Eighteen-Months May be a Reliable Storage Period for Direct Immunofluorescent Slides of Renal Biopsies at Room Temperatur
Tripti Kumar1, Thomas Fennell2 and Ping L. Zhang1*
1Division of Anatomic Pathology, Beaumont Laboratory, Beaumont Health, USA
2Division of Clinic Pathology, Beaumont Laboratory, Beaumont Health, USA
*Corresponding author:
Ping L. Zhang, MD, PhD, Division of Anatomic Pathology, Beaumont Laboratory, Beaumont Health, 3601 W. 13 Mile Road, Royal Oak, Michigan 48073-6769, USA, Tel: 248-898-9060, Fax: 248-898-8020, E-mail: Ping.zhang@beaumont.edu
J Clin Nephrol Ren Care,
JCNRC-1-001, (Volume 1, Issue 1),
Original Article
Received: July 23, 2015: Accepted: August 13, 2015: Published: August 17, 2015
Citation: Kumar T, Fennell T, Zhang PL (2015) Eighteen-Months May be a Reliable Storage Period for Direct Immunofluorescent Slides of Renal Biopsies at Room
Temperature. J Clin Nephrol Ren Care 1:001
Copyright: © 2015 Kumar T. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Context: After the initial diagnosis of a renal biopsy, direct immunofluorescent (DIF) slides would be ideally kept for later internal and external reviews. Although there is an assumption that rapid reduction in fluorescent intensity occurs after DIF staining, no renal DIF study has addressed how long DIF slides can be reliably stored for re-examination at room temperature.
Objective: In this study, we determined whether DIF staining intensity was reduced in renal biopsies over time, thus establishing a standard period for the storage.
Design: We retrospectively evaluated 21 control cases of focal segmental glomerulosclerosis (FSGS) and 35 study cases with immune complex mediated glomerulopathy (ICMG). The DIF slides were stored at lab room temperatures (set at 70℉ - 21 ± 1℃). DIF intensity for IgG, kappa and lambda was re-examined during two follow-up periods (1 to 17.5 months [m], and 41 to 57.5m, respectively). The re-examined scores were compared with initial intensity scores (0 to 3+) statistically.
Results: All control cases (FSGS) essentially showed no staining in both initial and re-examinations. In the study group, there was a small but non-significant reduction in staining intensity for IgG, kappa and lambda during the first follow-up. At the second follow-up examination 40m later, 50 to 70% reduction of staining intensity was noted for all study cases.
Conclusions: Our data suggest that DIF intensity was relatively well maintained from 1 to 17.5m in most cases. However, the staining intensity had significantly faded from 41 to 57.5m. Thus a reliable storage period for renal DIF slides appears to be 18m.
Introduction
DIF stains in renal biopsies has been introduced as early as 1960's [1,2] and a panel of DIF stains have fully developed as gold standard in the renal pathology field from 1970's to current practice [3-10]. Since the fluorescent power of dyes for staining are thought to have a fast decay once the staining is activated, most pathologic laboratories usually eliminate (within two weeks to one month) the DIF slides after diagnosis of renal biopsies has been reached. However, reviewing renal DIF slides can be important for re-evaluating a previously rendered diagnosis, either for an internal review or an outside consultation. Thus a reliable storage period for keeping renal IF slides, should be well established.
Storage periods for DIF stains related to skin diagnosis has been found adequate for only 2 to 9 months from 1975 to 1985 [11-13], and is found to last longer, up to 11 months, in a 2003 study [14]. However, according to our knowledge, the preservation period of DIF intensity in renal biopsies has not been reported. Several studies comparing IF staining in fresh tissue and other fixative tissue have been documented [15-17]. It is critical to know how long the DIF stained slides for renal biopsies can be saved at room temperature, as DIF for renal biopsies are at least different from skin DIF in two aspects. First, renal biopsies have more antibody stains including kappa and lambda that are not usually stained for skin DIF. Second, the kidney has unique glomerular basement membranes and mesangium to promote possible attachment of immune complex deposits; the two structures are not present in the skin. In our division, it is our "tradition" to keep the DIF at room temperature for 2 years before they are finally disposed from our archives. To justify the "tradition" for 2 year storage, in this quality control study we retrospectively evaluated DIF slides over 1 to 18 months after the renal biopsy diagnoses were reached. As the first review revealed valid intensity of DIF in renal biopsies over 18 months, the second review in the cases were conducted 40 months later. A significant retardation of DIF intensity was observed at the second follow-up examination. Our study indicates that an 18 month period may be a reliable period for staining preservation of renal DIF slides in cases with immune complex deposits.
