International Journal of Virology and AIDS
Organotropic Kinetics in Acute Sublethal Transient Cytomegalovirus Infection in Mice
Daniel Benharroch1*, Ory Rouvio2, Aharon Palmon3 and Bracha Rager2
1Department of Pathology, Soroka University Medical Center and Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer-Sheva, Israel
2Department of Microbiology-Immunology-Genetics, Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer-Sheva, Israel
3Department of Oral Biology, Hadassah Faculty of Dental Medicine, Hebrew University, Jerusalem, Israel
*Corresponding author: Daniel Benharroch, Department of Pathology, Soroka University Medical Center, P.O.Box 151, 1, Ytzhak Rager Avenue, Beer-Sheva, 84101, Israel, Tel: 972-8-6400920; Fax: 972-8-6232770; E-mail: benaroch@bgu.ac.il
Int J Virol AIDS, IJVA-3-015, (Volume 3, Issue 1), Research Article; ISSN: 2469-567X
Received: October 15, 2015 | Accepted: December 30, 2015 | Published: January 03, 2016
Citation: Benharroch D, Rouvio O, Palmon A, Rager B (2016) Organotropic Kinetics in Acute Sublethal Transient Cytomegalovirus Infection in Mice. Int J Virol AIDS 3:015. 10.23937/2469-567X/1510015
Copyright: © 2016 Benharroch D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We determined the primary level of dissemination of the cytomegalovirus into the mouse organs and its significance. This was completed before we engaged in a wider project on the immune and molecular aspects of the pathogenesis of MCMV infection in the Balb/c mice. By this venture, we may have opened opportunities to develop one or more vaccines against the virus. Mice were inoculated intraperitoneally with the virus. At day 1, 3, 7, 14 and 28 after infection, the murine organs including the spleen, salivary glands, inguinal lymph nodes, liver, bone marrow, and lungs, were collected. The organs were assessed for the expression of MCMV-DNA, for the infectious viral titer and part of the tissues was formalin-fixed and paraffin-embedded for morphological evaluation. Each organ was found to present a different configuration of the three components reflecting the viral spread. Thus, the lungs and the bone marrow showed high levels of DNA and low titers of infectious virus, taken to mean evidence of latency. The salivary glands revealed evidence of a persistent infection, as expressed by high levels of MCMV-DNA and constant titers of infective virus that lasted beyond day 28. However, the most remarkable finding was that the latency probably discerned in the spleen was localized in the stromal cells, but not in the lymphocytic component. To this extent, we may have determined the MCMV journey in the mouse.
Keywords
Cytomegalovirus, BALB/c mouse, MCMV-DNA, Latency, Persistence
Introduction
Murine cytomegalovirus (MCMV) shows many similarities with the human virus (HCMV). This is a ubiquitous DNA virus from the betaherpesvirus group. A self-limiting acute phase of viral infection and a persistent latent phase normally characterize the pathogenesis of MCMV infection in the immunocompetent host [1]. The MCMV widespread distribution occurs via the blood and its mononuclear cells [2,3]. In the more common congenital viral infection, the HCMV causes birth defects, including mal-development of the central nervous system. In addition, it becomes re-activated in immunocompromised patients [4]. Thus, these grave sequels underline the need for new therapeutic modalities and for effective anti-HCMV vaccines [5]. Murine cytomegalovirus is a natural pathogen of mice similar to HCMV in physical structure and in pathogenesis. Until the advent of the guinea-pig model, still to become widely adopted [6], the murine model was the closest to optimal for the study of HCMV.
In this study, we investigated the pathogenesis of MCMV infection in BALB/c mice aged 6-8 weeks, as younger animals are prone to die early of the infection. We included female mice only, as this gender has originally been considered to be more resistant to systemic viral infections [7,8]. In the selected mice, an acute, sublethal and transient infection is followed within 10-14 days from intraperitoneal inoculation, by apparent recovery.
We studied the organ-specific dissemination and involvement, in the framework of a time sequence. In the selected organs, we detected the MCMV-DNA by PCR, as it correlates with the productive MCMV infectivity, and with the histopathological changes in the organ. We also looked for evidence of apoptosis, using the TUNEL assay.
Acute primary MCMV infection inevitably develops into persistent and latent infection. Persistence is defined as a chronic infection with a relatively low level of virus replication. Latency refers to a complete absence of infectious virus, with evidence of at least one viral component such as viral DNA or proteins. There is growing evidence that cells in the bone marrow and peripheral blood are key reservoirs for HCMV through latent infection. It has been suggested that cells of hematopoietic origin play a major role in pathogenesis and latency [9].
Material and Methods
Mice
Female BALB/c mice were purchased from Harlan Olac (Jerusalem, IL), at the age of 14 days (10-12 g body weight). Mice were bred and maintained under specific-pathogen-free conditions at our institute, and used for experiments at 6-8 weeks of age. Until then, the mice were maintained in isolation in our vivarium and were fed and watered at libitum. All procedures with animals were carried out in accordance with institutionally approved protocols, according to guidelines of the Animal Ethics and Experimentation Committee of the Ben Gurion University of the Negev and were approved by it.
Virus, preparation of virus pools and virus titrations
The growth medium from each well in mouse embryo fibroblast (MEF) plates was aspirated, and duplicate wells were inoculated with 0.2 ml of diluted salivary gland suspension (SGS). After an adsorption period of 1 hour at 37°C, monolayers were overlayed with 0.8 ml of growth medium containing 0.75% carboxymethyl cellulose (CMC), incubated for 5 days at 37°C in a humidified 5% CO2 incubator, fixed in PBS-10% formaldehyde and stained with Crystal Violet to visualize the virus plaques.
