International Journal of Transplantation Research and Medicine
Influence of Metronidazole on Oral Busulfan Test Dose
Francine Attié de Castro1, Vera Lucia Lanchote1* and Belinda Pinto Simões2
1Departamento de Análises Clínicas, Toxicológicas e Bromatológicas, Faculdade de Ciências Farmacêuticas de Ribeirão Preto, Universidade de São Paulo, Ribeirão Preto, Brazil
2Departamento de Clínica Médica, Faculdade de Medicina de Ribeirão Preto, Universidade de São Paulo, Ribeirão Preto, Brazil
*Corresponding author:
Vera Lucia Lanchote, PhD, Departamento de Análises Clínicas, Toxicológicas eBromatológicas, Faculdade de Ciências Farmacêuticas de Ribeirão Preto-USP, Universidade de São Paulo, Avenida do Café, s/n, Campus da USP, 14040-903, Ribeirão Preto, São Paulo, Brazil, Tel: 55-16-3602-4195, Fax: 55-16-3602-4725, E-mail: lanchote@fcfrp.usp.br
Int J Transplant Res Med, IJTRM-2-019, (Volume 2, Issue 1), Case Report
Received: January 28, 2016: Accepted: July 05, 2016: Published: July 07, 2016
Citation: de Castro FA, Lanchote VL, Simões BP (2016) Influence of Metronidazole on Oral Busulfan Test Dose. Int J Transplant Res Med 2:019.
Copyright: © 2016 de Castro FA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Pharmacokinetics, Busulfan, Metronidazole, Oral test dose, Stem cell transplantation
Background and Objectives
Individualization of busulfan (BU) dosing during pre-transplant conditioning for hematopoietic stem cell transplantation (HSCT) has been performed worldwide for more than a decade. The pharmacokinetic parameters of BU show high variability and studies report that high plasma concentrations (> 900 ng/mL) are associated with sinusoidal obstructive syndrome (SOS), whereas low plasma concentrations (< 600 ng/mL) are associated with graft rejection and/or disease recurrence [1,2]. The high variability in BU pharmacokinetics can result in severe adverse reactions and can compromise the success of transplantation. One alternative used for the individualization of BU therapy is the application of a test dose days before the beginning of conditioning. On the basis of a small test dose of BU administered days before conditioning, drug clearance by the patient is calculated and the ideal dose to achieve the target plasma concentration is defined. The major disadvantage of this technique is that the BU test dose is administered prior to the use of different drugs present in the pre-HSCT conditioning regimen, such as antibiotics, glucocorticoids and anticonvulsants, among others [3-7].
Busulfan is metabolized by conjugation with glutathione which is mediated by glutathione-S-transferase, followed by oxidation catalyzed by CYP3A4 [2,4,5,8,9]. The main metabolites of BU are 3-hydroxysulfolane and tetrahydrothiophene 1-oxide, which do not significantly contribute to the efficacy or toxicity of the drug [10-12].
Several drugs that may have an impact on the clearance of BU have been described in the literature, including the antibiotic metronidazole which is used during pre-HSCT conditioning to prevent infections caused by Clostridium difficile and/or to avoid graft-versus-host disease [12-15]. We report a clear case report of drug-drug interaction between BU and oral metronidazole, in which the patient received two test doses of BU, one in combination with metronidazole and one without this drug.
Case Report
The study was approved by the Research Ethics Committee of the University Hospital of the School of Medicine of Ribeirao Preto (HCFMRP-USP) and the patient was only submitted to investigation after he had given free informed consent.
A 30-year-old male patient (73 kg, BMI: 24.9 kg/m2) with chronic myeloid leukemia was submitted to HSCT at the Bone Marrow Transplant Unit of HCFMRP-USP. On the third day of treatment with metronidazole (400 mg/8 h), the patient received an oral test dose of BU (0.25 mg/kg) (Myleran®, GlaxoSmithKline). Serial blood samples (volumes of 1 mL) were collected with heparinized syringes at times 0, 15, 30, 45, 60, 75, 90, 105, 120, 135, 150, 180, 240, 300, and 360 min after administration of the BU test dose. The plasma samples were immediately analyzed by LC-MS/MS [16] and the pharmacokinetic parameters were determined with the WiNonLin 5.2 software using a mono-compartmental model.
