International Journal of Virology and AIDS
Monocyte-Derived Dendritic Cells from Feline Immunodeficiency Virus Positive Cats are Productively Infected and Maintain CD8+ T Cell Stimulatory Capacity
Hood SF1, Thompson EM2, Akaronu NO3, Miller MM4 and Fogle JE5*
1Duke Human Vaccine Institute, Immunology and Virology Quality Assessment, USA
2Catalent Pharma Solutions, USA
3The Brody School of Medicine at East Carolina University, USA
4The Hamner Institutes of Health Sciences, USA
5Department of Population Health and Pathobiology, North Carolina State University, USA
*Corresponding author: Dr. Jonathan E. Fogle, Department of Population Health and Pathobiology, College of Veterinary Medicine, North Carolina State University, 1060 William Moore Drive, Raleigh, NC 27607, USA, Tel: 919-513-6248, Fax: 919-513-6464, E-mail: jefogle@ncsu.edu
Int J Virol AIDS, IJVA-2-007, (Volume 2, Issue 1), Research Article; ISSN: 2469-567X
Received: March 11, 2015 | Accepted: April 01, 2015 | Published: April 03, 2015
Citation: Hood SF, Thompson EM, Akaronu NO, Miller MM, Fogle JE (2015) Monocyte-Derived Dendritic Cells from Feline Immunodeficiency Virus Positive Cats are ProductivelyInfected and Maintain CD8+ T Cell Stimulatory Capacity. Int J Virol AIDS 2:007. 10.23937/2469-567X/1510007
Copyright: © 2015 Hood SF, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution,and reproduction in any medium, provided the original author and source are credited.
Abstract
Dendritic cells (DCs) have been utilized to enhance CD8+ T cell responses to pathogen-associated peptides for enhancement of vaccine efficacy. CD8+ T cells play a central role in the elimination of viruses during acute viral infection and control of viremia during chronic viral infection. For lentiviral infections such as HIV and FIV, dendritic cell vaccines may be useful for augmenting CD8+ T cell function. Therefore, we asked if monocyte-derived dendritic cells (moDCs) from FIV+ chronically infected cats maintained the ability to stimulate CD8+ T cells in the absence of exogenous antigen, when compared to uninfected controls. Using high speed cell sorting, monocytes were isolated from PBMCs based on forward versus side scatter gating. Cells were then cultured with IL-4 and GM-CSF over the course of 6 days, and LPS was added to stimulate maturation after the first 72 hours. We confirmed the identity and maturation status of cells by cytologic examination and FITC-dextran uptake. We then assessed differences in CD8+ T cell proliferation in the presence of monocytes / macrophages (M/M), immature dendritic cells (iDCs), and mature dendritic cells (mDCs). We demonstrated that CD8+ T cell proliferation is maintained in the presence of DCs from FIV+ cats and that production of inflammatory cytokines and chemokines by DCs from FIV+ cats is retained in vitro. Further, we confirmed the presence of virus in co-culture. These results suggest that moDCs from FIV-infected animals serve as viral reservoirs. More importantly, these results support our hypothesis that moDCs maintain the ability to stimulate CD8+ T cells during chronic lentiviral infection.
Keywords
Dendritic cells, CD8+ T Cells, Feline immunodeficiency virus.
Abbreviations
AIDS: Acquired Immunodeficiency Syndrome, APC: Antigen-Presenting Cell, CFSE: Carboxyfluorescein Succinimidyl Ester, DCs: Dendritic Cells, FIV: Feline Immunodeficiency Virus, GM-CSF: Granulocyte Macrophage Colony-Stimulating Factor, HIV: Human Immunodeficiency Virus, iDCs: Immature Dendritic Cells, LPS: Lipopolysaccharide, mDCs: Mature Dendritic Cells, moDC: Monocyte-Derived Dendritic Cell, MHC: Major Histocompatibility Complex, MM: Monocyte / Macrophage, PBMCs: Peripheral Blood Mononuclear Cells, qPCR: Quantitative Polymerase Chain Reaction, RPI: Relative Proliferative Index, SPF: Specific Pathogen Free, TCR: T cell Receptor
Introduction
DC presentation of lentiviral antigen to T cells is important to control infection. CD8+ T cells are especially critical for controlling viral replication, as this population eliminates virus-infected cells; therefore, DC-based immunotherapy has the potential to augment virus-specific CD8+ T cell responses [1,2]. DC therapy likely stimulates a more focused CD8+ T cell response, driving the expansion of antigen-specific lymphocyte populations [3,4]. HIV-1 infected patients on combination anti-retroviral therapy exhibit DCs capable of stimulating a potent T cell response and producing IL-12, IL-10, and β-chemokines [5,6]. They also demonstrate similar morphology to DCs from uninfected patients [5,6]. These factors suggest that DCs from chronically infected immune environments may remain functional. However, in contrast to these findings, other studies suggest that DCs from HIV infected patients have impaired ability to stimulate CD8+ T cell proliferation and that viral infection may inhibit DC maturation [6,7]. If DCs remain potent stimulators of T cell activation in the face of chronic lentiviral infection, monocyte-derived (moDCs) dendritic cells could provide an antigen-presenting source for augmenting T cell responses [7].
