International Journal of Virology and AIDS
Comprehensive Programme to Prevent Cervical Cancer-the Case of Hungary
Döbrőssy L*, Oroszi B, Kovács A and Budai A
Office of Chief Medical Officer, Budapest, Hungary
*Corresponding author: Döbrőssy L, MD, Office of Chief Medical Officer, Albert Flórián út 2-4, Budapest, H-1097, Hungary, Tel: + 36 30 536 0 375, E-mail: dobrossy.lajos@oth.antsz.hu
Int J Virol AIDS, IJVA-3-021, (Volume 3, Issue 1), Case Report; ISSN: 2469-567X
Received: February 12, 2016 | Accepted: April 28, 2016 | Published: April 30, 2016
Citation: Döbrőssy L, Oroszi B, Kovács A, Budai A (2016) Comprehensive Programme to Prevent Cervical Cancer-the Case of Hungary. Int J Virol AIDS 3:021. 10.23937/2469-567X/1510021
Copyright: © 2016 Döbrőssy L, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The overall purpose of a comprehensive cervical cancer prevention programme is to coordinate HPV vaccination (primary prevention) and cervical screening (secondary prevention). Harmonization of the HPV vaccination and population screening is expected to reduce the burden of cervical cancer by reducing human papillomavirus (HPV) infections, and detecting cervical pre-cancer lesions and invasive cancer earlier, providing timely treatment. In 2015, 80% of 7th class schoolgirls between 12-13 years of age received HPV vaccination in Hungary. Compliance with the offered screening needs to be increased.
Keywords
HPV infection, HPV vaccination, Cytology/HPV DNA screening, CIN 2-3 and cervical cancer
Introduction
Every year approximately 60,000 new cervical cancers (C53) are diagnosed, and 25,000 fatal cases are reported in Europe. Cervical cancer is the 7th most common cause of cancer death in Europe for females. The highest age-standardized mortality rates for cervical cancer are in Romania; the lowest rates are in Iceland. Hungarian cervical cancer mortality rates are estimated to be the 9th, among the highest one-third in Europe (6.9/100.000) [1,2].
Over the past 50 years, the single strategy of reduction of mortality of this disease of public health problem was early detection and early treatment by the organized or opportunistic screening by cytology. Recently, it has been recognized that the development of cervical cancer is causally connected to human papillomavirus (HPV) infection [3], against which recently vaccination has been introduced. All the authoritative international organisations, such as World Health Organization (WHO) [4], Centre for Disease Control and Prevention (CDC) [5]. American Cancer Society (ASC) [6], European Commission [7] recognize the importance of cervical cancer as global public health problem and reiterate recommendation that in addition to screening programmes, HPV vaccines be included in national immunization programmes.
Human Papillomavirus
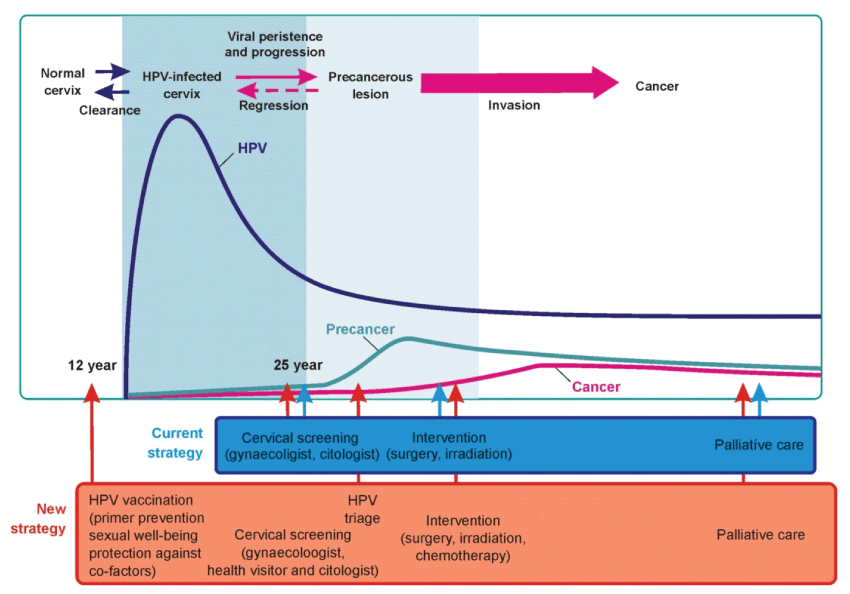
It is an old standing observation that cervical cancer behaves like a sexually transmitted disease. In the recent decades a vast body of evidence has emerged indicating the persistence of HPV infection as the necessary, although not sufficient cause of cervical cancer [3]. Other cofactors needed for progression range from cervical HPV infection to cancer [8]. Today, the causal role of Human Papillomavirus (HPV) in the development of cancer of the uterine cervix has been firmly established both biologically and epidemiologically [9-12]. There are over 100 types of HPV. Persistent viral infection with high-risk human papillomavirus genotypes causes virtually all cancers of the cervix, but two – types 16 and 18 – account for about 70% of cervical cancers [13,14]. The natural history of HPV infection and that of cervical cancer development has been shown on figure 1.
