Could Accidental Ischemic Attacks Protect Against Subsequent Ischemic Reperfusion Injury?
Mohamed S. A. Mohamed*
Department of Thoracic Transplantation, University Clinic Essen, Germany
*Corresponding author:
Mohamed S. A. Mohamed, MBBCh, MSc, MD, Department of Thoracic Transplantation, University Clinic Essen, Hufeland Straße 55. D- 45147 Essen, Germany, E-mail: Mohammed.Shehatta1@gmail.com
Int J Transplant Res Med,
IJTRM-1-009, (Volume 1, Issue 2),
Retrospective Study
Received: July 27, 2015: Accepted: August 17, 2015: Published: August 20, 2015
Citation: Mohamed MSA (2015) Could Accidental Ischemic Attacks Protect Against Subsequent Ischemic Reperfusion Injury?. Int J Transplant Res Med 1:009
Copyright: © 2015 Mohamed MSA. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Heart failure is the end result of various cardiac diseases that might be of ischemic or non-ischemic etiology. Implantation of ventricular assisting devices has been introduced as a bridge to heart transplantation, when heart failure reaches a critical degree of decompensation. The ischemic background of heart failure was suggested to have a protective effect in comparison to the non-ischemic background, based on the principles of ischemic conditioning. This manuscript tries to provide a short and concentrated answer to the question; whether or not the accidental ischemic attacks could provide a degree of protection during subsequent ischemic- reperfusion injury, based on basic and clinical knowledge.
Keywords
Cardiomyopathy, Heart failure, Ventricular assist device implantation, Ischemic preconditioning, Ischemic reperfusion injury
Introduction
Ischemic pre-conditioning (IPC) has been investigated in various animal species [1], where it has been found to protect against post-ischemic contractile dysfunction as well as ischemic- reperfusion injury (IRI) in the heart and the liver [2,3]. The significant protective effects of IPC have paid the attention to the question; whether some protection could be achieved through the accidental ischemic attacks.
The mechanisms underlying IPC are not totally clear. However, a considerable progress has been achieved towards the identification of many mediators involved in its mechanism of action. A role of paracrine mediators, released during the period of ischemia has been suggested. Various studies have also suggested a role of adenosine, acetylcholine, catecholamines, angiotensin II, bradykinin, endothelin, opioids, nitric oxide (NO) and reactive oxygen species (ROS) [2].
For instance, the breakdown of ATP in myocytes during periods of ischemia results in adenosine, which is involved in triggering pre-conditioning in most of the studied animal species and humans. Blockage of adenosine A1 receptors in myocytes was found to block the protective effects of IPC, while re-stimulation of these receptors re-established the protective effects [2]. In addition to adenosine, protein kinase C, Ca2+, opioid receptors, ROS and mitochondrial KATP could play important roles in mediating the protective effects achieved by IPC.
It has recently been established that the activation of KATP channels antagonizes the opening of mitochondrial permeability transition pore during reperfusion, thereby preventing uncoupling of the mitochondria. In addition, heme-oxygenase up regulation and NO have been identified to mediate IPC and remote IPC [2,4,5].
The half- lives of the above mentioned mediators and pathways could provide an answer to our addressed question. Accordingly, even if the accidental ischemic attacks were able to provide the protective effect of IPC, such protection would be expected to last for limited periods (2-3 hours) [6]. In other words, IPC should be applied shortly before the IRI to be able to exert its protective effects [7,8]. In addition, it is well established that the duration of the ischemia- reperfusion cycles plays an essential role in IPC, with cycles lasting less than 10 minutes are able to provide protection, while cycles of more than 10 minutes duration are not protective and may be harmful [7,8]. Accordingly, the accidental ischemic attacks don't seem to be able to provide the protective role of IPC, and the following is a clinical evidence of that.
Methodology
This is a retrospective randomized study, where 46 cardiomyopathy patients, subjected to left ventricular assist device (LVAD) implantation by our surgical team between 2011 and 2014, were randomized in 2 groups (23 patients each), based on the ischemic and the non-ischemic backgrounds of the advanced left ventricular systolic dysfunction, which was defined as left ventricular ejection fraction (LVEF) <40%, based on the echo-cardiographic findings. The classification into ischemic and non-ischemic cardiomyopathy patients was based on the referral data from the cardiology department, were careful history, examination and investigations were performed, including echocardiography and coronary angiography, when indicated. The ischemic background was claimed when the cardiomyopathy was associated with >75% stenosis in at least one major coronary artery, or when a history of myocardial infarction, and or infarct scar changes on the ECG, was identified. The non-ischemic cardiomyopathy (dilated CMP) was claimed as left ventricular dilatation and dysfunction in the absence of any of the above.