Using the FIV model for AIDS lentiviral infection, we compared proliferation of CD8+ T cells in the presence of monocyte/macrophages (M/M), immature dendritic cells (iDCs), and mature dendritic cells (mDCs) generated from peripheral blood mononuclear cells (PBMCs) of healthy and FIV-infected cats. Although CD8+ T cell function is altered in long-term lentiviral infection, we hypothesized that protracted immune stimulation during chronic infection heightens the ability of DCs to promote T cell function. Therefore, we asked if DCs from chronically infected FIV+ cats are able to stimulate CD8+ T cell proliferation.
Following protocols for in vitro DC derivation in other species, feline monocytes isolated and sorted from peripheral blood were cultured with a combination of granulocyte macrophage colony stimulating factor (GM-CSF) and IL-4 to generate DCs and with LPS to stimulate maturation [8-12]. The M/M, iDCs, and mDCs were co-cultured at a 1:1 ratio with autologous CD8+ T cells in the absence of exogenous antigen to assess differences in stimulation of CD8+ T cell proliferation in FIV+ cats and healthy control cats. Culture supernatant pro-inflammatory cytokine and chemokine concentration and copies per mL of virus were determined by multiplex analysis and qPCR, respectively.
After co-culture, CD8+ T cells from FIV+ cats exhibited greater proliferation than those from control cats in the presence of all three cell-types. Further, autologous M/M, iDCs, and mDCs maintained the ability to produce pro-inflammatory cytokines and chemokines in co-cultures of cells from FIV+ animals. Finally, we have demonstrated the presence of FIV viral RNA in co-cultures from FIV+ cats. CD8+ T cells cultured alone produced minimal or no FIV RNA, indicating that the M/M and moDCs were the source of virus. We propose that moDCs from FIV+ cats can serve as a viral reservoir providing an endogenous source of lentiviral antigen. More importantly, we have demonstrated that moDCs maintain the ability to stimulate CD8+ T cell proliferation in vitro.
Materials and Methods
Study animals
Specific pathogen free (SPF), 6 month-old, male cats were purchased from Liberty Research, Inc. (Waverly, NY). FIV+ animals were infected with the NCSU1 isolate of FIV. Each cat was administered 1X105 TCID50 intravenously of cell-free virus as previously described [13,14]. At the time of these studies, all cats had been infected for 1-3 years and were in the chronic stage of infection. During the course of these studies, some cats were euthanized for use in an unrelated study; therefore, the number of cats used in each experiment reported decreases somewhat. All procedures described in this manuscript were approved by the North Carolina State University Institutional Animal Care and Use Committee (Table 1).
![]()
Table 1: Number of animals used for each experiment
View Table 1
PBMC isolation and cell sorting
Whole blood was collected in heparinized containers by jugular venipuncture. Peripheral blood mononuclear cell isolation was performed on samples prior to staining for cell sorting. Cells were surface labeled with anti-CD14 (anti-CD14 FITC: Serotec MCA1568F), anti-CD5 (Clone 572, Feline anti-CD5 APC: Tompkins, NCSU clone) and anti-CD8 (Clone 357, Feline anti-CD8 PE: M. Tompkins) and purified into monocyte or CD8+ CD5+ T lymphocytes by high speed cell sorting (Moflo DakoCytomation ). Cells were gated based on forward versus side scatter characteristics. Anti-CD5 and anti-CD14 were used to confirm the monocyte gating strategy with >90% of the CD14+ cells inside and >90% of the CD5+ cells outside the gate (Figure 1). CD8+ T cells were sorted from a CD5+ population of lymphocytes.
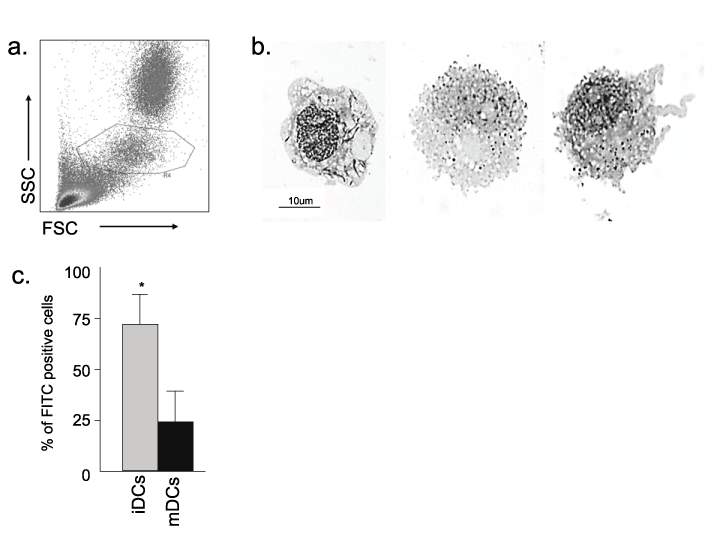
.