Materials and Methods
We retrospectively queried our database for control cases composed of 21 focal segmental glomerulosclerosis (FSGS) and 35 study cases with immune complex mediated glomerulopathy (ICMG), from 01/2009 to 06/2010. The study group mainly included idiopathic membranous glomerulopathy and varying types of lupus nephritis, with one post-infectious glomerulonephritis. The renal frozen sections were washed with Difco FA buffer (100 g Difco dried FA buffer from Fisher Scientific Company LLC, Pittsburgh, PA, dissolved in 1000ml deionized water) for 5 min, incubated with specific fluorescein labeled antibodies against IgG, IgA, IgM, C1q, C3, albumin, kappa and lambda (Dako Cytomation, Carpinteria, CA) for 30 min, rinsed with water, and coverslipped with Permafluor mounting media (Thermo Scientific, Fremont, CA). The DIF slides were kept in dark boxes at lab room temperature (the thermometer has been set at 70℉ in our immune laboratory - 21 ± 1℃), after the initial evaluation for diagnosis. As many types of immune complex diseases are characterized with deposits for mixed heavy chain IgG with both light chains, we decided to re-examine IgG, kappa and lambda for persistent findings in this study. DIF slides for IgG, kappa and lambda were re-examined for the fluorescent intensity after a storage time ranging from 1 to 17.5 months (m) and the fluorescent intensity was scored from 0 to 3+ (the first follow-up). The 35 study cases were further divided into three periods - 1-5 months (n=12), 6 - 11 months (n=10) and 12 to 18 months (n=13) for detail analysis.
The same cases with IgG, kappa and lambda stains underwent a second follow-up examination 40 months later (storage time ranging from 40 to 57.5 months). Similar to the first follow-up, the 35 study cases were further divided into three periods - 41-45 months (n=12), 46 - 51 months (n=10) and 52 to 57.5 months (n=13) during the second follow-up examination.
During the second follow-up period, to determine if there was good preservation of DIF staining in cases with direct antibody binding to glomerular basement membranes, 10 cases with crescentic glomerulonephritis, anti-glomerulus basement membranes (anti-GBM) type, were retrospectively retrieved. The initial scores of IgG, kappa and lambda were identified. The slides with IgG, kappa and lambda, stored at room temperatures, were reviewed.
Results were expressed as the mean ± SEM. Initial reported values (in each biopsy diagnosis) of IgG, kappa and lambda scores were compared with respective values recorded during the first follow-up and the second follow-up examinations, using one way analysis of variance (ANOVA) (Statview program). A p value less than 0.05 was considered statistically significant.
Results
The mean age of patients in FSGS group (56.3 ± 2.8 years, ranging from 40 to 79 years old) was significantly older than that in the ICMGN group (44.5 ± 3.5 years, ranging 5 to 79 years old). All staining scores of the ICMGN group from initial examination, the first follow-up and the second follow-up are listed in table 1. Each case with detailed examination months is also listed in table 1. The FSGS group as the negative control had low staining scores in all three episodes of examinations as expected (Table 2). Over 1 to 17.5 months of the first follow-up, there was no significant decline in staining intensity for all three markers (Table 2), although up to a 12% but non-significant reduction in kappa staining intensity was noted.