Calculation of the INTERMACS score
The INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support) scale provides a classification of the patients of the advanced heart failure, according to their hemodynamic profile and the level of target organ damage (Table 1) [9]. The INTERMACS classification is an outcome of the ventricular assist device, multi-center registry, which aimed at unifying the criteria of the clinical state of the advanced heart failure patients [9,10]. The development of the INTERMACS classification system optimized the pre-operative risk prediction, and accordingly, the decision making regarding the therapeutic strategy. The INTERMACS scale has been confirmed as a reliable predictor of mortality [9,10] and postoperative complications following the implantation of ventricular assist devices [11]. Based on the history, clinical examination and investigations, the patients were assigned a pre-operative INTERMACS score, reference to the criteria listed in table 1.
![]()
Table 1: INTERMACS scale for classifying patients with advanced heart failure.
View Table 1
LVAD implantation
Based on the INTERMACS score, the patients were indicated for LVAD implantation, either as a bridge to transplantation or as a destination therapy. The pre-, intra-, and post-operative management were performed for all patients of both groups according to our standard institutional protocols.
Study end points
In the present study, we concentrated on the INTEMACS score pre-operative and the 30-day survival post-operative as the main targets of comparison between the study groups (Figure 1).
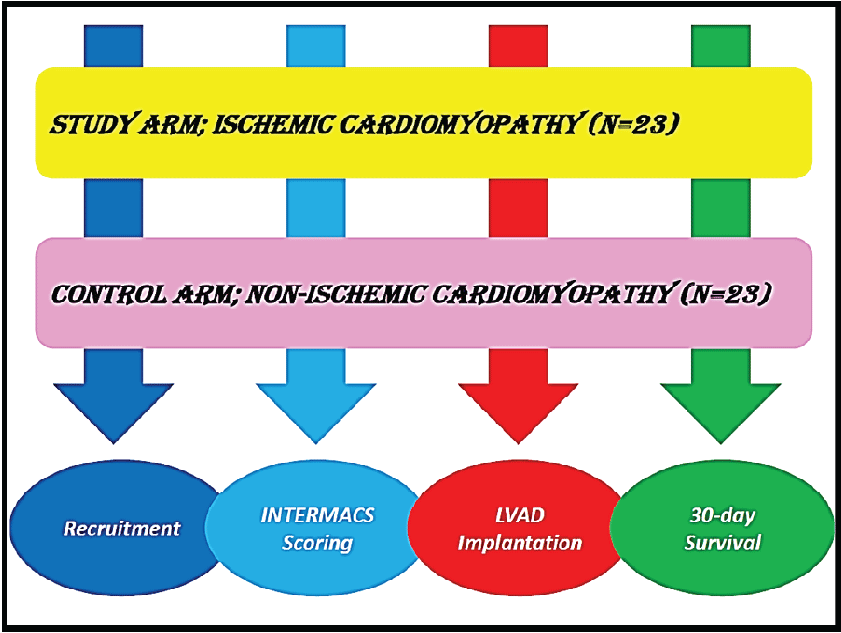
.
Figure 1: Diagrammatic representation of the study design.
Study design- Retrospective randomized clinical study
Study arm- LVAD patients with ischemic cardiomyopathy
Control arm- LVAD patients with dilated cardiomyopathy
Intervention- LVAD implantation
Study end points- INTERMACS score before LVAD implantation
Survival after LVAD implantation
View Figure 1
Statistical analysis
The statistical analysis was performed using Microsoft Excel 2007 for the calculation of means and standard deviations. Comparison between both groups regarding the study parameters was done using the GraphPad Prism 5 Demo and non- paired T test to identify significant differences. Kaplan-Meier survival analysis and the log rank test were applied to confirm the comparison between groups regarding the cumulative survival. P values ≤ 0.05 were considered significant.