Figure 1: Morphologic, functional and phenotypic assessment of feline monocyte-derived dendritic cells
(a) Sorting gate for monocytes. Monocytes were gated by typical FSC vs. SSC characteristics. PBMCs were surface immunophenotyped with anti-CD14 and
anti-CD5 prior to sorting, to confirm the monocyte gate (R4, outlined) with most (~90%) of the CD14+ cells inside and most (~90%) of the CD5+ cells outside of
the gate. (b) Morphologic analysis of M/M, iDCs, and mDCs after culture. Cells were fixed, stained and analyzed by light microscopy (100x shown here). M/M
have well-defined cell edges (left). iDCs have increased vacuolation and less well-defined cell edges (middle). mDCs display advanced vacuolation and less
well-defined cell edges due to cytosolic projections (right). (c) Flow cytometric assessment of FITC dextran uptake by iDCs and mDCs. Consistent with the cell
morphology noted above, iDCs exhibit far more FITC dextran internalization than mDCs. Results are representative of four measurements for each condition
from two FIV+ cats (asterisks p< 0.05).
View Figure 1
Monocyte culturing and cell stimulus
Sorted monocytes or CD8+ T cells were cultured in RPMI medium containing 15% FBS, 4mM L-glutamine, 1% non-essential amino acids, 50�g/mL Gentamicin, 1% Penicillin Streptomycin, 2% HEPES, 1% Sodium pyruvate and 50μM β-mercaptoethanol. All cells were cultured at 2.5x106 cells per mL of culture media in a 48 well plate (1x106 cells in 400μL per well). CD8+ T cells were cultured with 100U/mL IL-2. After 24 hours, wells of monocytes were washed. Fresh media was added to control cells and media containing 1μg/mL IL-4 (Catalogue # 984-FL R&D Systems) and 1μg/mL GM-CSF (Catalogue # 987-FL R&D Systems) was added to remaining wells. After 48 additional hours, 50μL of media containing 1U/mL IL-2 was added to T cell wells. Fresh media was added to unstimulated monocyte wells. To stimulated monocyte wells, 1μg/mL IL-4 and 1ug/mL GM-CSF were added in 50μL of media along with 50μg/mL LPS to induce maturation. After 72 additional hours of culture, monocyte wells were either scraped in preparation for surface-phenotypic analysis, viability analysis, and cytologic examination (Romanowsky staining), or washed in preparation for co-culture without removing from the dish. T cells were removed from wells, counted, and set aside for CFSE staining and co-culture.
Dendritic cell characterization and viability assays
Monocytes, iDCs, and mDCs were surface immunophenotyped with anti-MHC II Biotin (Serotec MCA2723), anti-MHC I FITC (Washington State B072001/cell line H58A), anti-CD14 FITC (Serotec MCA1568F), anti-B7-1 PE (BD Pharmingen 553769) and anti-CD5 APC (572, Tompkins). For cell viability assays, samples were stained with Annexin V APC (BD Biosciences 550424) and Propidium Iodide (Roche Diagnostics 10969300). Samples were assessed via flow cytometry using a Becton Dickson FacsCalibur. Unstained samples were used to assess auto-fluorescence. For cell surface characterization, CD5+ T cells were gated out of the analysis.
FITC Dextran uptake by iDC and mDC populations was measured by flow cytometry. Samples from uninfected animals were differentiated from monocytes into iDCs and mDCs as previously described. The populations were treated with FITC Dextran (Molecular Probes) and incubated at 0�C for 60 minutes. The samples were assessed by flow cytometry using a Becton Dickson FacsCalibur. Flow cytometry gating was established using a control population not treated with FITC Dextran.
Cytologic examination
Cytologic staining was used to visually characterize M/M, iDC, and mDC populations at the completion of culture. Samples were centrifuged in a microcentrifuge prior to pipetting onto microscope slides. Romanowsky staining was used to assess cellular morphology. Cell morphology was confirmed by a veterinary clinical pathologist.
Autologous co-culture/Proliferation assay
All co-culture assays were performed with autologous cells as described below. CD8+ T lymphocytes were stained with CFSE using the Cell Trace CFSE Cell Proliferation Kit (Molecular Probes C34554) according to the manufacturer�s instructions. Dendritic cell wells were washed thoroughly with RPMI media without removing the cells. Autologous T cells were added to each co-culture well at a 1:1 ratio (40,000 cells added) with 400uL culture media, and 1U/mL IL-2. Controls included CD8+ T cells plated with 5 μg/mL ConA and 1U/mL IL-2 and CD8+ T cells plated with 1μg/mL IL-4, 1μg/mL GM-CSF and 1U/mL IL-2. Cells without CFSE staining were maintained in culture to assess background auto-fluorescence. Samples were incubated at 37�C for 96 hours. Cells were gently scraped from wells and accessed via flow cytometry (Becton Dickson FacsCalibur). Proliferation was assessed via ModFit LT software. The CFSE positive population was gated to remove antigen-presenting cells from the analysis. Control groups were used to establish the position of the cells that have not divided (parent peak) before assessing the proliferative properties of experimental groups.