The clearance calculated based on the administration of the BU test dose combined with metronidazole was 10.3 L/h. The dose suggested to achieve Css within the target range of 600-900 ng/mL according to the equation dose/τ = Cl/f .Css, where τ corresponds to the 6-h dose interval, would be 37 to 55.6 mg/kg/6 h for 4 days. However, due to problems related to bone marrow donation, the patient did not start the pre-HSCT conditioning. Two months later, the same patient was rehospitalized for HSCT. A new test dose of BU (0.25 mg/kg) was administered; however, on this occasion, the test was performed before the use of metronidazole. On this second occasion, the apparent clearance value of the patient was 16.6 L/h and the ideal dose suggested was 60-90 mg/kg/6 h for 4 days. The patient was conditioned with an oral BU dose of 72 mg/kg administered at intervals of 6 h for 4 days.
After administration of the fifth BU dose during the conditioning regimen, serial blood samples were collected during the 6-h dose interval and immediately analyzed to determine whether the Css of BU was within the target range of 600-900 ng/mL. The AUC calculated for the 6-h dose interval using a non-compartmental model (steady state) was 4,516.5 ng.h/mL, corresponding to Css of 752.7 ng/mL.
Figure 1 shows the decay curves of plasma BU concentrations over time after administration of the test doses and of the fifth dose.
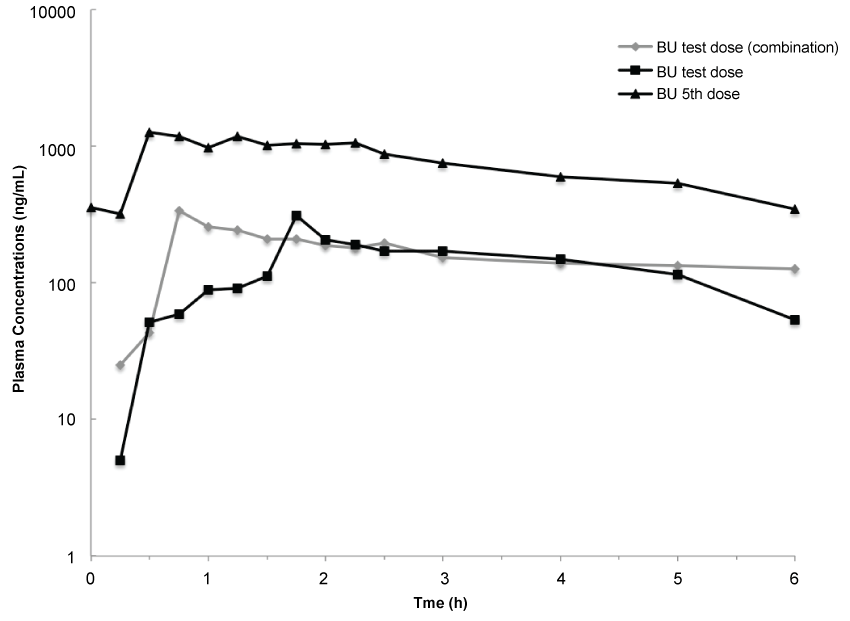
.
Figure 1: Plasma busulfan (BU) concentration versus time during the test dose (combined or not with metronidazole) and during the 5th BU dose.
View Figure 1
Discussion
The individualization of oral or intravenous BU therapy is a routine procedure at most HSCT centers. Some centers perform therapeutic monitoring on the first day of conditioning, whereas others use a test dose [3,4,17,18]. We found only two studies in the literature reporting BU - metronidazole interaction, one using oral administration and the other intravenous administration [6,12]. Nilsson et al. [12] compared the plasma concentrations of BU measured immediately before each dose (trough levels) between three groups of patients: group A received oral BU combined with metronidazole (400 mg/8 h), group B was treated with oral BU without metronidazole for 2 days and received the combination (400 mg/8 h) on the subsequent 2 days, and group C was only treated with BU and did not receive metronidazole. The authors observed a significant difference in plasma BU concentrations between groups A and C, an increase in plasma BU concentrations in group B when the patients began treatment with metronidazole, and marked hepatic alterations in patients receiving the combination. Gulbis et al. [6] reported the case of a 7-year-old child who received an intravenous test dose of BU (0.5 mg/kg) 7 days before HSTC and began treatment with oral metronidazole 6 days before HSTC. The authors observed a 46% reduction in clearance after the beginning of metronidazole treatment, which led to the discontinuation of the last BU dose in an attempt to prevent the occurrence of SOS. As a consequence, the patient did not develop SOS, but relapsed 3 months after HSCT.
