Journal of Clinical Nephrology and Renal Care
Outcomes Associated with a Heparin-Free Hemodialysis Protocol and Review of the Literature
Elizabeth Liang1,2, Mayra Rodriguez1,2, Melissa Mueller2, Matthew K Abramowitz1,2 and Michele H Mokrzycki1,2*
1Division of Nephrology, Albert Einstein College of Medicine and Montefiore Medical Center, New York, USA
2Department of Medicine, Albert Einstein College of Medicine and Montefiore Medical Center, New York, USA
*Corresponding author:
Michele H Mokrzycki, MD, MS, Division of Nephrology, Department of Medicine, Albert Einstein College of Medicine and Montefiore Medical Center, 3411 Wayne Ave, Suite 5-H, Bronx, NY 10467, USA, Tel: 718-920-5442, Fax: 718-652-8384, E-mail: mmokrzyc@montefiore.org
J Clin Nephrol Ren Care, JCNRC-2-010, (Volume 2, Issue 1), Review Article
Received: April 18, 2016: Accepted: May 18, 2016: Published: May 20, 2016
Citation: Liang E, Rodriguez M, Mueller M, Abramowitz MK, Mokrzycki MH (2016) Outcomes Associated with a Heparin-Free Hemodialysis Protocol and Review of the Literature. J Clin Nephrol Ren Care 2:010.
Copyright: © 2016 Liang E, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:We aimed to determine outcomes associated with a heparin-free hemodialysis (H-HD) protocol in hospitalized patients.
Methods:A retrospective study comparing 2 intra-dialytic heparin protocols: H + HD (routine heparin prime/bolus dosing) and H-HD (saline prime, heparin avoidance). HD circuit patency, need for intra-dialytic interventions and patient bleeding events were the outcomes.
Results:We extracted data from 1043 HD procedures performed in 365 patients. H-HD was associated with a higher rate of HD circuit clotting (9.1% vs. 2.4%, p = 0.019), most requiring a change in the HD circuit (7.3% vs. 0.8%). In a multivariate analysis, the significant predictors of HD circuit clotting were H-HD, OR = 5.11, blood flow rate, OR = 0.61, and platelet count, OR = 1.27. There was no reduction in bleeding events in the H-HD group.
Conclusions:A H-HD protocol was associated with excess HD circuit clotting resulting in more treatment interruptions, nursing interventions, blood loss, and total costs without a proven safety benefit.
Introduction
Intra-dialytic heparin is routinely used for anticoagulation of the extracorporeal hemodialysis (HD) circuit [1,2]. The theoretical benefit of intra-dialytic heparin is to prevent HD circuit clotting, thereby reducing treatment interruption and blood loss, and optimizing HD adequacy. However, in an inpatient population with heightened acuity of illness, the potential for adverse events attributable to heparin, such as hemorrhage and heparin-induced thrombocytopenia, is concerning [2,3]. Therefore, a heparin-free HD (H-HD) protocol use in all hospitalized HD patients may be a desirable alternative to the practice of “routine heparin-use” during HD, (H + HD).
Many previously published series of H-HD lacked a H + HD comparison group, and most were limited to unstable patients with relative contraindications to anticoagulation [4-10]. Although heparin was not administered as a bolus or maintenance dose during the HD procedure, in most studies a heparinized saline solution was used to prime the dialyzers pre-HD [4-6,8-10]. These studies reported that H-HD was feasible in HD patients at risk for bleeding and was associated with acceptable HD circuit clotting rates. Controlled trials comparing H-HD and H + HD reported similar outcomes or a trend toward greater HD circuit clotting in the H-HD treatments, but were not powered to detect a significant difference [11-16]. Furthermore, many studies utilized a prophylactic saline flush or infusion protocol, or prophylactic change of the dialyzer to reduce risk of HD circuit clotting [4-9,13,14].
In 2007, due to a heparin recall by the Food and Drug Administration, a H-HD protocol was implemented in our inpatient HD unit for all hospitalized HD patients, including both acute and chronic HD patients, independent of patient bleeding risk [6]. These procedures were completely heparin-free, and saline alone was used to prime the HD circuit. This H-HD protocol has been continued to the present day, despite the present availability of heparin. To compare outcomes associated with H-HD to those of H + HD, we conducted a retrospective chart review.