The Smith strain of MCMV was obtained from the American Type Culture Collection (Rockville, MD). Highly virulent virus pools, prepared from salivary glands of infected mice as previously described, were used throughout this study [10]. Virus stocks containing 0.5-1 × 108 PFU/0.1 g of tissue were stored at -70°C in 10% dimethyl sulfoxide (DMSO) until use. MCMV titers in SGS pools and in the various organs were determined by a semiquantitative plaque assay [11]. Briefly, confluent monolayers of secondary MEF were prepared in 24 well plates. Serial 10 fold dilutions of fluids containing MCMV were prepared in DMEM supplemented with 2% FCS. The growth medium from each well was aspirated, and duplicate wells were inoculated with 0.2 ml of sample dilution. After an adsorption period of 1 hour at 37°C monolayers were overlayed with 0.8 ml of growth medium containing 0.75% CMC, incubated for 5 days at 37°C in a humidified 5% CO2 atmosphere, fixed in 10% formaldehyde in PBS and stained with Crystal violet to visualize virus plaques. Titers were expressed as log10 PFU/0.1 g tissue.
Infection of mice with MCMV
Eight weeks old female BALB/c mice were infected intraperitoneally (i.p.) with 5 × 104 PFU of stock virus. Two mice of each group were sacrificed at 1, 3, 7, 14 and 28 days after infection and organs were dissected for PCR analysis, virus titration, histopathological examination and apoptosis. Liver, bone marrow (BM), inguinal lymph nodes (LN), spleen, lungs, and salivary glands (SG) were removed and 10% (w/v) homogenates were prepared. Extracts were partially clarified by low speed centrifugation, 10% DMSO was added and samples were stored at - 70°C, until assayed for infectious virus as described.
Preparation and detection of MCMV DNA (PCR)
DNA extraction from the organs was performed as previously reported [10]. In order to identify MCMV DNA, PCR amplification of mouse DNA containing viral DNA was used. DNA oligonucleotide primers that were selected from the published sequence of MCMV gB gene were synthesized [12]. The sense primer was based on the cDNA sequence no. 2416-2443 (5’-AAG-CAG-CAC-ATC-CGC-ACC-CTG-AGC-GCC) and the antisense no. 2745-2772 (5’-CCA-GGC-GCT-CCC-GGC-GGC-CCG-CTC-TCG-3’). This gB gene primer pair amplifying a 356bp segment has been found to be more sensitive than that of the IE gene, in previous studies [10,13]. Identification of PCR amplified DNA was done by hybridization with [32P]-labeled oligonucleotide probe synthesized according to gB gene sequence no. 2574-2594 (5’-TCG-TCA-GGA-AGC-CCG-TGA-CG-3’, reverse and complemented probe) as described previously [10]. PCR was normalized against Actin. Autoradiography was performed at -70°C with one intensifying screen and Fuji Medical X-ray film.
Light microscopy and histopathological examination
Liver, BM (from the femur), inguinal LN, spleen, lung, and SG were removed from infected mice, and placed in 10% formalin. Tissues were embedded in paraffin and 5 μ sections were stained with hematoxylin and eosin (H&E) for histopathological evaluation.
Apoptosis assay
Apoptosis was detected using the TUNEL method (terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling, and the ApopTag Kit, Intergen, Oxford, UK). The 3’-OH DNA ends generated by the DNA fragmentation of apoptosis were tagged with digoxigenin-dUTP catalyzed by terminal deoxynucleotidyl transferase. Anti-digoxigenin peroxidase was added. The resulting complex was then visualized with the diaminobenzidine substrate. Apoptotic cells were identified by golden-brown staining of their nuclei and apoptotic bodies.
Splenic cells fractionation
Splenic stromal and lymphoid cell fractions were prepared as described [14]. Briefly, spleens were removed and placed in RPMI 1640 medium plus 10% FCS. The spleen was extruded and washed through holes made in the capsule-end, using a 18-gauge bent needle. The empty capsule was discarded. Then, the spleen content was suspended in 12 ml medium and transferred to a 15 ml centrifuge tube. The stromal fragments were defined as those fragments which settled to the bottom of the tube in 10 min at 1 x g. The lymphoid fraction was defined as the splenocytes remaining in suspension.
Results
MCMV disease in liver
In this organ, the low amounts of MCMV DNA which were detected on day 1, rapidly increased reaching a peak on day 3 (Figure1A and Figure 1B). This was followed by a marked decrease in the amount of DNA on day 7 which persisted through day 14 at which time only minute amounts of DNA were observed. By day 28, no MCMV-DNA was detected. In these experiments we used two different sets of primers, gB and IE for the amplification of MCMV DNA. As shown in figure 1A, on day 1 MCMV DNA was detected using IE while on day 14 MCMV DNA was detected with the gB primers (Figure1 B). Parallel to the DNA, peak amounts of infectious virus (3 × 105 PFU/0.1g) were isolated from livers on day 3 (Figure 1C). A gradual decline in viral replication was detected on day 7 with virus titers were 6.3 × 103 PFU/0.1g and by day 14, 1 × 102 PFU/0.1 g of the virus were occasionally detected. On day 28, no infectious MCMV could be recovered.