Multiplex analysis of DC/CD8+ T cell co-cultures
Supernatants from co-cultures of CD8+ T cells and mDCs were thawed from -80oC on ice and treated with beads according to the manufacturer�s instructions (Milliplex Cat#SPRFCYTOPMX19). Samples were plated in duplicate in a 96 well plate and were read by Magpix Luminex Xmap multiplexing platform using Milliplex Analyte software. Supernatants from co-cultures of cells from infected and uninfected animals were both assessed (for each group n=2).
Determination of viral RNA levels in DC/CD8+ T cell co-cultures by qPCR
Supernatants from co-cultures of populations of cells from infected (n=5) and uninfected animals (n=7) and from cultures of CD8+ T cells alone (n=2) were thawed from -80oC. RNA was harvested from culture supernatants; reverse transcribed, and copies per mL of culture media assessed as described previously.
Statistical analysis
A Student�s T-test was used for pair-wise comparisons of relative proliferative indices and for comparisons of FITC Dextran uptake and number of FIV viral copies in culture supernatants. Differences were considered to be significant at p< 0.05. Relative proliferative indices were determined by dividing sample proliferative indices by those of the control. ModFit software (Verity Software, Topsham, ME) was used to analyze CD8+ T cell proliferation. The proliferative index is defined as the total number of divisions divided by the number of cells that went into division; cells that did not replicate once were not included in the analysis [15]. For analysis of viral copies in culture supernatants, a one-way ANOVA plus Tukey�s test was used to compare the means from each group with p< 0.05 considered significant.
Results
Morphologic, functional, and phenotypic analysis
Methods for generating feline moDCs have been previously described [16,17]. For the studies here, immature dendritic cells were generated by treating sorted monocytes with IL-4 and GM-CSF. Mature dendritic cells were then generated from iDCs by stimulating with LPS. As shown in figure 1a, monocytes were sorted from PBMCs using forward vs. side scatter gating. Residual lymphocytes were removed by washing of non-adherent cells on day 2, leaving a relatively pure M/M culture.
Cell morphology was characterized by cytologic examination. Figure 1b demonstrates the cell morphology for M/M (left), iDCs (middle) and mDCs (right) which is consistent with those described previously [16,17]. M/M exhibit crisp distinct borders, iDCs form cytosolic projections, and mDCs contain increased numbers of vacuoles and more extensive cytosolic projections. FITC Dextran uptake by iDC and mDC populations was assessed by flow cytometry. iDCs are reported to be more efficient at antigen uptake before differentiating into mDCs [1,5]. Consistent with our morphologic observations, figure 1c demonstrates greater uptake of FITC Dextran by iDCs than mDCs. Others have described changes in surface expression of CD14, MHC I, MHC II and B7.1 in mDCs during in vitro maturation [16,17]. Based upon these reports and the morphologic and functional studies described here, we asked if there were differences in expression of these surface receptors during in vitro maturation. There was a high degree of variability in surface receptor expression and no significant differences were identified between noninfected control cats and FIV+ cats. Therefore, data from noninfected controls and FIV-infected cats were combined for comparison of surface marker expression between M/M, iDCs, and mDCs. As shown in figure 2, there was a trend toward increased CD14 expression and decreased CD80 (B7.1) expression as maturation progressed. However, due to the variability of expression between the M/M, iDCs, and mDCs, neither of these trends reached significance. Both MHC I and MHC II expression appeared consistent between the groups.
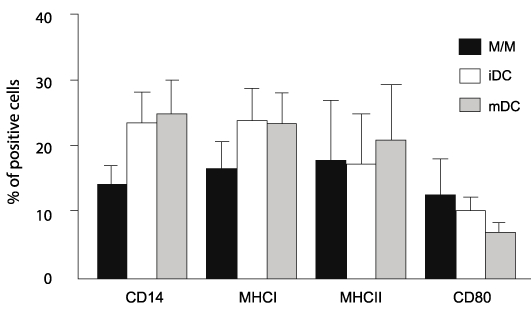
.
Figure 2: M/M, iDCs, and mDCs exhibit similar surface phenotype following
in vitro culture
Following coculture, eac h group of cells was analyzed by flow cytometry. No
significant difference in CD14, MHCI, MHCII, and CD80 expression was noted
between cell types. FIV+ and FIV- cats are grouped together as no significant
differences were observed between the two (n=13). Error bars represent +
SEM for each surface marker.
View Figure 2
Autologous co-culture
After confirming that M/M, iDCs, and mDCs exhibited morphological traits consistent with the desired cell types, we asked if DCs from chronically infected FIV+ cats retained the ability to stimulate CD8+ T cell proliferation, when compared to DCs from uninfected control cats. Aside from LPS to promote DC maturation, there was no additional exogenous antigenic or mitogenic stimulation of DCs. We hypothesized that DCs from FIV+ cats possessed heightened stimulatory capacity as a result of chronic immune activation. Further, we asked if M/M, iDCs, and mDCs served as a source of endogenous virus. CD8+ T cells were isolated from FIV+ or control cats and labeled with CFSE. Labeled CD8+ T cells were then cultured at a 1:1 ratio with autologous M/M, iDCs or mDCs. After 96 hours, CD8+ T cell proliferation was assessed by flow cytometry.