Material and Methods
This study was approved by the Institutional Review Board of Montefiore Medical Center. (IRB # 10-05-151E)
Prior to 2007, intra-dialytic heparin dosing was prescribed at the discretion of the treating nephrologist (H + HD period). During this period prophylactic intra-dialytic heparin was ordered at the discretion of the treating nephrologist and given as a bolus dose (range 500-2000 units) in the first 15 minutes of initiating HD. A maintenance hourly heparin dose was not commonly prescribed.
In 2007, due to a heparin recall by the Food and Drug Administration, a heparin-free hemodialysis (H-HD) protocol was implemented [6]. During this period, prophylactic intra-dialytic heparin was not available or prescribed as either a bolus or maintenance dose.
Inclusion and exclusion criteria: Data from all inpatient HD treatment flow charts and hospital charts from June 1 - July 31, 2006 during a period of routine heparin-use (H + HD), and from January 1 - January 31, 2010 during a heparin recall/shortage (H-HD) were included. We excluded data from HD treatments performed in the critical care units due to the higher acuity of patients. We excluded patients receiving continuous renal replacement therapy. We also excluded the first and second HD treatments in a new ESRD patient, as they were intentionally prescribed with lower blood flow rates and treatment times, however subsequent HD treatments in these patients were included in the analysis.
All HD procedures were performed at the Montefiore Medical Center inpatient HD unit. All treatments were performed using Fresenius 2008K machines (Fresenius Medical Care North America, Waltham, MA) with Optiflux® F 180 NR single use, hollow fiber dialyzers which were primed with normal saline alone before starting HD. HD treatment times ranged between 3 to 4 hours. Hemodiafiltration was not used during any of the study periods. Rescue heparin was defined as heparin given after the first 15 minutes of starting dialysis in response to poor blood flow or increased pressure in venous line. Saline bolus given in response to hypotension was distinguished from saline flush given for anticipated clotting and not included under intervention. We did not implement a prophylactic saline flush or infusion protocol.
The following data were collected: patient demographic information including age, sex, race, diabetes status, admission diagnosis, use of systemic anticoagulation (warfarin or intravenous therapeutic heparin), use of heparin for prophylaxis, use of heparin catheter lock. Baseline laboratory values recorded were hemoglobin, platelet count, partial thromboplastin time (PTT), and international neutralization rate (INR). Blood flow rate, venous pressures, and arterial pressures (average of 3 recordings) for the first hour of dialysis were recorded.
The primary outcome analyzed was the rate and extent of extracorporeal HD circuit clotting, which was recorded by the dialysis nurse on the treatment flow sheet by indicating that dialysis lines had to be changed or the treatment was terminated early. Secondary outcomes include: 1) the rinse condition of the dialyzer upon treatment termination (i.e., clean, streaky, clotted), and 2) an intra-dialytic therapeutic intervention, including saline flush or rescue heparin used for poor blood flow rate. Documented hemorrhagic events such as gastrointestinal (GI) bleeding, HD access bleeding, and surgical wound hemorrhage during the hospitalization were recorded.
Data are reported as the mean +/- standard deviation. Continuous variables were evaluated using independent Student's t-test and categorical variables were analyzed using Chi-square analysis. A general estimated equation (GEE) was performed using the following predetermined variables: intra-dialytic heparin use, blood flow rate, diabetes mellitus, hemoglobin, platelet count, the use of systemic anticoagulation, type of dialysis access, GI bleeding and non-GI bleeding during hospitalization. A GEE was used to control for repeated measures within the same patient. Statistical significance was set at P < 0.05. All data were performed on SPSS® 10.0 and STATA® 12.
Results
Data from 1043 inpatient HD procedures performed in 365 hospitalized patients were reviewed: 911 H-HD treatments (298 patients), and 132 H + HD (67 patients). (Table 1) During the H-HD period, there was a higher percentage of patients who were prescribed anticoagulation on the wards, (including the therapeutic use of systemic anticoagulation (warfarin, intravenous heparin, and dalteparin) and prophylactic subcutaneous heparin use to prevent deep venous thrombosis), and the mean PTT and INR values were higher in these patients.