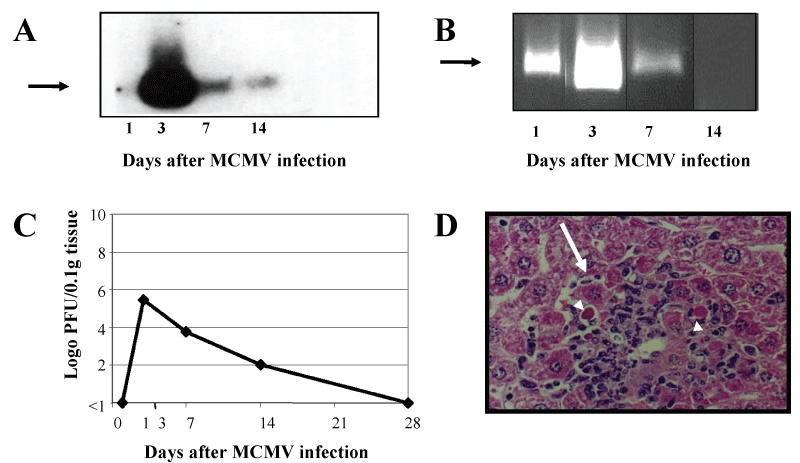
.
Figure 1: MCMV disease in liver. Groups of 6-8, eight-week-old Balb/c female mice were infected i.p. with 5 × 104 PFU of virus and were sacrificed on days 1,3,7,14 and 28 days. (A) PCR amplification of 356 bp product (arrow) of MCMV gB DNA (data presented is from pooled liver DNA of 3 mice). Neg-liver DNA from an uninfected mouse); (B) PCR amplification of 280 bp product (arrow) of MCMV IE DNA (data presented is from pooled liver DNA of 3 mice); (C) Virus titers at different time points after infection (calculated as log10 PFU/0.1 g tissue, data represent the average of 5 experiments); (D) Histopathological examination of a liver section 3 days after infection. Note a focus of hepatocellular necrosis with acute inflammation (long arrow), and several inclusion bodies (IB, arrow heads), (H&E X420).
View Figure 1
From day 3 to day 7, the livers from MCMV infected mice were macroscopically either congested or yellowish in color. The histopathological examination showed typical signs of MCMV disease on day 1 after infection. They included foci of markedly enlarged hepatocytes (long arrow; Figure 1D), containing large typical eosinophilic intranuclear MCMV inclusion bodies (IB) surrounded by a clear halo (the "owl eye" appearance - arrow heads; Figure 1D). Occasionally, smaller granular cytoplasmic inclusions were also detected. As the infection progressed, days 3-5, numerous tiny foci of hepatocellular necrosis surrounded by a few granulocytes, lymphocytes and histiocytes were detected. By that time a substantial increase in the number of hepatocytes containing IB was also observed (Figure 1D). The morphologic changes in the liver subsided by day 7, when only rare foci of necrosis were detected, but no IB nor inflammatory infiltrates. Throughout these experiments very few apoptotic Kupffer cells, but no apoptosis related to the viral infected areas was seen.
MCMV disease in bone marrow
In contrast with the liver, high amounts of MCMV DNA were detected in the BM on day 1. The high amounts of MCMV-DNA in this organ were sustained until day 14 (Figure 2A), and by day 28, no MCMV-DNA was detectable. In contrast to the high levels of viral DNA, only very low levels of infectious virus was detected by plaque assay. Neither histological evidence of MCMV infection nor apoptosis could be detected at any time after inoculation.
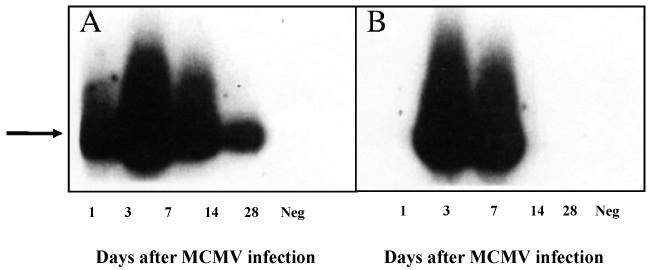
.
Figure 2: MCMV infection in bone marrow and inguinal lymph nodes. Groups of 6-8, eight-week-old Balb/c female mice were infected i.p. with 5 × 104 PFU of virus and were sacrificed on days1, 3, 7, 14, and 28 days. PCR amplification of 356 bp product (arrow) of MCMV gB DNA (data presented is from pooled DNA of 3 mice). Neg-liver DNA from an uninfected mouse). (A) Bone marrow; (B) Lymph node.
View Figure 2
MCMV disease in Lymph nodes
In this secondary lymphoid organ, significantly large amounts of MCMV DNA were detected from day 3 to 7 after infection. Unlike the BM, these amounts rapidly declined after day 7, and by day 14 no MCMV-DNA was detected (Figure 2B). By plaque assay, no infectious virus could be recovered from this organ at any time after inoculation.
Histological sections from lymph nodes on day 1 after infection, showed only primary lymphoid follicles, as lack of evidence of antigenic stimulation. This feature persisted until day 14, at which time we noted a reactive follicular hyperplasia with focal acute inflammation (data not shown).
The first signs of apoptosis became apparent on day 7, when apoptotic bodies were detected in the germinal centers of the lymph nodes of infected mice. On day 14, the number of apoptotic bodies increased, evidence that extensive apoptotic activity has taken place in this organ, as is usually the case in tingible bodies of germinal centers (data not shown).
MCMV disease in spleen
No MCMV-DNA was detected in this organ on day 1. Peak levels were detected in whole spleen homogenates on day 3, followed by a substantial decline by days 7-14. Following separation of the spleen into stromal and lymphoid cell fractions, similar amounts of MCMV DNA were detected in both cell fractions on day 3 after infection.
While in the stomal cell fraction, viral DNA persisted at least till day 14, a profound decrease in the amounts of viral DNA was detected in the lymphoid cell fraction by days 7 and 14 (Figure 3A).