As described in figure 3, M/M, iDCs, and mDCs from FIV+ cats stimulated approximately 50% greater CD8+ T cell proliferation when compared to uninfected control cats. As a positive control for CD8+ T cell proliferation, CD8+ T cells were cultured alone in the presence of IL-2 and ConA. As a control for DC culture conditions, CD8+ T cells were also treated with IL2, IL4, and GM-CSF and did not exhibit significant proliferation (not shown).
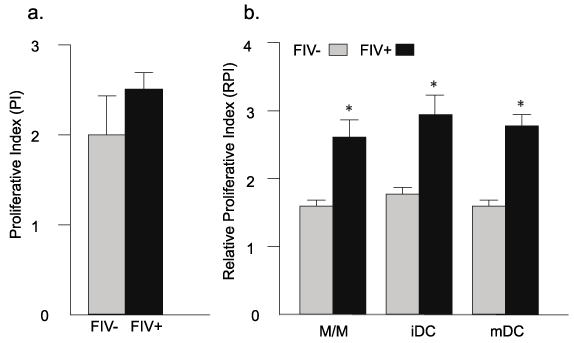
.
Figure 3: CD8+ T cells from FIV+ cats exhibit greater proliferation than CD8+
T cells from healthy controls following co-culture with autologous M/M, iDCs,
and mDCs
(a) CD8+ T cells from FIV+ and FIV- cats were cultured in the presence of
ConA and IL-2 (positive control, CTRL). Although the mean proliferative
index (PI) was somewhat different, it did not reach significance. (b) For each
experimental group (M/M, iDC, mDC), CD8+ T cells from FIV+ cats (black
bars) exhibit approximately 50% greater proliferation than CD8+ T cells
from control cats (gray bars). (n=6 FIV- cats and n=5 FIV+ cats). Relative
proliferative indices (RPI) were determined by dividing sample proliferative
indices by those of the control. Error bars represent + SEM for each group,
p< 0.05 (asterisks).
View Figure 3
Because monocyte-derived APCs from FIV+ cats retained the ability to stimulate CD8+ T cell proliferation, we asked if this may be due, in part, to the production of stimulatory cytokines and chemokines. Figure 4 demonstrates that there was a trend towards increased concentrations of IL-12 p40 and TNFα, CXCL1 and CCL5 in mDC co-cultures from FIV+ cats. Collectively, these data support our hypothesis that DCs remain functional during chronic FIV infection and suggest that monocyte-derived antigen presenting cells from FIV+ cats exhibit heightened CD8+ stimulatory ability.
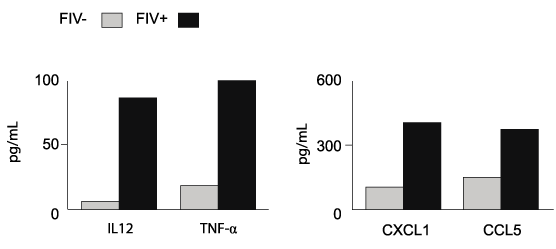
.
Figure 4: mDCs from FIV+ cats maintain the ability to produce proinflammatory
cytokines and chemokines
Cytokines were measured in culture supernatants using a multiplex cytokine
assay (see methods section). There was a trend towards increased
concentrations of pro-inflammatory cytokines (IL-12 p40 and TNF-alpha) and
chemokines (CXCL1 and CCL5) in co-cultures of mDCs and CD8+ T cells
from FIV+ cats (n=2) compared to those from FIV- cats (n=2). Results from CD8+ T
cells in culture alone were at the lower limit or below limit of detection.
View Figure 4
Determination of viral RNA levels in DC/CD8+ T cell co-cultures by qPCR
The data reported in figure 2 suggests there was no difference in the expression of costimulatory molecules between FIV+ cats and control cats, while figure 4 suggests that mDCs from FIV+ cats produce stimulatory chemokines and cytokines to induce proliferation. However, in the absence of exogenous antigen, we asked if this was a direct result of production of these stimulatory molecules or if the M/M, iDCs, and mDCs from infected cats might be a source of viral antigen, which would enhance proliferation of virus-specific T cells in culture. To answer this question, we quantified FIV in culture supernatants. Figure 5 demonstrates that FIV was present in all co-culture supernatants from FIV+ cats, but not in CD8+ T cells cultured alone, and that iDCs supported a productive viral infection approximately three times greater than that of M/M or mDCs. Collectively, these observations suggest that M/M, iDCs, and mDCs are the source of virus in culture. Further, these results suggest that heightened proliferation of CD8+ T cells from FIV-infected animals is multifactorial, resulting from both increased cytokine and chemokine production by M/M, iDCs, and mDCs and the processing and presentation of FIV antigen.