![]()
Table 1: Patient characteristics, laboratory data and dialysis characteristics for each HD session.
View Table 1
In the H + HD treatments the recorded pressures in the arterial and venous chambers were significantly higher than in the H-HD procedures, and there were more women represented in the H + HD period. All other variables were similar between the patients in the H + HD and H-HD periods, including hemoglobin, platelet count, the incidence of red blood cell transfusions, and the mean dialysis blood flow rates.
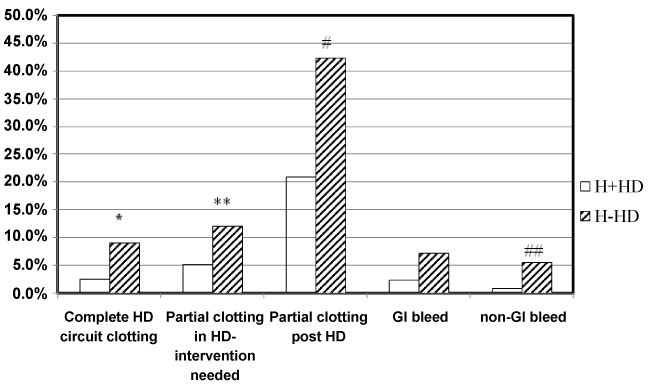
The primary and secondary outcomes are provided in table 2 and figure 1. There was a significant increase in HD circuit clotting in the H-HD period (9.1%) compared to the H + HD period (2.4%) (P = 0.008). In the H-HD period, HD circuit clotting resulted in a change of the extracorporeal circuit in 7.3% of sessions, whereas this was required in only 0.8% of H + HD sessions. Early termination of HD treatments was similar between the periods. Dialyzer fiber clotting post-HD was more commonly reported in the H-HD period (42.0 vs. 20.9%), (P < 0.001). Furthermore, nursing interventions (saline flush or heparin bolus) were required more frequently in the H-HD sessions (12 vs. 5.1%), (P = 0.025).
There was no statistically significant difference in gastrointestinal (GI) bleeding events during hospitalization between the heparin periods, however the risk of non-GI bleeding was significantly higher in the H-HD period in a univariate analysis, (H-HD 5.6% vs. H + HD 0.8%, P = 0.017) (Table 2).
![]()
Table 2: Comparison of outcomes for H + HD and H-HD treatments.
View Table 2
In a multivariate analysis, after adjusting for other significant covariates, H-HD was associated with a 5-fold risk of extracorporeal HD circuit clotting (Table 3). There was no significant difference in GI or non-GI bleeding between the two periods in the multivariate analysis. Other variables associated with a higher risk of HD circuit clotting were elevated platelet count and hemoglobin. The use of systemic anticoagulation for non-dialysis indications and higher dialyzer blood flow rates were associated with lower HD circuit clotting risks. Diabetics had a lower HD circuit clotting risk.
Discussion
In this study, H-HD was associated with a significant risk of extracorporeal HD circuit clotting resulting in the need for nursing intervention during the HD procedure, such as a saline flush, or change of the complete HD circuit. Complete HD circuit clotting requiring change of HD circuit or early treatment termination occurred in 9.1% of H-HD procedures. Partial clotting requiring an intervention during HD (saline flush) occurred in 12%, and partial fiber clotting assessed as the post HD dialyzer appearance occurred in 42.3% of H-HD dialysis procedures (Table 2 and Figure 1). Factors associated with a higher risk of HD circuit clotting were low blood flow rate, elevated hemoglobin and platelet count. Heparin avoidance in HD was not associated with a reduction in reported bleeding events, in fact there was a higher incidence of non-GI bleeding in the H-HD group which may be attributable to the use of anticoagulation unrelated to the HD procedure. In a multivariate analysis, when controlling for blood flow rate, platelet count, hemoglobin, diabetes mellitus, systemic anticoagulation, and clinical bleeding events, H-HD was associated with a 5-fold risk of HD circuit clotting (Table 3).