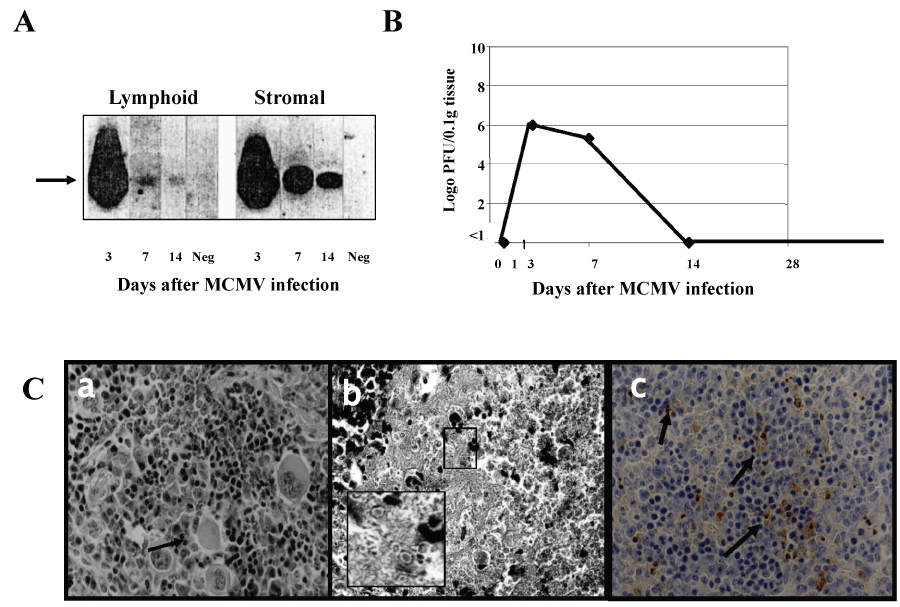
.
Figure 3: MCMV infection in spleen. Groups of 6-8, eight-week-old Balb/c female mice were infected i.p. with 5 × 104 PFU of virus and were sacrificed on days1, 3, 7, 14, and 28 days. (A) Splenic lymphoid and stromal cell fractions, prepared as described in M&M and subjected to PCR amplification of 356 bp product of MCMV gB DNA (data presented is from pooled DNA of 5 mice). Neg- spleen DNA from uninfected mouse; (B) Virus titers at different time points after infection (calculated as log10 pfu/0.1 g tissue, data represent the average of 5 experiments; (C) Histopathological examination of a spleen section at different time points after infection (a) a spleen section on day 5 after infection. Note the extramedullary hematopoiesis and poorly preserved large cells containing inclusion bodies in the red pulp (arrow heads) and hypoplasia of the white pulp (H&E X420); (b) a spleen section 7 days post infection shows residual white pulp surrounded by areas of hemorrhagic necrosis with poorly preserved inclusion bodies (inset) (H&EX320); (c) a spleen section 7 days post infection shows apoptotic cells dispersed in the residual white pulp and in the adjacent white pulp (arrow heads) (TUNEL assay X420).
View Figure 3
On day 3 after infection, virus titers of whole spleen homogenates were high (5 × 106 PFU/0.1 g). A gradual decline of these titers started on day 7, and no infectious virus could be recovered beyond day 14 (Figure 3B).
One of the hallmarks of acute MCMV infection in mice is splenomegaly followed by shrinkage and necrosis. By days 5-10 after infection the spleens of MCMV infected mice were considerably enlarged (mean weight 179 ± 10.8 mg), compared with those of uninfected animals (103.5 ± 5 mg). Spleens became soft with hyperemic cut surface, followed by necrosis and shrinkage.
Histologically, antigenic stimulation of the splenic white pulp was evident as early as on day 1 after infection. By day 3, the red pulp showed large mononuclear cells, possibly poorly preserved, hematopoietic cells with intranuclear IB (Figure 3C(a)). From that time and until day 7, the white pulp was hypoplastic to atrophic and surrounded by hemorrhagic necrosis and by isolated ghost cells with IB (Figure 3C(b)). By day 14, the white pulp was restored to normal and neither IB nor necrosis, were observed in the spleen. As expected apoptotic activity in the germinal centers of the white pulp was seen on day 3. This increased by day 7 to include hematopoietic cells of the red pulp (Figure 3C(c)). On day 14, apoptosis was prominent, mainly in the areas where extramedullary hematopoiesis of the red pulp occured.
MCMV disease in the lungs
In the lungs, low levels of MCMV-DNA were detected using both the more sensitive gB primer but also the IE primer. As the infection progressed, the amount of viral DNA reached a peak by day 7. This level of MCMV-DNA was maintained until day 14 and followed by a profound decrease thereafter. In this organ, MCMV-DNA was still detected by day 28 using the gB primer (Figure 4A), while titers of infectious virus that could be detected by the plaque assay were considerably lower than in liver and spleen (Figure 4 B and Table 1). Virus replication was detected from days 3 to 14 reaching a peak of only 1 × 102 PFU/0.1 g tissue on day 7, and which subsequently declined (Figure 4B).
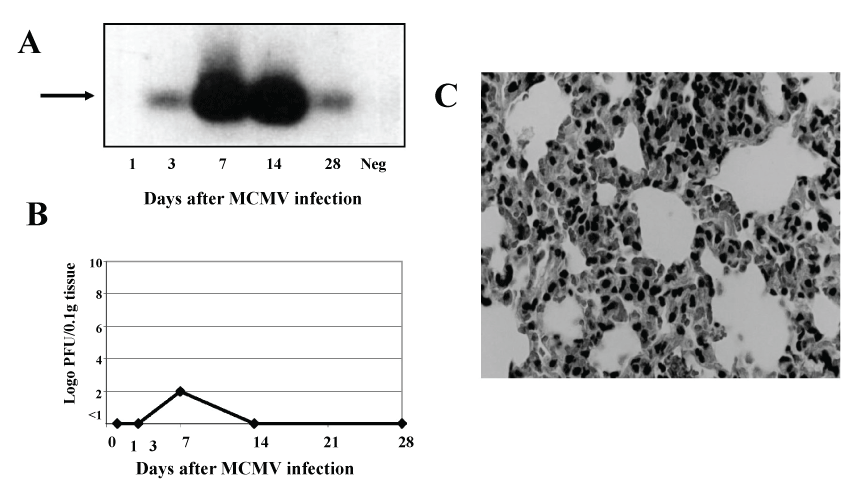
.