Results
INTERMACS score
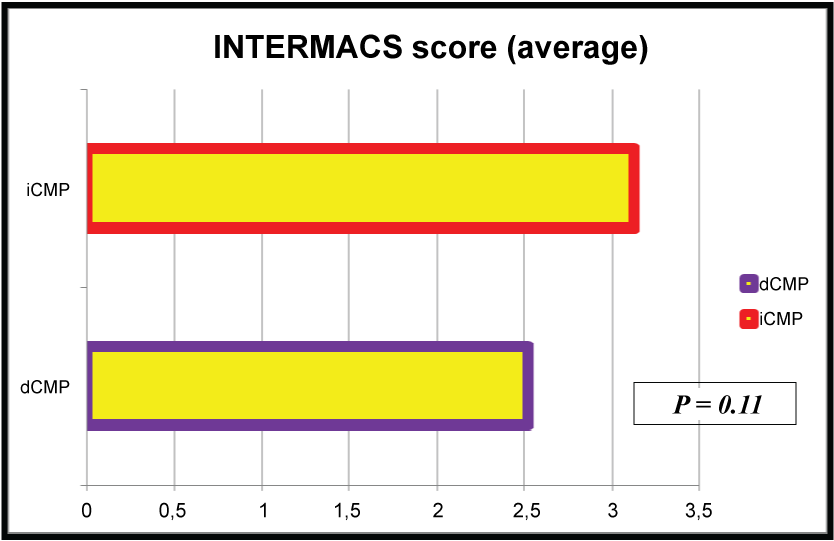
The average INTERMACS score of the dCMP group was 2.5 ± 1.3, compared to 3.1 ± 1.2 of the iCMP group. The difference between both groups was non-significant (P=0.11) (Figure 2).
.
Figure 2: The average INTERMACS scores of both study groups. The difference was non-significant (P>0.05)
View Figure 2
Postoperative survival
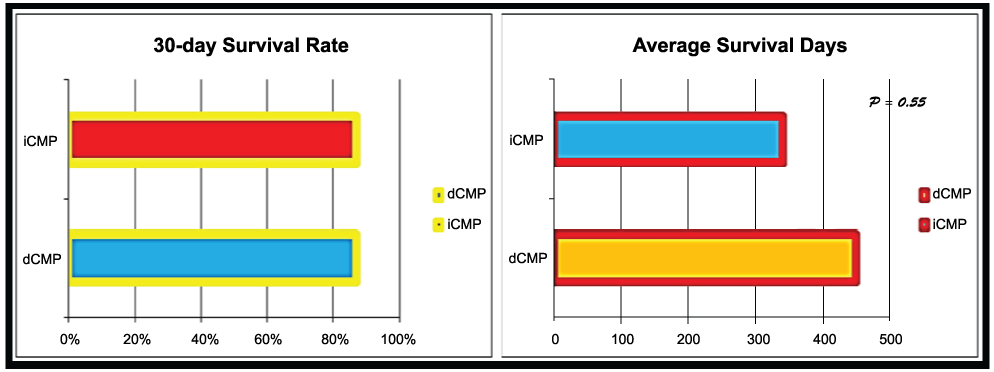
The 30-day survival rates of both groups were similar (87%), where 3 patients died within the first 30 days postoperative in each group. However, the average survival of the dCMP patients was 449.39 days, compared to 399.86 days of the iCMP patients. The difference between both groups was not significant (P=0.55) (Figure 3).
.
Figure 3: 30-day survival rates and the average survival days of both study groups. All rates showed non-significant differences (P>0.05).
View Figure 3
Kaplan-Meier analysis
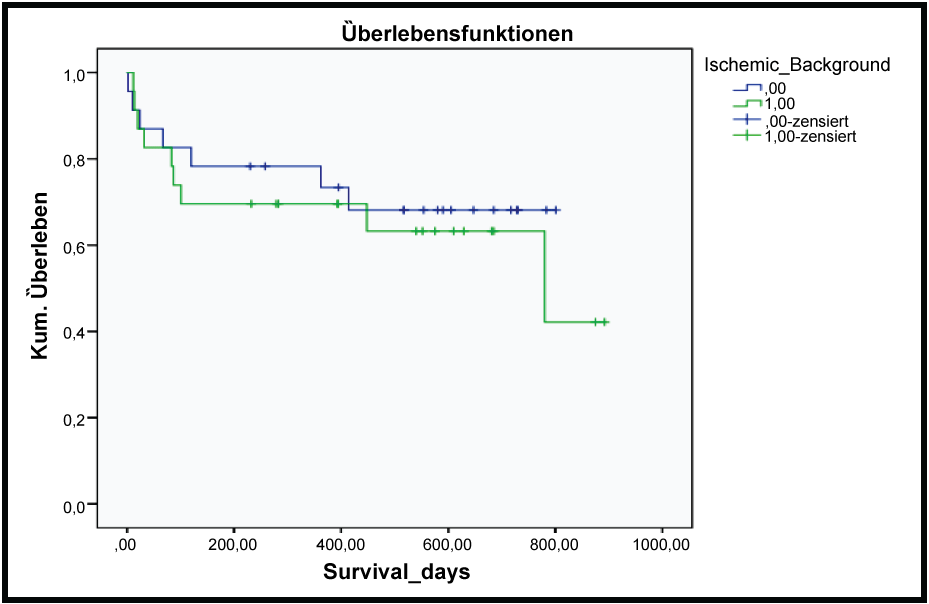
The Kaplan-Meier survival analysis and the log rank test confirmed the absence of a significant difference between both groups regarding the postoperative survival, which denies the ischemic background to be an independent predictor of patient survival (Chi-square=0. 314 and P=0.575) (Figure 4).