.
Figure 1: Comparison of clinical outcomes in H + HD and H-HD protocols.
HD: Hemodialysis, H+HD: Routine heparin hemodialysis, H-HD: Heparin-free hemodialysis, GI: Gastrointestinal.
*p = 0.008, **p = 0.025, #p < 0.001, ##p = 0.017
View Figure 1
![]()
Table 3: Multivariate Analysis for Primary Outcome Filter Clotting.
View Table 3
Comparisons between previous studies in H-HD are difficult due to differences associated with the HD procedure, including the dialyzer type, vascular access type, prescribed blood flow rates and treatment times. There were also differences in the priming solution type, prescribed use of prophylactic saline flush/infusion protocols, or prophylactic change of the dialyzer in these studies. Most importantly, outcome definitions vary between studies.
Parallel plate or coil dialyzers were used in some of the earliest trials, as were dialyzers with smaller surface areas and non-synthetic, cellulosic-based dialyzers [4,5,8,9-11,15]. In the only study comparing dialyzer membrane materials, using a retrospective trial design Barth et al. reported a lower clotting incidence using a polyacrylonitrile membrane than found with regenerated cellulose or cellulose acetate dialyzers [14]. Catheters were also associated with a higher incidence of clotting episodes, compared to arteriovenous fistulae or grafts, in this study [14,16]. Raja et al. reported that lower prescribed blood flow rates (< 250 ml/min), longer dialysis treatment times (> 3.5 hours), and blood transfusion administration during HD were shown to be an important risk factors for post-HD partial fiber clotting (> 20% fiber bundle volume). This was a relatively small study in 70 dialysis procedures [4]. In contrast, in a large retrospective study in 17,000 H-HD treatments, Stamatiadis et al. reported, in a multivariate analysis, that only blood flow significantly affected HD treatment failure, which was defined as HD requiring treatment termination or change of the dialyzer [9].
The rate of HD circuit clotting reported by Glaser et al. in H-HD (9.6%) is similar to that reported in our study, however in the former study this was not significantly different from the clotting rate in the H + HD group (6.9%) [11]. The use of prophylactic saline flushes or continuous saline infusion protocols have been implemented in anticipation of extracorporeal clotting in H-HD, although the benefit is uncertain. The rate of HD dialyzer clotting reported using a saline flush H-HD protocol in trials in which the priming solution was saline was between 5-10% [12,16]. Preuschof et al. reported outcomes in H-HD procedures using either saline bolus, continuous saline infusion, or both. Saline was used as the priming solution in all treatments, however no comparisons were made between the groups [7]. In addition, prophylactic change of the HD circuit was performed when impending clotting of the dialyzer was noted. Complete and partial HD circuit clotting occurred in 3% and 5% of treatments, however 52% of treatments required prophylactic change of the dialyzer. Caruna et al. compared dialyzer clotting, defined as post-HD % fiber loss, in 4 groups: H-HD without prophylactic saline, H-HD with routine saline flush, H-HD with constant saline infusion, and H + HD [13]. All dialyzers were primed with low dose heparin and saline. Prophylactic saline flushes or continuous saline infusion was not associated with a reduction in fiber loss or dialyzer clotting.
In clinical trials of H-HD in which a heparinized priming solution was used pre-HD, but without a prophylactic saline flush or infusion protocol, the incidence of partial and complete circuit clotting, defined as necessitating change of HD circuit, occurred in 5.8-20% and 2-7% of treatments [6,8,13]. Stamatiadis reported similar outcomes in a large series of H-HD in which both a protocol of routine saline flushes and the use of a heparinized priming solution was associated with a 5% incidence of complete HD circuit clotting, defined as clotting requiring change of circuit or early termination of HD [9].
Studies in which comparisons were made between H + HD and H-HD procedures are few, and outcome definitions are not uniform. Romao et al. compared H-HD and H + HD in 10 patients who served as their own controls, and reported more dialyzer 125I-fibrin deposition upon completion of dialysis in the H-HD treatments. In a comparison of 200 total HD treatments, McGill et al. reported dialyzer clotting (details not provided) in 5% H-HD and 0% in H + HD [16]. Barth et al. reported a higher incidence of HD circuit clotting in H-HD (1.3-26%) compared to H + HD (0.5-11%), which was defined as the need to change HD circuit or early treatment termination, however this outcome varied markedly depending upon the dialyzer membrane characteristics (as described earlier), and a statistical comparison of outcomes between H-HD and H + HD was not performed [14].