Figure 4: MCMV disease in lungs. Groups of 6-8, eight-week-old Balb/c female mice were infected i.p. with 5 × 104 PFU of virus and were sacrificed on days 1,3,7,14 and 28 days. (A) PCR amplification of 356bp product (arrow) of MCMV gB DNA (data presented is from pooled lung DNA of 3 mice). Neg-lung DNA from an uninfected mouse); (B) Virus titers at different time points after infection (calculated as log10pfu/0.1gr tissue, data represent the average of 5 experiments); (C) Histopathological examination of a lung section on day 7 after infection. Note the congestion, and interstitial chronic inflammatory infiltrate in the absence of inclusion bodies (H&E X320).
View Figure 4
Macroscopically, the appearance of MCMV infected lungs is normal at an early stage, they became slightly hyperemic and atelectatic as the infection progressed. On histology of the lungs of MCMV infected mice revealed mild to severe pulmonary congestion with areas of atelectasis as early as on days 1 and 3. At this time, no histological evidence of MCMV, such as IB or inflammation was observed. By day 7, small lymphocytic infiltrates surrounding the bronchi were evident (Figure 4C). This inflammatory process persisted until day 14 after which the histologic appearance of the lung became normal. No sign of apoptosis was evident at any time during the course of the infection.
MCMV disease in the Salivary glands
In contrast with all other organs examined, the SG showed evidence of virus infection starting as late as day 7. High levels of MCMV-DNA were detected and reached a peak on day 14. This level was maintained until day 28-30 using the gB primer (Figure 5A and Figure 5C). MCMV DNA could still be detected beyond that period in isolated mice only (Figure 5D).
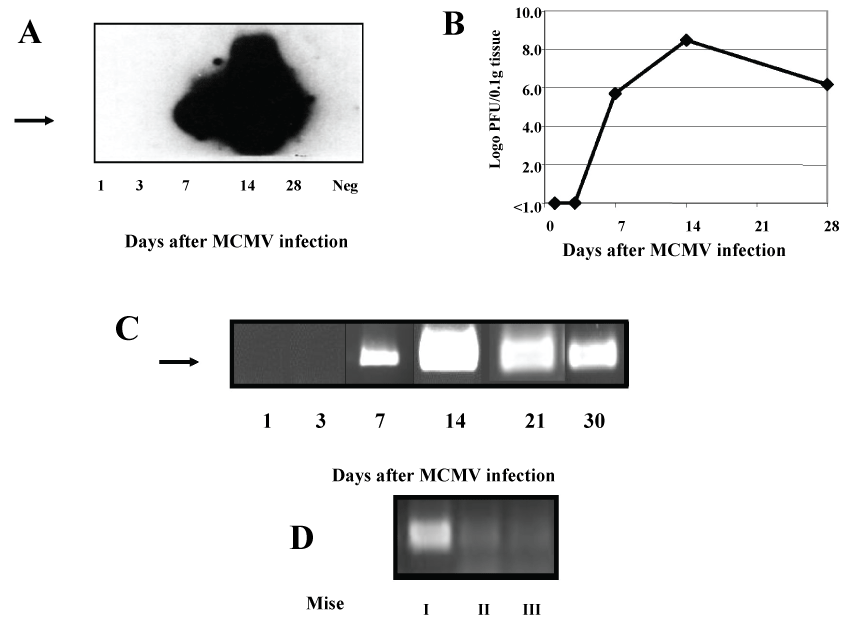
.
Figure 5: MCMV disease in SG. Groups of 6-8, eight-week-old Balb/c female mice were infected i.p. with 5 × 104 PFU of virus and were sacrificed on days1, 3, 7, 14, and 28 days. (A) PCR amplification of 356bp product (arrow) of MCMV gB DNA (data presented is from pooled SG DNA of 3 mice). Neg- salivary gland DNA from an uninfected mouse). (B) Virus titers at different time points after infection (calculated as log10pfu/0.1gr tissue, data represent the average of 5 experiments). (C) PCR amplification of 280 bp product (arrow) of MCMV IE DNA (data presented is from pooled SG DNA of 3 mice). (D) PCR amplification of 280 bp product (arrow) of MCMV IE DNA 42 days after infection (data presented is from SG DNA of 3 individual mice).
View Figure 5
Viral replication in this organ steadily increased from day 7, reaching peak titers of 3 × 108 PFU/0.1 g tissue by day 14 (Figure 5B). On day 28, 1.5 × 106 PFU/0.1 g of the virus were isolated. Upon histology, no apparent change of this organ was noted until days 7 or 14 after infection, when presence of intranuclear and more subtle, intracytoplasmic IB in serous acinar cells were detected. By day 14 a patchy severe chronic inflammation, composed mainly of small round lymphocytes surrounded an acinar epithelium containing numerous IB (Figure 6A and Figure 6B). This extensive chronic inflammation persisted through day 28, but the number of IB in the epithelial cells had decreased (Figure 6D). In serous acinar areas rich in IB, we focally noted oxyphilic granules in the acinar epithelium (Figure 6A and Figure 6D). Evidence of MCMV infection was nearly absent in the mucinous acinar areas. Throughout the different stages, a moderate amount of apoptotic cells was detected in stromal cells and in the lymphoid cells infiltrates (Figure 6C). Interestingly, no anatomic relation between the sites of apoptosis and the IB distribution was evident.