.
Figure 4: Kaplan-Meier analysis of postoperative survival based on the presence or absence of the ischemic background.
View Figure 4
Discussion
Most of the previous clinical studies reported the benefit of the medical therapy for both iCMP and dCMP, however, with higher mortality and weaker response to treatment in the patients of the ischemic background, particularly in response to beta-blockers [12-14] and ACE inhibitors [15,16]. Accordingly, it was interesting to address this question on the level of the surgical therapy and postoperative survival, especially with the increasing evidence of the value of the ischemic preconditioning technique.
In a study of 3787 systolic heart failure patients, the ischemic background was a significant independent predictor of mortality during follow-up [17]. Supportively, Stevenson et al. [18] concluded the coronary heart disease as an independent mortality predictor and stated the ischemic background as an indication of the priority listing for heart transplantation [18]. Similar findings were reported by several studies, where the overall all statements were that; the ischemic heart failure has a worse prognosis than the non-ischemic heart failure [19,20]. Ng et al. [21] showed a greater symptomatic improvement during follow-up for the non-iCMP patients, who showed better ventricular remodeling than the patients of iCMP, however, the difference in mortality was not significant [21].
Such worse prognosis associated with iCMP was referred to the older age of the patients, the presence of many other co-morbidities, and the potential association with a higher level of neuro-hormonal and immunological activation [21-23], where, in both dCMP and iCMP, there is a risk of sudden death due to heart failure, with an additional risk arising from the coronary disease, which might result in necrotic and or scarring tissue that might have the potential to impede the ischemic preconditioning [18,19].
On the other side, data from the Framingham community study, claimed the ischemic heart failure to be associated with better clinical outcomes than those of the non-ischemic heart failure [24]. In addition, the current guidelines do not distinguish between the heart failure etiologies [25,26], because whatever the basic etiology was, the subsequent pathophysiology of heart failure would be similar [27].
In the middle between both claims, Lourenco et al. [28] reported greater in-hospital mortality of iCMP, based on a univariate analysis. However, the multivariate analysis revealed that the etiology is not an independent mortality predictor [28]. Accordingly, they stated no significant difference between iCMP and non-iCMP regarding the long-term prognosis. The results that go with those of the recent registries of acute heart failure, such as ADHERE [23], OPTIMIZE-HF [24] and The Euro Heart Failure Survey [29], where the increased mortality risk associated with iCMP may be practically observed in patients with preserved systolic functions, however, in patients with advanced systolic heart failure, the ischemic background has no specific impact on the prognosis [30].
While all the previous studies focused on the prognosis and survival of heart failure in response to medical therapy, in the present study, we provided a statement regarding the survival following LVAD implantation. As LVAD implantation involves exposing the heart to an ischemic reperfusion injury (due to cardiopulmonary bypassing), the principles of ischemic preconditioning raised the attention towards the notion that the previous ischemic attacks might have provided some degree of protection that could be reflected on the postoperative survival.
In the present study, the INTERMACS scoring reflected the prognosis of heart failure based on the ischemic and non-ischemic etiologies. However, the survival after LVAD implantation reflected whether or not the accidental ischemic attacks could provide a degree of protection against the subsequent ischemic reperfusion injuries. Our results clearly showed similarity between the ischemic and the non-ischemic cardiomyopathy patients in the INTERMACS scoring pre-operative and in the survival postoperative.
As the present study is a retrospective short comment, whose study groups are of a relatively small size, with many clinical parameters (such as ventilation time, ICU stay, requirement of ECMO and total in hospital stay) were not included, a prospective randomized study is required for further confirmation of our results.
References
-
Wever KE, Menting TP, Rovers M, van der Vliet JA, Rongen GA, et al. (2012) Ischemic preconditioning in the animal kidney, a systematic review and meta-analysis. PLoS One 7: e32296.
