 61 years) sample [27]. Thus, cognitively unimpaired persons but not MCI patients may be able to compensate for daily fluctuations in cognitive performance in this speed test for cognitive flexibility, which would imply that the "signal to noise ratio" for MCI patients might be high enough to reflect underlying endogenous rhythms in the latter groups' test performance. Moreover, a recent prospective study found that weaker circadian activity rhythms are associated with poorer EF of the participants five years later but not with global cognition, verbal episodic memory or working memory. One measure used in the study was the TMT B and performance in this test was sensitive to these disruptions, even when sleep, cognitive performance level and DMC were controlled statistically [20].
Shortcomings
Our patients were assessed either in the morning or in the late afternoon. Clearly, it would have been preferable to test the same subjects twice at optimal or non optimal times. Unfortunately, no parallel versions exist for the standard neurocognitive test battery used in this study. Nevertheless, this study should be replicated using repeatable cognitive assessments.
Despite the fact, that the vast majority of elderly persons are "morning types", it cannot be ruled out, that subjects with deviating individual chronotypes were included. Future studies should therefore assess the chronotype of each participant.
In former studies, TOD fluctuations were found not only to be linked with the flexibility subdomain of EF covered by the TMT B, but also with EF subdomains like working memory, set shifting and inhibition [28]. Therefore, it would be interesting to include a broader array of EF tests in future studies investigating possible synchrony effects in test batteries for the diagnosis of dementia syndromes.
As our subgroup of MCI patients was very small, the discovery of a synchrony effect for the TMT B in this subgroup could be a random finding. However, in case of replication using larger samples, parallel versions of such brief EF tests could be administered at optimal and non optimal times of day to see if marked synchrony effects can be observed and whether such fluctuations in TMT performance might be an early predictor of cognitive decline. In this case, an extended test battery for the screening of early signs of dementia could be administered and even in absence of such signs, subjects could be reassessed on a regular basis.
Acknowledgement/Disclosure
The authors would like to thank Bruno Weisenberger, Michaela Wachter and Maren Lindsiepe for administration of the tests, Karin Divers for proofreading of the manuscript and all patients and control subjects for their participation.
References
-
Harrison Y, Horne JA, Rothwell A (2000) Prefrontal neuropsychological effects of sleep deprivation in young adults-a model for healthy aging? Sleep 23: 1067-1073.
-
Hedden T, Gabrieli JD (2004) Insights into the ageing mind: a view from cognitive neuroscience. Nat Rev Neurosci 5: 87-96.
-
Blatter K, Cajochen C (2007) Circadian rhythms in cognitive performance: methodological constraints, protocols, theoretical underpinnings. Physiol Behav 28: 196-208.
-
Stranahan AM (2012) Chronobiological approaches to Alzheimer's disease. Curr Alzheimer Res 9: 93-98.
-
Munch M, Knoblauch V, Blatter K, Schroder C, Schnitzler C, et al. (2005) Age-related attenuation of the evening circadian arousal signal in humans. Neurobiol Aging 26: 1307-1319.
-
Dijk DJ, Duffy JF, Czeisler CA (2001) Age-related increase in awakenings: impaired consolidation of Non REM sleep at all circadian phases. Sleep 24: 565-577.
-
Dijk DJ, Duffy JF (1999) Circadian regulation of human sleep and age-related changes in its timing, consolidation and EEG characteristics. Ann Med 31: 130-140.
-
Brown SA, Schmitt K, Eckert A (2011) Aging and circadian disruption: causes and effects. Aging 3: 813-817.
-
May CP (1999) Synchrony effects in cognition: The costs and a benefit. Psychon Bull Rev 6: 142-147.
-
Yoon C, May CP, Hasher L (1999) Aging, circadian arousal patterns, and cognition. In: Schwarz NPD, Knauper B, Sudman S. Aging, cognition and self reports. Washington, DC: Psychological Press.
-
Schmidt C, Collette F, Cajochen C, Peigneux P (2007) A time to think: circadian rhythms in human cognition. Cogn Neuropsychol 24: 755-789.
-
Yang L, Hasher L, Wilson DE (2007) Synchrony effects in automatic and controlled retrieval. Psychon Bull Rev 14: 51-56.
-
Hasher L, Zacks RT, Rahhal TA (1999) Timing, instructions, and inhibitory control: some missing factors in the age and memory debate. Gerontology 45: 355-357.
-
Tang B (2007) Living by a biological clock: age-related functional changes of sleep homeostasis in people aged 65-88.5 years. Sleep and Biological Rhythms 5: 180-195.
-
Montplaisir J, Petit D, Gauthier S, Gaudreau H, Decary A (1998) Sleep disturbances and EEG slowing in Alzheimer’s disease. Sleep Res Online 1: 147-151.
-
McClung CA (2011) Circadian rhythms and mood regulation: Insights from pre-clinical models. Eur Neuropsychopharmacol 21: S683-S693.
-
Bonanni E, Maestri M, Tognoni G, Fabbrini M, Nucciarone B, et al. (2005) Daytime sleepiness in mild and moderate Alzheimer's disease and its relationship with cognitive impairment. J Sleep Res 14: 311-317.
-
Oosterman JM, van Someren EJ, Vogels RL, Van Harten B, Scherder EJ (2009) Fragmentation of the rest-activity rhythm correlates with age-related cognitive deficits. J Sleep Res 18: 129-135.
-
Tranah GJ, Blackwell T, Stone KL, Ancoli-Israel S, Paudel ML, et al. (2011) Circadian activity rhythms and risk of incident dementia and mild cognitive impairment in older women. Ann Neurol 70: 722-732.
-
Walsh CM, Blackwell T, Tranah GJ, Stone KL, Ancoli-Israel S, et al. (2014) Weaker circadian activity rhythms are associated with poorer executive function in older women. Sleep 37: 2009-2016.
-
Folstein MF, Folstein SE, McHugh PR (1975) ''Mini-mental state''. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12: 189-198.
-
Brown MA, Newson M, Haworth J, Wilcock GK (2005) Exploring time of day effects on mini-mental state examination performance in Alzheimer’s disease and age-associated cognitive decline. Brain Impairment 6: 212-218.
-
Madhusoodanan S, Madhusoodanan N, Serper M, Sullivan SJ, D'Antonio E, et al. (2010) Cognitive status changes based on time of day in nursing home patients with and without dementia. Am J Alzheimers Dis Other Demen 25: 498-504.
-
Aebi C (2002) Validierung der Neuropsychologischen Testbatterie CERAD-NP: Eine Multi-Center Studie, Thesis, University of Basel.
-
Meltzer CC, Francis PT (2001) Brain aging research at the close of the 20th century: from bench to bedside. Dialogues Clin Neurosci 3: 167-180.
-
Horne JA (1993) Human sleep, sleep loss and behavior. Implications for the prefrontal cortex and psychiatric disorder. Br J Psychiatry 162: 413-419.
-
Paradee CV, Rapport LJ, Hanks RA, Levy JA (2005) Circadian preference and cognitive functioning among rehabilitation inpatients. Clin Neuropsychol 19: 55-72.
-
West R, Murphy KJ, Armilio ML, Craik FI, Stuss DT (2002) Effects of time of day on age differences in working memory. J Gerontol B Psychol Sci Soc Sci 57: 3-10.