![]()
Table 1: Clinical and Direct Immunofluorescent S of All patients from group B (immune complex mediated glomerulopathy)
View Table 1
![]()
Table 2: Mean Value of Original scores of IgG, kappa and lambda, and Current Reading of 3 markers over Time in group A (FSGS) and group B cases (immune complex mediated glomerulopathy [ICMG])
View Table 2
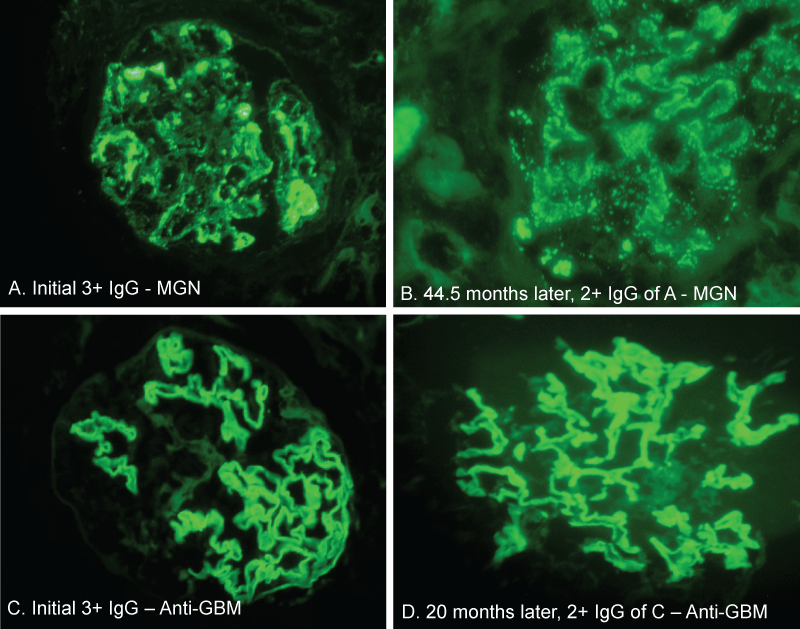
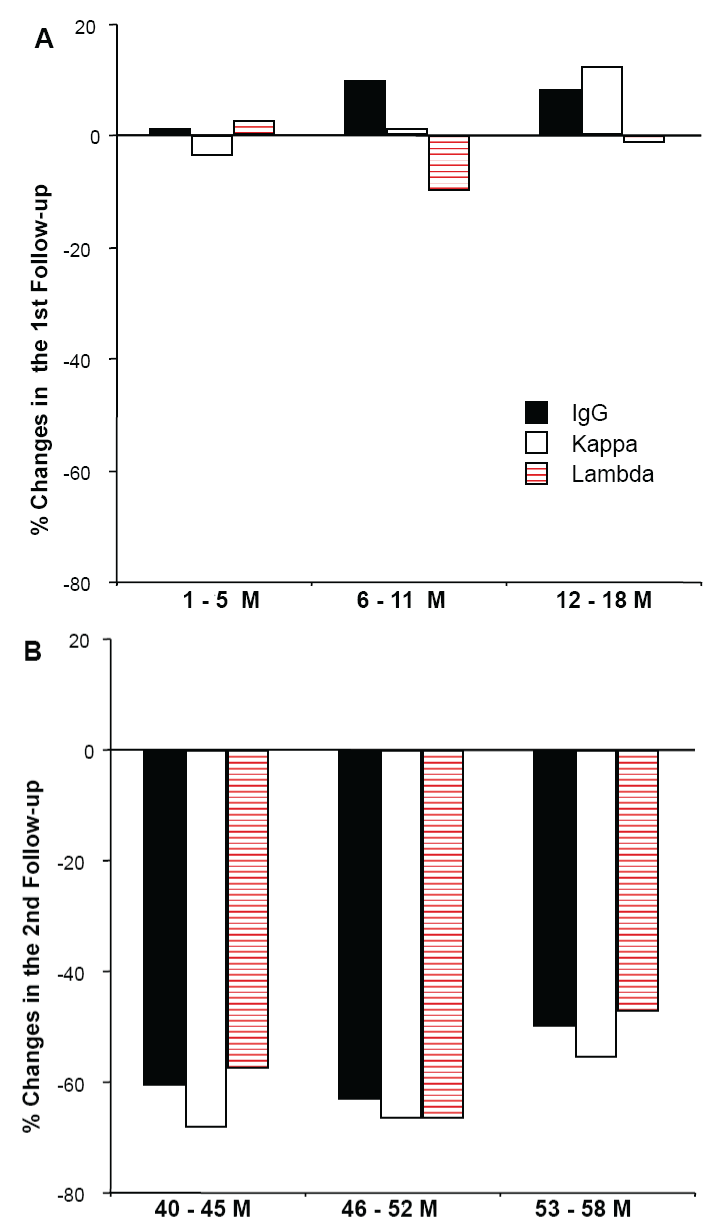
When compared to a 3+ intensity of IgG in a patient with membranous glomerulopathy, the second repeat examination after 45 month storage still revealed a 2+ IgG staining along the glomerular basement membranes (Figure 1A,1B). But in the majority of cases during the second follow-up (41 to 57.5 months), significant decline in staining intensity for all 3 markers was found (Table 2), with up to 68.2% reduction of kappa staining intensity (Figure 2).
.