Secondary Prevention: Screening
The screening makes use of natural history of cervical cancer. There is a shorter or longer period of time when the still asymptomatic process has become detectable by suitable method, in this case cytology. This period is called preclinical detectable phase (PCDC); this period lends itself to screening [15]. The target conditions of screening are the precursor lesions and the invasive cancer of the uterine cervix. The sensitivity and specificity of morphology-based screening tests is somewhat limited, but satisfactory [16]. The screening services have led to major reductions of mortality from invasive cervical cancer in most developed countries; however, due to lack of resources or inadequate infrastructure, or some other reason, many countries have failed to reduce cervical cancer mortality through screening [17].
However, cytology, as a screening tool, is based on a highly subjective interpretation of morphologic alterations present in cervical samples. Recently, HPV DNA testing, as the molecular marker of infection, has emerged as a likely candidate to replace traditional cytology for primary screening. It is less prone to human error and more sensitive in detecting high-grade cervical lesions than morphology-based cytology [18,19]. HPV testing has clearly demonstrated a higher sensitivity but somewhat lower specificity than traditional cytology; therefore, it is likely to cause more adverse effects, such as over diagnosis and overtreatment, and psychological side-effects, as well [20]. The HPV DNA test can be used in combination with the traditional cytology test to screen for cervical cancer, particularly for women 30 years of age and older, For younger women, false positive test results might be common. However, it does not replace the Pap test for most women. Appropriate policy and programme organization remain essential to achieve an acceptable balance between benefit and harm of any screening programme [21].
Primary Prevetion: Vaccination Programmes
Because persistent HPV infections can cause cervical cancer, the public health goal of vaccination is to reduce HPV infections. Three prophylactic vaccines are currently available and marketed in many countries worldwide for the prevention of HPV-related diseases: a quadrivalent vaccine, a bivalent vaccine and a 9-valent vaccine, all the three directed against oncogenic genotypes [4,22].
The primary target group for vaccination in Europe is girls of an age before sexual activity starts. HPV vaccine is recommended for girls at age 9 or 12 so they are protected before ever being exposed to the virus. Catch up vaccination is also recommended for females 13 through 26 years of age who have not been previously vaccinated. School-based immunisation, as part of school vaccination programmes, is likely to be the lowest-cost and most effective option for delivery of HPV vaccines to pre-adolescent girls.
Since 2008, HPV vaccination programmes have been implemented in most EU countries. By 2012, 19 out of 29 countries in the EU (including Norway and Iceland) had implemented routine HPV vaccination programmes, and 10 countries had also introduced catch-up programmes. Despite the efforts made by individual Member States, coverage rates - where data are available - are lower than expected in many EU countries. Furthermore, target age, system of financing and delivery of the vaccines differ from one country to another and coordination among EU countries is lacking [23].
Comprehensive Preventive Strategies
Harmonisation of primary and secondary prevention, i.e. vaccination and screening promises a better chance for prevention of cervical cancer. Until recently, cervical cancer strategies focussed on screening. However, as adolescents become sexually active at a much younger age, the focus is on the use of vaccination as an effective measure to prevent HPV infection, and development of cancer [24,25]. In addition, the vaccines can reduce the need for medical care, biopsies, and invasive procedures associated with the follow-up from abnormal cytology tests, thus helping to reduce health care costs and anxieties related to abnormal cytology and follow-up procedures.