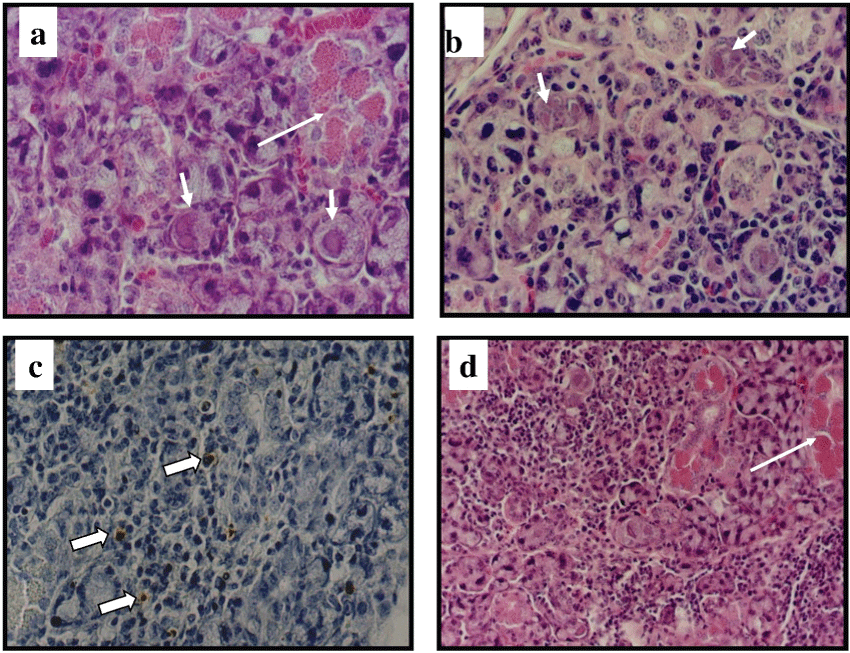
.
Figure 6: Histopathology of MCMV disease in SG.
Histopathological examination of a SG section 14 days after infection (A) note single or double inclusion bodies (short arrows) and oxyphilic changes in acinar cells (arrow) (H&E X420); (B) same section with inclusion bodies (arrow) and chronic inflammation (H&E X320); (C) SG on day 14 post infection, show apoptotic lymphocytes in the interstitium in the absence of inclusion bodies (black arrows) (TUNEL assay X320); (D) SG on day 28 post-infection, show persistent chronic inflammation and oxyphilic changes in acinar cells (arrows) (H&E X220).
View Figure 6
Table 1 exhibits the association between the peak MCMV replication and the culmination of MCMV-DNA levels in the various murine organs. Figure 7 is a flow chart which summarizes the organotropic kinetics of the CMV in the Balb/c mouse.

.
Figure 7: MCMV “Journey in the mouse”. Flow chart of virus dissemination in different organs after infection. Bars show the time period when MCMV DNA could be detected and the arrows show the time point in which peak infectious virus was found.
View Figure 7
![]()
Table 1: Peak infectious viral titer correlated to viral DNA PCR product*.
View Table 1
Discussion
Cytomegalovirus is one of the most ubiquitous of pathogens, imposing, in man a huge toll on health care organizations. This burden is mainly attributed to congenital infection and to opportunistic infections in immunocompromised hosts. This emphasizes the need for more potent anti-CMV drugs and for vaccines. In order to develop advanced therapeutic modalities, we have striven to expand our understanding of the organotropic and kinetic aspects of the MCMV infection. This has been done by establishing a data base on the dynamics of viral spread and more specifically, the time sequences and the organ-specific trafficking of the MCMV in the mouse.
Although ginea pig CMV has supplanted occasionally the MCMV as an animal model for HCMV, the murine virus has been the most extensively investigated model to date. Moreover, the guinea pig CMV model has mainly been studied in relation with congenital CMV disease and with the CMV-related hearing loss [6].
As often demonstrated, susceptibility of BALB/c mice to MCMV infection is a function of age, sex, viral dose, route of inoculation, immune status of the mouse and of the virus origin (tissue culture versus salivary gland-derived virus), although female mice have been classically regarded as more resistant than males to systemic viral infections, including that caused by MCMV, it has been suggested that the reverse may be more accurate [7,8].
We have examined systematically the dynamics of MCMV-organs interactions in MCMV salivary gland-derived viral infection of mice. Six to eight weeks-old mice were inoculated with 5 × 104 pfu of MCMV. All mice developed an acute sublethal transient infection, with typical clinical patterns followed by an apparent recovery.
We assayed viral DNA, infectious virus and examined the histopathological changes in the affected organs. Our results demonstrate that the virus behaves differently in each of the organs examined (Figure 7).
Within the first 24 hours of infection we were able to detect MCMV in liver and BM. These results indicate that both organs are the primary sites of dissemination. In the other organs such as spleen, LN and lungs, detectable amounts of DNA appeared, on day 3 of the infection. The last organ that was invaded by the virus was the SG (Figure 7).
In the liver, an accelerated replication occurred with a sharp peak followed by a rapid decline. The kinetics of MCMV-DNA expression and of the infectious virus titers occured in parallel (Table 1). Total viral clearance occurred by day 14. Morphological changes in immunocompetent mice included hepatocytes with typical inclusion bodies with an acute and chronic inflammatory reaction. The relatively short lived MCMV infection of the liver (day 1 to 14) emphasize the pivotal role of the inflammatory response in the eradication of MCMV infection from the liver of the immune competent animal.