Figure 1: Direct immunofluorescent stains of IgG at the initial and later follow-up period. There was 3+ IgG staining in a membranous glomerulopathy (panel A) and its follow-up 2+ IgG staining at 45 month later (case 25 in Table 1) (panel B). In an anti-glomerular basement membrane crescentic glomerulonephritis case, 3+ linear IgG staining was seen initially (panel C) and IgG staining remained nearly 3+ strong 20 months later (panel D) (case 7 in Table 3). Note, some storage artifacts (or sign of decay) were somehow seen as shadows along glomerular basement membranes in panel B and panel D. (Magnification x 400 in A - D)
View Figure 1
.
Figure 2: Percent of direct immunofluorescent changes in IgG, kappa and lambda during the first follow-up without significant alteration in staining intensity (panel A), but with significant reductions (p<0.05) of staining intensity for all three makers during the second follow-up (penal B). M - months.
View Figure 2
During the second period of follow-up, we also examined 10 cases with anti-GBM diseases (4 men and 6 women, with a range of age from 45 to 83 year old). Figure 1C and 1D show an example of initial IgG linear staining and its follow-up at 20 months in a patient with anti-GBM crescentic glomerulonephritis (Case 7 in table 3). With a limited number of cases, we observed a maintained 3+ linear IgG staining from 4.5 months to 20 months, but 2 cases around this period almost lost all staining intensity (Table 3).
![]()
Table 3: Follow-up of Immunofluorescent (IF) Stains in Renal Biopsies with Anti-Glomerular Basement Membranes (GBM) Crescentic Glomerulonephritis (GN)
View Table 3
Discussion
Our current study revealed that an 18 month period appears to be a reliable period to store DIF slides that continue to generate sufficient intensity of fluorescent signals for identifying immune complex deposits. The best confirmation of previous DIF staining intensity can be achieved by examining IgG, kappa and lambda stains concurrently as a panel, as we see a small but non-significant variations of staining intensity changes among three markers over time (small changes during the first follow-up and remarkable and significant reductions during the second follow-up). This observation justifies our current departmental "tradition" to store DIF slides for two years at 70℉ room temperature. DIF slides from skin biopsies can be reliably stored for up to 11 months [14]. We speculate that the additional 6 -month period in renal biopsies may be somehow related to the magnetic effects of glomerular basement membranes and mesangium to trap immune complex deposits. In fact, we did not notice any immune complex deposits in IgA associated leukocytoclastic vasculitis of skin by electron microscopy, while electron microscopy in their respective renal biopsies clearly showed immune complex deposits in mesangial areas and focal subendothelial spaces of the two IgA nephropathy cases (unpublished observation). This implies a larger quantity of immune complex deposits in glomeruli than in skin from the beginning of the disease.
During the second follow-up examination (40 months from the initial biopsies) of our renal DIF slides, significant reduction of immunofluorescent staining intensity was indeed seen in all three markers IgG, kappa and lambda. This finding is compatible with the notion that DIF intensity will fade away over time. Repeat examination of the same slides during the initial diagnosis and during the first follow-up examination may also contribute to the staining decay during the second follow-up examination.
Our study has a few limitations. Our limited cases of anti-GBM biopsies also support that DIF intensity showed some staining preservation, although two cases faded away quickly than expected (see Table 3 and text for details). Anti-GBM antibodies directly attack against the target antigen along glomerular basement membranes and electron microscopy usually does not show obvious deposits due to their smaller antigen-antibody complex size (thus linear staining for IgG). Therefore anti-GBM biopsies do not have the initial sum of deposit aggregates as seen in immune complex mediated glomerulopathies. Second, we only examined some types of immune complex mediated glomerulopathies, but we did not examine IgA nephropathy, monoclonal light chain nephropathy and complement mediated glomerulopathies, namely dense deposit disease and C3 dominant glomerulopathy, therefore we are uncertain how long DIF intensity in those entities would last reliably.
Many of clinical laboratories worldwide still use Buffered Glycerol as a mounting medium for DIF. The permamnent coverslipping mounting media may not only ensure no move between the coverslips and glass slides, but may be also related to a better preservation of DIF intensity. Therefore some natures of the mounting media we used including anti-fading capacity, viscosity, pH and antifungal additives of the mounting would be important to know. But when we wrote a request regarding the mounting medium components to the company, we did not get a reply from them. These components in the mounting medium appear to be their trade secrets as the information is not available on their website either. Another issue is that many clinical laboratories may keep the DIF slides in refrigerators at 0 - 4℃ for a longer preservation of DIF intensity. However, we have hundreds of renal biopsies and skin biopsies with DIF stains each year, and it is not practical to keep all of them in a refrigerator over 2 years of period, thus we do not have data to determine whether a low temperature would have an advantage over the room temperature for maintaining a better preservation of DIF intensity.