It is clear that combination of cervical screening together with HPV vaccination in comprehensive cervical cancer prevention programme will offer the most effective long-term protection against cervical cancer. However, discontinuation of screening for precancerous lesions or early cervical cancer of cervix is not justified because vaccination will not protect against HPV types not included in the first generation of vaccines. Furthermore, cervical screening is still needed for women who have not yet been vaccinated.
In addition to HPV vaccination and screening, the coordinated approach to prevention of cervical cancer requires complex interventions including education on healthy sexual practices for girls and boys, tailored as appropriate to age and culture, with the aim of reducing the risk of HPV transmission. Essential messages should include delay of sexual initiation, and reduction of high-risk sexual behaviors. Social mobilization to accept the offered vaccination is important.
Harmonization of Primary and Secondary Preventive Programme in Hungary
In Hungary, opportunistic cervical screening goes back to the early 1960s. By the end of 1970s, sufficient capacity was in place to screen the eligible women every 3 years. However, due to the lack of individual identification of women screened, only the number of smears analyzed was registered, and nobody knew who had been screened and who has never been screened. Opportunistic screening, even though very extensive, did not achieve its goal. In the 1990s, the "close the gap" programme cosponsored by the World Bank created a favorable policy environment for integrating organized population screening as a core function of healthcare system, and a golden opportunity presented itself for reorganizing and updating cervical screening in line with international recommendations [26].
In Hungary, the "supply side" of organized mass screening for early detection of cervical cancer and its precursors has been established. The responsibility for management, coordination, monitoring and evaluation has been delegated to the Chief Medical Officer. The existing system would be capable of carrying out the screening of the whole population eligible by age in the recommended intervals. Since 2003, organized screening programme for cervical cancer has been in operation [27], following the recommendations of the European Council [7]. The notification list for personal invitation is managed by National Screening Registry established in the frame of Screening Coordination Department of the Chief Medical Officer's Office. The notification list is set up using the population list received from the database maintained by the National Health Insurance Fund Administration (OEP). Women between 25-64 years of age are invited three-yearly for cytology-based screening by personalized invitation letter. Smears are taken by gynecologists, and in rural areas by primary care personnel (district health visitors), and analyzed in contracted cytology laboratories. Those with negative test results are recalled for repeated test in three years' time; those with non-negative (positive) test results are referred to gynecological services for colposcopy and other diagnostic procedures to confirm or to rule out premalignant lesions (CIS 1-3) or early cancers of uterine cervix.
However, the "demand side"- that is, the compliance of the target population with the offered screening - has remained below expectations. It is fair to say that the organized cervical screening facilities in Hungary are unexploited, as a consequence, both the effectiveness and economic viability of the programmed is damaged. According to the National Screening Registry, in 2013, 29.6% of those invited attended the organized cervical screening. During the same period, the National Health Insurance Fund Administration paid for diagnostic cervical cytology ten-times more often, meaning that the majority of screened women received screening outside the organized programme, such as in gynecological services and private clinics, which failed to report the screening test performed to the National Screening Registry. According to learned estimates, approximately three-fourths of the eligible female population received organized or opportunistic cervical screening; and about one-fourth of the female population has never been screened. Between the years 2003-2012, the National Cancer Registry unpublished data report indicated, that incidence of new cervical cancer cases decreased from 1333 (17.7/100.000) in 2003, to 1120 (13.9/100.000) in 2012. As the Central Statistical Office annual report says, in the same period of time, the cervical cancer mortality figures reduced from 465 (5.4/100.000) in 2003 to 405 (4.6/100.000) in 2013. The moderate reduction of incidence and mortality is obviously attributable to organized and opportunistic screening activity.
There are various causes of non-attendance, such as novelty of the concept of "organized screening", limited access to screening facilities, particularly in rural areas. Socio-economic status has an influence: the well-to-do, more highly educated women are more likely to be screened ("selection bias") as compared with those living on the shadowy side of the life.
During the last decades, in Hungary, there has been a growing interest in the diagnostic hybrid capture HPV DNA (HC2) test, at first, to investigate prevalence of genital human papillomavirus (HPV) infection, secondly, as a primary screening test, solely or in combination with traditional, morphology-based cytology to detect cervical cancer precursors in asymptomatic women. Several cytology laboratories, particularly those run by gynecological departments, and private clinics carry out such investigations to get experience in performing the test [28-30]. However, the HPV DNA test is not reimbursed, and it is not yet applied routinely in organized population screening programme.