Simultaneously, the bone marrow was submitted to an early stage invasion which lasted until day 14. In contrast with the significant amounts of viral DNA found in the BM, very low levels of infectious MCMV were found here (data not shown). This reflects possibly latency in this organ [1,15], which remained histologically normal at all times.
While very low levels of infectious virus were detected in the inguinal LN, the levels of MCMV DNA were relatively high. These results, similar to those found in the BM, are in contrast to those found by Cavanaugh et al. who showed high titers of infectious virus in the sub-maxillary gland draining lymph nodes. These nodes were probably infected during the virus clearing process from the salivary gland [16]. A variable effect on the LNs, depending on their anatomical location, may prevail.
High levels of MCMV-DNA were detected in whole spleen cell suspensions, with comparable levels of viral replication in this organ. MCMV replication peaked on day 3 and decreased thereafter. On histology, the spleen revealed the rapid onset of an inflammatory process. Inflammation peaked between days 3-5, when numerous activated lymphocytes were detected in the white pulp followed by hypoplasia and with poorly preserved large cells, possibly hematopoietic cells containing intranuclear IB in the red pulp. By day 14 the spleen resumed its normal appearance. Of note, when the spleen was fractionated to lymphoid and stromal cells subsets, viral DNA persisted longer in the stromal cells than in the lymphoid cells. The findings suggest that acute MCMV infection is cleared from the spleen, by effective inflammatory and adaptive immune responses, while a latent infection is established in the stromal cells. Clearing of the acute infection, followed by the development of latency have both been attributed to the lymphoid tissue stroma cells, as early as 1991 and have recently been related to type I interferon activity [15,17]. The thesis of bidirectional transmission of infectious virus between monocytes/macrophages and stromal cells (endothelial and littoral cells) is sustained by our experiment [15,18].
The lungs which demonstrated low DNA-MCMV levels detected on day 3 after infection, reached peak levels on days 7 and 14. Viral DNA was still detected in this organ by day 28. The levels of infectious virus were low, and by day 14, its complete clearance was evident. Latency is therefore suggested for the lungs as well [19,20]. To sustain this possible occurence, inflammation of the lungs was scarce on histology. MCMV shows a unique tropism to SG in mice. This organ has been extensively studied and has been implicated as the major site of MCMV replication and latency. Salivary glands are the last site to be invaded by the virus, as neither MCMV DNA nor infectious virus could be detected before day 7 after inoculation. However from this time onwards, a highly productive infection occurs. By day 14, virus replication reaches the peak value of 3 × 108 pfu/0.1 g tissue, the highest ever detected in any of the studied organs. This high level of replication was maintained at least until day 28 with abundant IB in the serous acinar cells together with moderate to severe chronic inflammation (Figure 6A and Figure 6B). Persistence of the virus in the SG is therefore suggested [21].
Unusual accumulation of oxyphilic granules was found on day 14 in areas of the SG serous acinar cells (Figure 6A and Figure 6B). This finding, together with severe chronic inflammation may be the consequence of stess and damage to the tissue, possibly inflicted by the high viral load present at that time in the SG.
MCMV dissemination in organs of BALB/c mice appears to take place in a very orderly manner. The appearance of the virus in the SG only by day 7 after infection is intriguing since the viral spread is hematogenous and reaches most organs by day 3 (Figure 7). Attempts to directly infect the SG by injecting MCMV via a SG ducts did not succeed in establishing of a direct infection of this organ [10]. In these experiments no virus could be recovered for several days after inoculation of the SG, although the virus did disseminate through the usual pathway and infected the spleen and liver on day 1-3 after infection.
The salivary glands might not become permissive to MCMV infection until day 7 of infection, after the inflammatory process in other organs has thrived [22]. We propose that invasion of the SG by free viruses or virus-infected mononuclear cells is delayed until MCMV induces systemic cytokines, which in turn elicit a break in through a putative “salivary gland-blood barrier”.
An alternative interpretation is that there is an obligatory period of “host maturation” due to both evoked host and viral chemokines and chemokine receptors and which results in gain of virulence by the virus [23-25]. The speculated need for virus-host maturation resembles that found with several other organisms which have a distinct life cycle in their hosts. Thus, some correlations between HCMV and MCMV pathogenetic pathways of dissemination may be found, acknowledging the limits of our understanding of the HCMV pathogenesis [21,26].
Our study is predominantly descriptive. Only if and when our analysis of the immune and molecular aspects of the related MCMV infection is completed, will we be able to establish the relevance of our present study to the development of new anti-HCMV therapy and vaccines. This primary level pathogenesis is also meant to confirm the latency and/or persistence of the MCMV infection, as they relate with several of the murine organs. This, in spite of the possible premature conclusion about latency, since the follow up in our study later rarely more than 28 days. It may, on the other hand, serve as a reference to eventual reparative changes induced by newly developed therapeutic agents and vaccines [27].
In conclusion, the pathogenesis of MCMV is dynamic, with a different profile, depending on the organ infected. This profile is influenced by the nature of the tissue invaded, its histological structure and the ability of individual cells to sustain a timely viral replication. Some organs reveal the presence of acute inflammation, with inclusion bodies or in their absence. In contrast, other organs show little inflammation, in spite of evidence of the replicating virus with or without viral DNA. Biological barriers and viral sanctuaries, as suggested in the SG, highlight the complex nature of the host-organism interactions, and the role of the microenvironment has a direct impact on the outcome, as seen in the spleen [28,29].
Acknowledgement
This study was supported in part by the Israel Ministry of Health, through the Office of the Chief Scientist and by Kibbutz Sde Boker, Israel.
Conflict of Interest
No conflict of interest exists.
References
-
Yuhasz SA, Dissette VB, Cook ML, Stevens JG (1994) Murine cytomegalovirus is present in both chronic active and latent states in persistently infected mice. Virology 202: 272-280.