The present study reports the case of an adult patient with chronic myeloid leukemia who received two oral test doses of BU, one combined with metronidazole (400 mg/8 h) and the other without the drug. A reduction in clearance of 38% was observed for the combination of oral BU and metronidazole. If this dosage had been continued, the patient would probably have presented low plasma concentrations of BU, a fact that could have resulted in recurrence of the disease and/or graft rejection. Since the same patient was enrolled, the possibility that this altered clearance was due to genetic changes, the influence of age, or even the influence of the underlying condition could be ruled out. Therefore, the new oral test dose of BU was crucial to the success of the treatment. Plasma BU concentrations were evaluated after the fifth BU dose and the results showed Css values within the target range. The patient was in clinical remission in the end of this study.
The mechanism whereby metronidazole interferes with the elimination of BU is still unclear and, although BU is metabolized through conjugation with glutathione, most drugs that interfere with the metabolism of BU are inducers or inhibitors of CYP enzymes that are involved in the elimination of intermediate metabolites of BU [19,20]. In vitro data have shown that metronidazole can inhibit CYP3A [21], however, recent in vivo data using high doses of metronidazole are contrary to these results [22,23]. The hypothesis of BU as a substrate of P-gp and of metronidazole as an inhibitor of P-gp was considered, but rapidly discarded since previous studies reported the influence of metronidazole on the metabolism of BU during both intravenous and oral administration [6,12]. Another possible and strong hypothesis is related to the inhibition of CYP2C9. Although the participation of CYP2C9 in the metabolism of BU has never been reported in the literature, according to the recent study by Uppunguduri et al. [24], patients genotyped as CYP2C9*2 and *3 show higher metabolic ratios (BU/3-hydroxysulfolane) than patients who did not carry these allelic variant, suggesting a relationship between CYP2C9 activity and BU metabolism. These results clarify the influence of metronidazole on the pharmacokinetics and metabolism of BU, since the former is a weak inhibitor of CYP2C9 in in vivo studies [25].
The main objective of the present case report was to alert HSCT centers that perform BU dose adjustments based on a test dose to observe whether metronidazole is administered during the test dose and/or during the conditioning period with BU in order to prevent a wrong dose prediction as a consequence of drug-drug interaction.
Acknowledgements
The authors thank Prof. Dr. Julio Cesar Voltarelli (in memoriam), Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for financial support, and the team of the Bone Marrow Transplant Unit of HCFMRP-USP.
References
-
McCune JS, Gooley T, Gibbs JP, Sanders JE, Petersdorf EW, et al. (2002) Busulfan concentration and graft rejection in pediatric patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant 30: 167-73.
-
McCune JS, Gibbs JP, Slattery JT (2000) Plasma concentration monitoring of busulfan: does it improve clinical outcome? Clin Pharmacokinet 39: 155-165.
-
Takamatsu Y, Sasaki N, Eto T, Nagafuji K, Abe Y, et al. (2007) Individual dose adjustment of oral busulfan using a test dose in hematopoietic stem cell transplantation. Int J Hematol 86: 261-268.
-
Beri R, Chunduri S, Sweiss K, Peace DJ, Mactal-Haaf C, et al. (2010) Reliability of a pretransplant i.v. BU test dose performed 2 weeks before myeloablative FluBu conditioning regimen. Bone Marrow Transplant 45: 249-253.
-
Nath CE, Earl JW, Pati N, Stephen K, Shaw PJ (2008) Variability in the pharmacokinetics of intravenous busulphan given as a single daily dose to paediatric blood or marrow transplant recipients. Br J Clin Pharmacol 66: 50-59.
-
Gulbis AM, Culotta KS, Jones RB, Andersson BS (2011) Busulfan and metronidazole: an often forgotten but significant drug interaction. Ann Pharmacother 45: e39.
-
de Castro FA, Piana C, Simões BP, Lanchote VL, Pasqua O, Della (2015) Busulfan dosing algorithm and sampling strategy in stem cell transplantation patients. Br J Clin Pharmacol 80: 618-629.