Both HPV vaccines, the quadrivalent and the bivalent, have been registered in Hungary since 2006 [31]. (There is no experience so far with 9-valent vaccine.) The Chief Medical Officer brought together a vaccination Advisory Board to study the experiences accumulated in other countries, and the feasibility, including cost effectiveness, of a national vaccination programme. In the meantime, two modelling studies have confirmed the cost effectiveness of adding the available vaccines [32,33]. Since 2010, HPV vaccination had been implemented as local initiative in some 300 (out of 3.600) municipalities, supported fully or partly by local governments; in addition, parents might have decided to buy the vaccine, available in pharmacies, to get their children vaccinated. During this period, about 32.000 girls have received vaccination. Data on these early local initiatives are not at our disposal.
Finally, the Advisory Board produced a recommendation to the Government, taken into account that cervical cancer is a considerably important public health concern in the country, and the accumulated evidence proves that cervical cancer is preventable by HPV vaccination, furthermore, a large body of evidence proves that the vaccines are effective, safe, efficient, and cost-effective, the Government has decided to integrate HPV vaccination into the national immunization programme.
Since 2014, a nation-wide vaccination programme is in operation. The vaccination, fully financed by national health authorities, has been carried out as a school-based programme: female students of 7th grade of primary schools (age group of 12-13 years of age) are being vaccinated. The early experiences show that success in terms of coverage of the target groups has been reasonably high: compliance with the offered vaccination was 80%.
HPV vaccination does not eliminate the need for cervical cancer screening, National authority and management of screening programmes should continue their efforts to organize and improve the coverage and quality of ongoing programmes. Vaccinated women might have a false sense of security, resulting in lower attendance at screenings. Women need to be informed and motivated to attend screening programmes, even if they are vaccinated. To achieve synergy between vaccination and screening is a cost-effective way and with the maximum benefit for women.
Conclusion
HPV vaccination and screening are not alternatives to each other, and should be treated as an integrated system. Introducing the HPV vaccine more widely, in combination with cervical screening, has the potential to further lower mortality rates, particularly among younger women. However, in the future, HPV vaccination may allow reducing screening programmes: deferring of age starting screening, lengthening the time interval between screening rounds or modulating the proper timing between vaccination and screening. All these modulations might translate into new screening algorithms for immunised women and could provide evidence of cost-effectiveness of papillomavirus vaccine.
References
-
European Cancer Observatory IARC. 2012.
-
Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JW, et al. (2013) Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer 49: 1374-1403.
-
Bosch FX, Lorincz A, Muñoz N, Meijer CJ, Shah KV (2002) The causal relation between human papillomavirus and cervical cancer. J Clin Pathol 55: 244-265.
-
(2014) Human papillomavirus vaccines: WHO position paper, October 2014. Wkly Epidemiol Rec 89: 465-491.
-
Center for Disease Control and Prevention (2011) HPV and cancer. MMWR 60: 1-64.
-
Saslow D, Castle PE, Cox JT, Davey DD, Einstein MH, et al. (2007) American Cancer Society Guideline for human papillomavirus (HPV) vaccine use to prevent cervical cancer and its precursors. CA Cancer J Clin 57: 7-28.
-
Council of the European Union (2003) Council recommendation of 2 December 2003 on cancer screening. Office J Eur. Union L 327: 85-89.
-
Wang SS, Zuna RE, Wentzensen N, Dunn ST, Sherman ME, et al. (2009) Human papillomavirus cofactors by disease progression and human papillomavirus types in the study to understand cervical cancer early endpoints and determinants. Cancer Epidemiol Biomarkers Prev 18: 113-120.
-
zur Hausen H, de Villiers EM, Gissmann L (1981) Papillomavirus infections and human genital cancer. Gynecol Oncol 12: S124-128.
-
zur Hausen H (1986) Intracellular surveillance of persisting viral infections. Human genital cancer results from deficient cellular control of papillomavirus gene expression. Lancet 2: 489-491.
-
zur Hausen H (2000) Papillomaviruses causing cancer: evasion from host-cell control in early events in carcinogenesis. J Natl Cancer Inst 92: 690-698.
-
Parkin DM, Bray F (2006) Chapter 2: The burden of HPV-related cancers. Vaccine 24 Suppl 3: S3/11-25.
-
Franco EL, Rohan TE, Villa LL (1999) Epidemiologic evidence and human papillomavirus infection as a necessary cause of cervical cancer. J Natl Cancer Inst 91: 506-511.