-
Schrier RD, Nelson JA, Oldstone MB (1985) Detection of human cytomegalovirus in peripheral blood lymphocytes in a natural infection. Science 230: 1048-1051.
-
Sinzger C, Jahn G (1996) Human cytomegalovirus cell tropism and pathogenesis. Intervirology 39: 302-319.
-
Jean Beltran PM, Cristea IM (2014) The life cycle and pathogenesis of human cytomegalovirus infection: lessons from proteomics. Expert Rev Proteomics 11: 697-711.
-
Griffiths P, Baraniak I, Reeves M (2015) The pathogenesis of human cytomegalovirus. J Pathol 235: 288-297.
-
Schleiss MR (2013) Developing a Vaccine against Congenital Cytomegalovirus (CMV) Infection: What Have We Learned from Animal Models? Where Should We Go Next? Future Virol 8: 1161-1182.
-
Krmpotic A, Bubic I, Polic B, Lucin P, Jonjic S (2003) Pathogenesis of murine cytomegalovirus infection. Microbes Infect 5: 1263-1277.
-
Geurs TL, Hill EB, Lippold DM, French AR (2012) Sex differences in murine susceptibility to systemic viral infections. J Autoimmun 38: J245-253.
-
Sinzger C, Plachter B, Grefte A, The TH, Jahn G (1996) Tissue macrophages are infected by human cytomegalovirus in vivo. J Infect Dis 173: 240-245.
-
Palmon A, Blagerman S, Tel-Or S, Pecht M, Trainin N, et al. (1996) Treatment of murine cytomegalovirus salivary-gland infection by combined therapy with ganciclovir and thymic humoral factor gamma 2. Antiviral Res 33: 55-64.
-
Rager-Zisman B, Merigan TC (1973) A useful quantitative semimicromethod for viral plaque assay. Proc Soc Exp Biol Med 142: 1174-1179.
-
Rapp M, Messerle M, Bühler B, Tannheimer M, Keil GM, et al. (1992) Identification of the murine cytomegalovirus glycoprotein B gene and its expression by recombinant vaccinia virus. J Virol 66: 4399-4406.
-
Busche A, Marquardt A, Bleich A, Ghazal P, Angulo A, et al. (2009) The mouse cytomegalovirus immediate-early 1 gene is not required for establishment of latency or for reactivation in the lungs. J Virol 83: 4030-4038.
-
Mercer JA, Wiley CA, Spector DH (1988) Pathogenesis of murine cytomegalovirus infection: identification of infected cells in the spleen during acute and latent infections. J Virol 62: 987-997.
-
Koffron AJ, Hummel M, Patterson BK, Yan S, Kaufman DB, et al. (1998) Cellular localization of latent murine cytomegalovirus. J Virol 72: 95-103.
-
Cavanaugh VJ, Deng Y, Birkenbach MP, Slater JS, Campbell AE (2003) Vigorous innate and virus-specific cytotoxic T-lymphocyte responses to murine cytomegalovirus in the submaxillary salivary gland. J Virol 77: 1703-1717.
-
Verma, S, Wang Q, Chodaczek G, Benedict CA (2013) Lymphoid-tissue stromal cells coordinate innate defense to cytomegalovirus. J Virol 87: 6201-10.
-
Waldman WJ, Knight DA, Huang EH, Sedmak DD (1995) Bidirectional transmission of infectious cytomegalovirus between monocytes and vascular endothelial cells: an in vitro model. J Infect Dis 171: 263-272.
-
Kurz SK, Rapp M, Steffens HP, Grzimek NK, Schmalz S, et al. (1999) Focal transcriptional activity of murine cytomegalovirus during latency in the lungs. J Virol 73: 482-494.
-
Kurz SK, Reddehase MJ (1999) Patchwork pattern of transcriptional reactivation in the lungs indicates sequential checkpoints in the transition from murine cytomegalovirus latency to recurrence. J Virol 73: 8612-8622.
-
Goodrum F, Caviness K, Zagallo P (2012) Human cytomegalovirus persistence. Cell Microbiol 14: 644-655.
-
Campbell AE, Cavanaugh VJ, Slater JS (2008) The salivary glands as a privileged site of cytomegalovirus immune evasion and persistence. Med Microbiol Immunol 197: 205-213.
-
Ghazal P, Messerle M, Osborn K, Angulo A (2003) An essential role of the enhancer for murine cytomegalovirus in vivo growth and pathogenesis. J Virol 77: 3217-3228.
-
Bubic I, Wagner M, Krmpotic A, Saulig T, Kim S, et al. (2004) Gain of virulence caused by loss of a gene in murine cytomegalovirus. J Virol 78: 7536-7544.
-
Scalzo AA, Corbett AJ, Rawlinson WD, Scott GM, Degli-Esposti MA (2007) The interplay between host and viral factors in shaping the outcome of cytomegalovirus infection. Immunol Cell Biol 85: 46-54.
-
Sacher T, Mohr CA, Weyn A, Schlichting C, Koszinowski UH, et al. (2012) The role of cell types in cytomegalovirus infection in vivo. Eur J Cell Biol 91: 70-77.
-
Sissons JG, Wills MR (2015) How understanding immunology contributes to managing CMV disease in immunosuppressed patients: now and in future. Med Microbiol Immunol 204: 307-316.
-
Doherty PC (1995) Anatomical environment as a determinant in viral immunity. J Immunol 155: 1023-1027.
-
Recher M, Lang KS, Navarini A, Hunziker L, Lang PA, et al. (2007) Extralymphatic virus sanctuaries as a consequence of potent T-cell activation. Nat Med 13: 1316-1323.