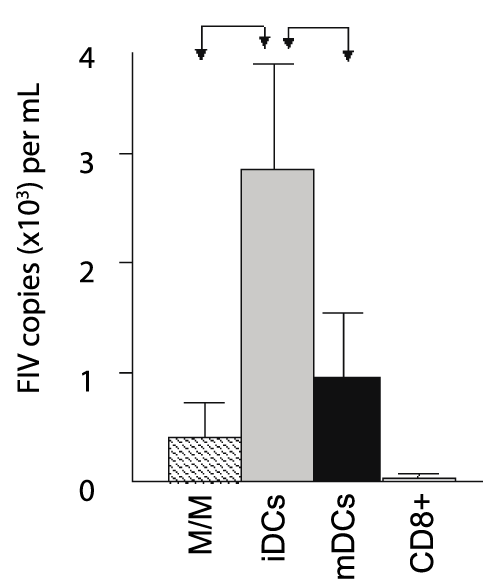
.
Figure 5: M/M, iDCs, and mDCs from FIV+ cats support a productive infection
following in vitro culture and differentiation
The number of copies per ml in culture supernatants was assessed by PCR
following co-culture of CD8+ T cells with autologous M/M, iDCs, and mDCs
from FIV+ cats. All groups produced virus in culture supernatants; however
iDCs supported the highest production of virus (gray bar) when compared
to M/M (hatched bar) and mDCs (black bar). CD8+ T cells from FIV+ cats
cultured alone produced almost no virus in culture supernatants (white bar).
Error bars represent + SEM from 5 FIV+ cats for each group (M/M, iDCs,
mDCs, p< 0.05, arrows).No virus was detected in culture supernatants from
FIV- cats (n=7, not shown).
View Figure 5
Discussion
As described in figure 1, we sorted monocytes from peripheral blood mononuclear cells (PBMCs) for differentiation into immature and mature dendritic cell populations using IL-4 and GM-CSF for the former and IL-4, GM-CSF, and LPS for the latter. The identity and characteristics of these populations were confirmed by FITC dextran uptake and cytology. As shown in figure 3, when M/M, iDCs, and mDCs from healthy control cats were compared to each other or when M/M, iDCs, and mDCs from FIV+ cats were compared to each other, there was no difference in the ability of each respective cell type to stimulate CD8+ T cell proliferation. This observation is interesting, as immature dendritic cells are reputed to have a lower cell surface concentration of MHC complexes and co-stimulatory molecules necessary for stimulation [19]. However, as demonstrated in figures 2 and 3, morphologic and phenotypic analysis suggests that in vitro cell maturation exists as a continuum. Distinguishing between the three stages of maturation outlined here is difficult due to intermediary phenotypes and this overlap between groups could account for the lack of significance in comparisons of surface molecule expression or stimulatory ability [19]. Further, the estimates for MHC I and MHC II expression were conservative because we elected to utilize a higher positive threshold based upon the degree of background auto-fluorescence in cultured cells. Recently, Huang et al have demonstrated that HIV-1 infected patients on combination anti-retroviral therapy maintain DCs capable of stimulating a potent T cell response [5]. Consistent with this, the data in figure 3 demonstrates that following co-culture with M/M, iDCs, and mDCs, there is increased CD8+ T cell proliferation in FIV+ cats when compared to healthy control cats. These findings support our hypothesis that DCs from FIV+ cats maintain their ability to stimulate CD8+ T cell proliferation during the course of lentiviral infection.
Based upon these observations, we asked if DCs from FIV+ cats retained the ability to produce inflammatory signals that promote CD8+ T cell proliferation. Figure 4 shows that mDCs from FIV+ cats retained the ability to produce IL12, TNFα, CXCL1, and CCL5, and concentrations in the culture supernatants were approximately twice that of healthy control cats. Similar to our findings, Chougnet et al. reported that DCs from HIV-infected patients retained the ability production of IL-12 or IL-10 and exhibited similar morphologic features when compared to healthy controls [5,6]. Collectively, these findings suggest mDCs from FIV+ cats exhibit a heightened ability to produce inflammatory cytokines and chemokines. More importantly, these data clearly answer the original question, demonstrating that mDCs from FIV+ cats retain the functional ability to stimulate CD8+ T cell proliferation.
As described in the methods, the M/M cultures were washed to remove non-adherent cells. While we anticipated a small amount of virus might be produced during in vitro culture, the level of virus detected in our assays was higher than expected. Similar studies from HIV-infected patients suggest mDCs do not harbor significant levels of virus [11]. However, other studies suggest that monocytes, particularly CD16+ monocytes, may serve as a viral reservoir and source of productive infection following monocyte differentiation [20-23]. Interestingly, Freer et al. previously reported that feline CD14+ monocytes exhibited poor ability to differentiate into dendritic cells in vitro [16]. As shown in figure 1, monocytes were isolated by forward vs. side light scatter characteristics; therefore, all monocyte subsets were included in our experiments. The presence of FIV in co-culture supernatants of CD8+ T cells with autologous M/M, iDCs, and mDCs from FIV+ cats but not in cultures of CD8+ T cells alone indicates that the monocytes isolated for use in this study served as a viral reservoir. Collectively, these data suggest that certain monocyte subsets, such as CD14lo or CD14- subset were the source of in vitro virus production following differentiation and likely provided FIV antigens for MHC-processing, and CD8+ T cell stimulation. Our original design was to compare DC �basal� stimulatory activity of CD8+ T cells, without adding antigen. Although it is a confounding factor for these experiments, the antigen-specific population still represents a small fraction of the total cell population within the DC culture. It is clear that DCs from FIV+ cats maintain CD8+ stimulatory ability.