-
Althouse R, Huff J, Tomatis L, Wilbourn J (1979) Chemicals and industrial processes associated with cancer in humans. IARC Monographs, Volumes 1 to 20. IARC Monogr Eval Carcinog Risk Chem Hum Suppl: 1-71.
-
Miller AB (1984) Biological aspects of natural history and its relevance to screening. In: Prorock PC, Miller AB (eds). Screening for cancer. UICC Techn Rep Series 78: 44-53.
-
Ronco G, Cuzick J, Pierotti P, Cariaggi MP, Dalla Palma P, et al. (2007) Accuracy of liquid based versus conventional cytology: overall results of new technologies for cervical cancer screening randomized controlled trial. Brit Med J 335: 28-31.
-
Anttila A, Ronco G, Working Group on the Registration and Monitoring of Cervical Screening Programmes in the European Union; within the European Network for Information on Cancer (2009) Description of the national situation of cervical cancer screening in the member states of the European Union. Eur J Cancer 45: 2685-2708.
-
Coste J, Cochand-Priollet B, de Cremoux P, et al. (2003) Cross sectional study of conventional cervical smear; monolayer cytology, and human papillomavirus DNA testing for cervical cancer screening. Brit Med J 326: 733-736.
-
Naucler P, Ryd W, Törnberg S, Strand A, Wadell G, et al. (2007) Human papillomavirus and Papanicolaou tests to screen for cervical cancer. N Engl J Med 357: 1589-1597.
-
Hakama M, Coleman MP, Alexe DM, Auvinen A (2008) Cancer screening: evidence and practice in Europe 2008. Eur J Cancer 44: 1404-1413.
-
von Karsa L, Arbyn M, De Vuyst H, A Anttila, J Jordan, et al. (2015) European guidelines for quality assurance in cervical cancer screening. Summary of the supplements on HPV screening and vaccination. Papillomavirus Res 1: 22-31.
-
Petrosky E, Bocchini Jr JA, Hariri S (2015) Use of 9-Valent Human Papillomavirus (HPV) Vaccine: Updated HPV Vaccination Recommendations of the Advisory Committee on Immunization Practices. Morbidity and Mortality Weekly Report (MMWR) 64: 300-304.
-
European Centre for Disease Prevention and Control (2012) Introduction of HPV vaccines in European Union countries -an update. ECDC. Stockholm.
-
World Health Organization (2013) Comprehensive cervical cancer prevention and control: a healthier future for girls and women, Geneva
-
Louvanto K, Chevarie-Davis M, Ramanakumar AV, Franco EL, Ferenczy A (2014) HPV testing with cytology triage for cervical cancer screening in routine practice. Am J Obstet Gynecol 210: 474.
-
Döbrőssy L (1999) Efforts to update the cancer screening in Hungary. The catalytic role of secondary prevention of World Bank supported programme. Népegészségügy 5: 23-35.
-
Kovács A, Döbrőssy L, Budai A, Boncz I, Cornides A, et al. (2007) The state of the organized cervical screening programmes in Hungary in 2006, Orv. Hetil 148: 35-540. (In Hungarian)
-
Nyári T, Cseh I, Woodward M, Szöllösi J, Bak M, et al. (2001) Screening for human papillomavirus infection in asymptomatic women in Hungary. Hum Reprod 16: 2235-2237.
-
Koiss R. Siklós P (2010) A HPV and cervical cancer. LAM 20: 96-102.
-
Döbrőssy L, Kovács A, Budai A. Strategy of cervical screening era. Orvostovábbképző Szemle 19: 49-56.
-
Seme K, Maver PJ, Korać T, Canton A, Částková J, et al. (2013) Current status of human papillomavirus vaccination implementation in central and eastern Europe. Acta Dermatovenerol. AP 22: 21-25.
-
Dasbach EJ, Nagy L, Brandtmüller A, Elbasha EH (2010) The cost effectiveness of a quadrivalent human papillomavirus vaccine (6/11/16/18) in Hungary. J Med Econ 13: 110-118.
-
Vokó Z, Nagyjánosi L, Kaló Z (2012) Cost-effectiveness of adding vaccination with the AS04-adjuvanted human papillomavirus 16/18 vaccine to cervical cancer screening in Hungary. BMC Public Health 12: 924.