Several studies that assessed the effect of H-HD on HD efficiency have not observed a significant loss of solute clearances or ultrafiltration rate between H-HD and H + HD [5,6,8,12,15,16].Studies evaluating coagulation and platelet function report no significant differences between H-HD and H + HD with respect to platelet count, platelet aggregation testing, fibrinogen, fibrin degradation products, antithrombin III level, prothrombin time and partial thromboplastin time [6,10,12] (Table 4).
![]()
Table 4: Studies evaluating extracorporeal circuit clotting in heparin-free hemodialysis.
View Table 4
A major limitation of the present study is the retrospective study design in which existing data was extracted from chart reviews. The dialysis prescription was determined at the discretion of the nephrologist and HD adequacy was not determined, as this was an inpatient HD population. The available data about the filter clotting was based on a subjective account documented by the HD staff and not based on a formal scoring system. The decisions on the need to change the HD filter or on the need administer intradialytic saline flushes were made on clinical grounds by the HD staff, and were not based on a formal protocol. In addition, the control group was selected from a historical period during which routine heparin anticoagulation during HD was available. Lastly, the number of patients and HD treatments between the treatment groups is unequal. Future large, prospective, randomized controlled trials are needed to control for all of the variables described above. It should be noted however, that one of the strengths of the study is that the nephrology attending staff and the HD nursing staff members, HD equipment and all other inpatient HD practices remained constant between the different time periods and heparin groups.
The clinical benefits of heparin avoidance in HD, specifically reduced bleeding incidence, are difficult to prove in high bleeding risk patients, as there are no randomized control trials for this patient population. In the current study, there was no difference in clinical bleeding during hospitalization in the H-HD and H + HD periods. Shen et al. observed similar findings in heparin-free outpatient dialysis [17]. Using the United States Renal Data System and the electronic medical records, and Medicare claims, they identified 836 incident HD patients aged 67+, not on warfarin, who were receiving H-HD. In a multivariate analysis, they found no significant association of H-HD with all-cause mortality, bleeding, or arterial/venous thrombosis.
The need for heparin avoidance in HD for high bleeding risk patients is intuitive however complete heparin avoidance as a standard protocol in an inpatient HD unit for all patients (both low and high bleeding risk) may be desirable. The potentitial advantages of routine heparin avoidance in an inpatient HD setting include elimination of unintentional heparin prescribing errors, reduction in heparin-induced thrombocytopenia (HIT), and the staff's ease in adherence with uniform protocols. In the current study, heparin avoidance in HD was involuntary, as there was a shortage of heparin due to FDA recall. To date, a H-HD protocol remains in active use in our inpatient HD unit for all hospitalized patients, however the potential loss of blood due to circuit clotting, need for increased nursing interventions for saline flushes and/or change of the HD circuit, and additional costs incurred do not appear to be justified by a documented benefit.
In addition to unfractionated heparin, anticoagulant options for intermittent HD include low-molecular weight heparins (Enoxaparin, Tinzaparin, Bemiparin), direct thrombin inhibitors (argatroban, hirudin, bivalirudin), and regional citrate anticoagulation [2,18]. Citrate anticoagulation is not commonly used for outpatient HD due to its relative complexity, namely the need for specialized dialysate containing low calcium, low magnesium and low bicarbonate. Furthermore, citrate intoxication may occur in patients with hepatic failure, rhabdomyolysis and when isolated ultrafiltration is used due to reduced citrate metabolism. Heparinoids, such as danaparoid and fondaparinux have prolonged half-lives in HD and dose adjustment and monitor are advised based on the anti-Xa activity pre-HD [2,17]. Dabigatran, apixaban and rivaroxaban are associated with increased bleeding risk in the ESRD population, and their use is not recommended [19,20].