Heightening DC antigen presentation holds promise for enhanced T cell stimulation [24]. Based upon this, different investigations have utilized DCs pulsed with antigen-specific peptides to amplify T cell responses [25,26]. However, DC immunotherapy trials for protection against lentiviral challenge have demonstrated mixed results, ranging from little measurable T cell responses or no reduction in viral loads in some studies, to promotion of T cell proliferation and reduction in viral load in other studies [4,5,27-33]. Collectively, these results underscore the need for identifying novel methods to enhance cross-presentation of exogenous antigen onto MHC class I molecules, which in turn will boost the stimulatory ability of DCs for vaccine and immunotherapy applications.
The FIV vaccine has demonstrated robust protection from virus challenge [34,35]. Using the FIV model for AIDS lentivirus infection offers a unique opportunity to identify key events contributing to vaccine success and to identify novel methods for enhancing cellular immune responses. While methods for loading DCs with antigens were not a focus of this manuscript, DC-based immunotherapy may yet hold promise for the optimization of antigen presentation. Using the well-characterized FIV lentiviral model, ongoing studies in our laboratory are aimed at enhancing cross presentation to stimulate CD8+ T cell immunity that exceeds that for the already successful FIV vaccine. The findings reported here indicate that M/M, iDCs and mDCs from FIV+ cats are productively infected and are a source of viral antigen and more importantly, that even during the course of chronic FIV infection, DCs maintain the capacity to produce stimulatory molecules and promote CD8+ T cell proliferation.
Acknowledgements
The authors would like to acknowledge Dr. Tayn Sibley and Dr. Jaime Tarigo for their assistance with cytologic identification and photographing of the various cell types. The authors would like to thank Deb Anderson, Linda English, and Janet Dow for their excellent technical assistance. The authors thank Dr. Kristen Messenger for her assistance with the feline multiplex assay.
This study was supported in part by National Institute of Health grants AI080288, AI074445, AI027757, and 5R25GM083242. Additionally, this research was supported in part by the Creative and Novel Ideas in HIV Research Program (CNIHR) through a supplement to the University of Alabama at Birmingham (UAB) Center for AIDS Research funding (P30 AI027767-24). CNIHR funding was made possible by collaborative efforts of the Office of AIDS Research, the National Institutes of Allergies and Infectious Diseases, and the International AIDS Society.
References
-
Bhardwaj N (2001) Processing and presentation of antigens by dendritic cells: implications for vaccines. Trends Mol Med 7: 388-394
-
Kato M, Igarashi H, Takeda A, Horie S, Higashihara E, et al. (2004) Stimulation of virus-specific T cell responses by dendritic cell vaccination in the chronic phase of simian AIDS models. Jpn J Infect Dis 57: 220-223
-
Letvin N L (2005) Progress toward an HIV vaccine. Annu Rev Med 56: 213-223
-
Goulder PJ, Watkins DI (2004) HIV and SIV CTL escape: implications for vaccine design. Nat Rev Immunol 4: 630-640
-
Huang XL, Fan Z, Colleton BA, Buchli R, Li H, et al. (2005) Processing and presentation of exogenous HLA class I peptides by dendritic cells from human immunodeficiency virus type 1-infected persons. J Virol 79: 3052-3062
-
Chougnet C, Cohen SS, Kawamura T, Landay AL, Kessler HA, et al. (1999) Normal immune function of monocyte-derived dendritic cells from HIV-infected individuals: implications for immunotherapy. J Immunol 163: 1666-1673
-
Altfeld M, Fadda L, Frleta D, Bhardwaj N (2001) Dcs and NK cells: critical effectors in the immune response to HIV-1. Nat Rev Immunol 11: 176-186
-
Heath WR, Belz GT, Behrens GM, Smith CM, Forehan SP, et al. (2004) Cross-presentation, dendritic cell subsets, and the generation of immunity to cellular antigens. Immunol Rev 199: 9-26
-
Mehlhop E, Villamide LA, Frank I, Gettie A, Santisteban C, et al. (2002) Enhanced in vitro stimulation of rhesus macaque dendritic cell activation of SIV-specific T cell responses. J Immunol Methods 260: 219-234
-
Inaba K, Schuler G, Witmer MD, Valinksy J, Atassi B, et al. (1986) Immunologic properties of purified epidermal Langerhans cells. J Exp Med 164: 605-613
-
Sapp M, Engelmayer J, Larsson M, Granelli-Piperno A, Steinman R, et al. (1999) Dendritic cells generated from blood monocytes of HIV-1 pateints are not infected and act as competent antigen presenting cells eliciting potent T-cell responses. Immunol Lett 66: 121-128