-
Johnson-Davis KL, McMillin GA, Juenke JM, Ford CD, Petersen FB (2010) Which dose of busulfan is best? Clin Chem 56: 1061-1064.
-
Nath CE, Shaw PJ (2007) Busulphan in blood and marrow transplantation: dose route frequency and role of therapeutic drug monitoring. Curr Clin Pharmacol 2: 75-91.
-
Czerwinski M, Gibbs JP, Slattery JT (1996) Busulfan conjugation by glutathione S-transferases alpha, mu, and pi. Drug Metab Dispos 24: 1015-1019.
-
Galaup A, Paci A (2013) Pharmacology of dimethanesulfonate alkylating agents: busulfan and treosulfan. Expert Opin Drug Metab Toxicol 9: 333-347.
-
Nilsson C, Aschan J, Hentschke P, Ringdén O, Ljungman P, et al. (2003) The effect of metronidazole on busulfan pharmacokinetics in patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant 31: 429-435.
-
Hassan M, Svensson JO, Nilsson C, Hentschke P, Al-Shurbaji A, et al. (2000) Ketobemidone may alter busulfan pharmacokinetics during high-dose therapy. Ther Drug Monit 22: 383-385.
-
Hassan M, Oberg G, Björkholm M, Wallin I, Lindgren M (1993) Influence of prophylactic anticonvulsant therapy on high-dose busulphan kinetics. Cancer Chemother Pharmacol 33: 181-186.
-
Buggia I, Zecca M, Alessandrino EP, Locatelli F, Rosti G, et al. Itraconazole can increase systemic exposure to busulfan in patients given bone marrow transplantation. GITMO (Gruppo Italiano Trapianto di Midollo Osseo). Anticancer Res 16: 2083-2088.
-
dos Reis EO, Vianna-Jorge R, Suarez-Kurtz G, Lima ELDS, Azevedo DDA (2005) Development of a rapid and specific assay for detection of busulfan in human plasma by high-performance liquid chromatography/electrospray ionization tandem mass spectrometry. Rapid Commun Mass Spectrom 19: 1666-1674.
-
Kangarloo SB, Naveed F, Ng ES, Chaudhry MA, Wu J, et al. (2012) Development and validation of a test dose strategy for once-daily i.v. busulfan: importance of fixed infusion rate dosing. Biol Blood Marrow Transplant 18: 295-301.
-
Malär R, Sjöö F, Rentsch K, Hassan M, Güngör T (2011) Therapeutic drug monitoring is essential for intravenous busulfan therapy in pediatric hematopoietic stem cell recipients. Pediatr Transplant 15: 580-588.
-
Czerwinski M, Gibbs JP, Slattery JT (1996) Busulfan conjugation by glutathione S-transferases alpha mu and pi. Drug Metab Dispos 24: 1015-1019.
-
Gibbs JP, Czerwinski M, Slattery JT (1996) Busulfan-glutathione conjugation catalyzed by human liver cytosolic glutathione S-transferases. Cancer Res 56: 3678-3681.
-
Maurice M, Pichard L, Daujat M, Fabre I, Joyeux H, et al. (1992) Effects of imidazole derivatives on cytochromes P450 from human hepatocytes in primary culture. FASEB J 6: 752-758.
-
Dilger K, Fux R, Röck D, Mörike K, Gleiter CH (2007) Effect of high-dose metronidazole on pharmacokinetics of oral budesonide and vice versa: a double drug interaction study. J Clin Pharmacol 47: 1532-1539.
-
Roedler R, Neuhauser MM, Penzak SR (2007) Does metronidazole interact with CYP3A substrates by inhibiting their metabolism through this metabolic pathway? Or should other mechanisms be considered? Ann Pharmacother 41: 653-658.
-
Uppugunduri CRS, Rezgui MA, Diaz PH, Tyagi AK, Rousseau J, et al. (2014) The association of cytochrome P450 genetic polymorphisms with sulfolane formation and the efficacy of a busulfan-based conditioning regimen in pediatric patients undergoing hematopoietic stem cell transplantation. Pharmacogenomics J 14: 263-271.
-
Drug Development and Drug Interactions: Table of Substrates Inhibitors and Inducers US Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research.