Probably due to widespread medical knowledge to patients through internet search, we have encountered 2-fold increase in requests for second opinions of our surgical pathology cases, including renal biopsies over the past 10 years (unpublished observation). The reliable storage period at 18 months for the renal DIF slides found is a useful guideline for us to decide whether we should send out DIF slides together with light microscopy slides and a CD with the electron microscopy images, in face of a second opinion request. After two years of storage in room temperature (70℉ or 21 ± 1℃), our data showing a huge decay during the second review have also provided us with a confident information that the DIF slides can be permanently removed from our laboratory by then. We still store the frozen blocks of renal biopsies for 10 years, in case re-stains are needed for a review.
References
-
Vassalli P, Morris RH, McCluskey RT (1963) The pathogenic role of fibrin deposition in the glomerular lesions of toxemia of pregnancy. J Exp Med 118: 467-478.
-
Morris RH, Vassalli P, Beller FK, McCluskey RT (1964) Immunofluorescent studies of renal biopsies in the diagnosis of toxemia of pregnancy. Obstet Gynecol 24: 32-46.
-
Gagliano RG, Costanzi JJ, Beathard GA, Sarles HE, Bell JD (1976) The nephrotic syndrome associated with neoplasia: an unusual paraneoplastic syndrome. Report of a case and review of the literature. Am J Med 60: 1026-1031.
-
Moriyama M, Fukuda Y, Ishizaki M, Sugisaki Y, Masugi Y (1976) Membranous glomerulonephritis associated with active liver cirrhosis both involved by HBs antigen. Acta Pathol Jpn 26: 237-250.
-
Roy S 3rd, pitcock JA, Etteldorf JN (1976) Prognosis of acute poststreptococcal glomerulonephritis in childhood: prospective study and review of the literature. Adv Pediatr 23: 35-69.
-
Collins AB, Schneeberger EE, Pascual MA, Saidman SL, Williams WW, et al. (1999) Complement activation in acute humoral renal allograft rejection: diagnostic significance of C4d deposits in peritubular capillaries. J Am Soc Nephrol 10: 2208-2214.
-
Colvin RB (2007) Antibody-mediated renal allograft rejection: diagnosis and pathogenesis. J Am Soc Nephrol 18: 1046-1056.
-
Sethi S, Fervenza FC (2012) Membranoproliferative glomerulonephritis--a new look at an old entity. N Engl J Med 366: 1119-1131.
-
Pickering MC, D'Agati VD, Nester CM, Smith RJ, Haas M, et al. (2013) C3 glomerulopathy: consensus report. Kidney Int 84: 1079-1089.
-
Cook HT (2013) Complement and kidney disease. Curr Opin Nephrol Hypertens 22: 295-301.
-
Soltani K, Taylor ME, Choy RK (1976) Preservation of specific fluorescence by sealing with nail polish. J Invest Dermatol 66: 332.
-
Balaton AJ, Dalix AM, Oriol R (1985) An improved mounting medium for immunofluorescence microscopy. Arch Pathol Lab Med 109: 108.
-
Valnes K, Brandtzaeg P (1985) Retardation of immunofluorescence fading during microscopy. J Histochem Cytochem 33: 755-761.
-
Dikicioglu E, Meteoglu I, Okyay P, Culhaci N, Kacar F (2003) The reliability of long-term storage of direct immunofluorescent staining slides at room temperature. J Cutan Pathol 30: 430-436.
-
Suzuki Y, Maesawa A, Matsui K, Shimizu F, Oite T, et al. (1984) Restoration of antigenicity of tissue antigens, cell-bound immunoglobulins and immune deposits in paraffin-embedded tissue. The influence of fixation and proteolytic enzymatic digestion. Acta Pathol Jpn 34: 563-574.
-
Tulunay O (1982) Immunofluorescent staining of plastic-embedded renal tissue. Arch Pathol Lab Med 106: 632-634.
-
Fogazzi GB, Bajetta M, Banfi G, Mihatsch M (1989) Comparison of immunofluorescent findings in kidney after snap-freezing and formalin fixation. Pathol Res Pract 185: 225-230.