-
Pickl WF, Majdic O, Kohl P, St�ckl J, Riedl E, et al. (1996) Molecular and functional characteristics of dendritic cells generated from highly purified CD14+ peripheral blood monocytes. J Immunol 157: 3850-3859
-
Davidson MG, Rottman JB, English RV, Lappin MR, Tompkins MB (1993) Feline immunodeficiency virus predisposes cats to acute generalized toxoplasmosis. Am J Pathol 143: 1486-1497
-
Miller MM, Thompson EM, Suter SE, Fogle JE (2013) CD8+ clonality is associated with prolonged acute viremia and altered mRNA cytokine profiles during the course of Feline Immunodeficiency Virus infection. Vet Immunol Immunopathol 152: 200-208
-
Givan AL, Fisher JL, Waugh MG, Bercovici N, Wallace PK (2004) Use of cell-tracking dyes to determine proliferation precursor frequencies of antigen-specific T cells. Methods Mol Biol 263: 109-124
-
Freer G, Matteucci D, Mazzetti P, Bozzacco L, Bendinelli M ( 2005) Generation of feline dendritic cells derived from peripheral blood monocytes for in vivo use. Clin Diagn Lab Immunol 12: 1202-1208
-
Bienzle D, Reggeti F, Clark ME, Chow C (2003) Immunophenotype and functional properties of feline dendritic cells derived from blood and bone marrow. Vet Immunol Immunopathol 96: 19-30
-
Muul LM, Silvin C, James SP, Candotti F (2008) Measurement of proliferative responses of cultured lymphocytes. Curr Protoc Immunol 82: 1-7
-
Le�n B, L�pez-Bravo M, Ardav�n C (2005) Monocyte-derived dendritic cells. Semin Immunol 17: 313-318
-
Sonza S, Mutimer HP, Oelrichs R, Jardine D, Harvey K, et al. (2001) Monocytes harbour replication-competent, non-latent HIV-1 in patients on highly active antiretroviral therapy. AIDS 15: 17�22
-
Lambotte O, Taoufik Y, de Go�r MG, Wallon C, Goujard C, et al. (2000) Detection of infectious HIV in circulating monocytes from patients on prolonged highly active antiretroviral therapy. J Acquir Immune Defic Syndr 23: 114�119
-
Ellery PJ, Tippett E, Chiu YL, Paukovics G, Cameron PU, et al. (2007) The CD16+ monocyte subset is more permissive to infection and preferentially harbors HIV-1 in vivo. J Immunol 178: 6581-6589
-
Dong C, Kwas C, Wu L (2009) Transcriptional restriction of human immunodeficiency virus type 1 gene expression in undifferentiated primary monocytes. J Virol 83: 3518-3527
-
Tanchot C, Lemonnier FA, P�rarnau B, Freitas AA, Rocha B ( 1997) Differential requirements for survival and proliferation of CD8 na�ve or memory T cells. Science 276: 2057-2062
-
Weissman D, Fauci AS (1997) Role of dendritic cells in immunopathogenesis of human immunodeficiency virus infection. Clin Microbiol Rev 10: 358-367
-
Steinman R M, Pope M (2006) Exploiting dendritic cells to improve vaccine efficacy. J Clin Invest 109: 1519-1526
-
Lu W, Wu X, Lu Y, Guo W, Andrieu JM ( 2003) Therapeutic dendritic-cell vaccine for simian AIDS. Nat Med 9: 27-32
-
Freer G, Matteucci D, Mazzetti P, Tarabella F, Catalucci V, et al. ( 2008) Evaluation of Feline Monocyte-Derived Dendritic Cells Loaded with internally Inactivated Virus as a Vaccine against Feline Immunodeficiency Virus. Clin Vaccine Immunol 15: 452-459
-
Garc�a F, Climent N, Guardo AC, Gil C, Le�n A, et al. (2013) A dendritic cell-based vaccine elicits T cell responses associated with control of HIV-1 replication. Sci Transl Med 5: 166-174
-
Gandhi RT, O'Neill D, Bosch RJ, Chan ES, Bucy RP, et al. (2009) A randomized therapeutic vaccine trial of canarypox-HIV-pulsed dendritic cells vs. canarypox-HIV alone in HIV-1-infected patients on antiretroviral therapy. Vaccine 27: 6088-6094
-
Kloverpris H, Karlsson I, Bonde J, Thorn M, Vinner L, et al. (2009) Induction of novel CD8+ T cell repsonses during chronic untreated HIV-1 infection by immunization with subdominant cytotoxic T-lymphocyte epitopes. AIDS 23: 1329-1340
-
Allard SD, De Keersmaecker B, de Goede AL, Verschuren EJ, Koetsveld J, et al. (2012) A phase I/IIa immunotherapy trial of HIV-1-infected patients with Tat, Rev, and Nef expressing dendritic cells followed by treatment interruption. Clin Immunol 142: 252-268
-
Lu W, Arraes LC, Ferreira WT, Andrieu JM (2004) Therapeutic dendritic-cell vaccine for chronic HIV-1 infection. Nat Med 10: 1359-1365
-
Yamamoto JK, Pu R, Sato E, Hohdatsu T (2007) Feline immunodeficiency virus pathogenesis and development of a dual-subtype feline-immunodeficiency-virus vaccine. AIDS 21: 547-563
-
Coleman JK, Pu R, Martin M, Sato E, Yamamoto JK (2005) HIV-1 p24 vaccine protects cats against feline immunodeficiency virus infection. AIDS 19: 1457-1466.