Acknowledgements
Dr. Maria Coco and Dr. Joel Neugarten for reviewing this manuscript.
References
-
Fischer KG (2007) Essentials of anticoagulation in hemodialysis. Hemodial Int 11: 178-189.
-
Davenport A (2011) What are the anticoagulation options for intermittent hemodialysis? Nat Rev Nephrol 7: 499-508.
-
Sonawane S, Kasbekar N, Berns JS (2006) The safety of heparins in end-stage renal disease. Semin Dial 19: 305-310.
-
Raja R, Kramer M, Rosenbaum JL, Bolisay C, Krug M (1980) Hemodialysis without heparin infusion using Cordis Dow 3500 hollow fiber. Proc Clin Dial Transplant Forum 10: 39-42.
-
Agresti J, Conroy JD, Olshan A, Conroy JF, Schwartz A, et al. (1985) Heparin-free hemodialysis with cuprophan hollow fiber dialyzers by a frequent saline flush, high blood flow technique. Trans Am Soc Artif Intern Organs 31: 590.
-
Sanders PW, Taylor H, Curtis JJ (1985) Hemodialysis without anticoagulation. Am J Kidney Dis 5: 32-35.
-
Preuschof L, Keller F, Seemann J, Offermann G (1988) Heparin-free hemodialysis with prophylactic change of dialyser and blood lines. Int J Artif Organs 11: 255-258.
-
Schwab SJ, Onorato JJ, Sharar LR, Dennis PA (1987) Hemodialysis without anticoagulation. One-year prospective trial in hospitalized patients at risk for bleeding. Am J Med 83: 405-410.
-
Stamatiadis DN, Helioti H, Mansour M, Pappas M, Bokos JG, et al. (2004) Hemodialysis for patients bleeding or at risk for bleeding, can be simple, safe and efficient. Clin Nephrol 62: 29-34.
-
Ivanovich P, Xu CG, Kwaan HC, Hathiwala S (1983) Studies of coagulation and platelet functions in heparin-free hemodialysis. Nephron 33: 116-120.
-
Glaser P, Guesde R, Rouby JJ, Eurin B (1979) Haemodialysis without heparin is possible. Lancet 2: 579-580.
-
Casati S, Moia M, Graziani G, Cantaluppi A, Citterio A, et al. (1984) Hemodialysis without anticoagulants: efficiency and hemostatic aspects. Clin Nephrol 21: 102-105.
-
Caruana RJ, Raja RM, Bush JV, Kramer MS, Goldstein SJ (1987) Heparin free dialysis: comparative data and results in high risk patients. Kidney Int 31: 1351-1355.
-
Barth RH, Sodden S, Berlyne GM (1989) Heparin-free hemodialysis with a polyacrylonitrile membrane. ASAIO Trans 35: 597-600.
-
Romao JE Jr, Fadil MA, Sabbaga E, Marcondes M (1997) Haemodialysis without anticoagulant: haemostasis parameters, fibrinogen kinetic, and dialysis efficiency. Nephrol Dial Transplant 12: 106-110.
-
McGill RL, Blas A, Bialkin S, Sandroni SE, Marcus RJ (2005) Clinical consequences of heparin-free hemodialysis. Hemodial Int 9: 393-398.
-
Shen JI, Mitani AA, Chang TI, Winkelmayer WC (2013) Use and safety of heparin-free maintenance hemodialysis in the USA. Nephrol Dial Transplant 28: 1589-1602.
-
Ciccone MM, Cortese F, Corbo F, Corrales NE, Al-Momen AK, et al. (2014) Bemiparin, an effective and safe low molecular weight heparin: a review. Vascul Pharmacol 62: 32-37.
-
Chan KE, Edelman ER, Wenger JB, Thadhani RI, Maddux FW (2015) Dabigatran and rivaroxaban use in atrial fibrillation patients on hemodialysis. Circulation 131: 972-979.
-
Heibuchel H, Verhamme P, Alings M, Antz M, Diener HC, et al. (2015) Updated European heart rhythm association practical guide on the use of non-vitamin K antagonist anticoagulants in patients with non-valvular atrial fibrillation. Europace 17: 1467-1507.