-
Sivaraman V, Yellon DM (2014) Pharmacologic Therapy That Simulates Conditioning for Cardiac Ischemic- Reperfusion Injury. J Cardiovasc Pharmacol Ther 19: 83-96.
-
Liu A, Fang H, Wei W, Dirsch O, Dahmen U (2014) Ischemic preconditioning protects against liver ischemia/reperfusion injury via heme oxygenase-1-mediated autophagy. Crit Care Med 42: e762-771.
-
Hausenloy DJ, Maddock HL, Baxter GF, Yellon DM (2002) Inhibiting mitochondrial permeability transition pore opening: a new paradigm for myocardial preconditioning? Cardiovasc Res 55: 534-543.
-
Juhaszova M, Zorov DB, Kim SH, Pepe S, Fu Q, et al. (2004) Glycogen synthase kinase-3beta mediates convergence of protection signaling to inhibit the mitochondrial permeability transition pore. J Clin Invest 113: 1535-1549.
-
Muscari C, Giordano E, Bonafè F, Govoni M, Pasini A, et al. (2013) Molecular mechanisms of ischemic preconditioning and postconditioning as putative therapeutic targets to reduce tumor survival and malignancy. Med Hypotheses 81: 1141-1145.
-
Iliodromitis EK, Lazou A, Kremastinos DT (2007) Ischemic preconditioning: protection against myocardial necrosis and apoptosis. Vasc Health Risk Manag 3: 629-637.
-
Ferdinandy P, Schulz R, Baxter GF (2007) Interaction of Cardiovascular Risk Factors with Myocardial Ischemia-Reperfusion Injury, Preconditioning, and Postconditioning. Pharmacol Rev 59: 418-458.
-
Stevenson LW, Pagani FD, Young JB, Jessup M, Miller L, et al. (2009) INTERMACS profiles of advanced heart failure: the current picture. J Heart Lung Transplant 28: 535-541.
-
Holman WL, Pae WE, Teutenberg JJ, Acker MA, Naftel DC, et al. (2009) INTERMACS: interval analysis of registry data. J Am Coll Surg 208: 755-761.
-
Alba AC, Rao V, Ivanov J, Ross HJ, Delgado DH (2009) Usefulness of the INTERMACS scale to predict outcomes after mechanical assist device implantation. J Heart Lung Transplant 28: 827-833.
-
Poole-Wilson PA, Swedberg K, Cleland JG, Di Lenarda A, Hanrath P, et al. (2003) Comparison of carvedilol and metoprolol on clinical outcomes in patients with chronic heart failure in the Carvedilol Or Metoprolol European Trial (COMET): randomised controlled trial. Lancet 362: 7-13.
-
(1999) Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF) Lancet 353: 2001-2007.
-
(1994) A randomized trial of beta-blockade in heart failure. The Cardiac Insufficiency Bisoprolol Study (CIBIS). CIBIS Investigators and Committees. Circulation 90: 1765-1773.
-
Garg R, Yusuf S (1995) Overview of randomized trials of angiotensin-converting enzyme inhibitors on mortality and morbidity in patients with heart failure. Collaborative Group on ACE Inhibitor Trials. JAMA 273: 1450-1456.
-
Nony P, Boissel JP, Girard P, Leizorovicz A, Lievre M, et al. (1992) Relative efficacy of angiotensin converting enzyme inhibitors on mortality of patients with congestive heart failure: implications of randomized trials and role of the aetiology (ischaemic or nonischaemic) of heart failure. Eur Heart J 13: 1101- 1108.
-
Bart BA, Shaw LK, McCants CB Jr, Fortin DF, Lee KL, et al. (1997) Clinical determinants of mortality in patients with angiographically diagnosed ischemic or nonischemic cardiomyopathy. J Am Coll Cardiol 30: 1002-1008.
-
Stevenson LW, Tillisch JH, Hamilton M, Luu M, Chelimsky-Fallick C, et al. (1990) Importance of hemodynamic response to therapy in predicting survival with ejection fraction less than or equal to 20% secondary to ischemic or nonischemic dilated cardiomyopathy. Am J Cardiol 66: 1348-1354.
-
Likoff MJ, Chandler SL, Kay HR (1987) Clinical determinants of mortality in chronic congestive heart failure secondary to idiopathic dilated or to ischemic cardiomyopathy. Am J Cardiol 59: 634-638.
-
Miller WL, Hartman KA, Burritt MF, Burnett JC Jr, Jaffe AS (2007) Troponin, B-type natriuretic peptides and outcomes in severe heart failure: differences between ischemic and dilated cardiomyopathies. Clin Cardiol 30: 245-250.
-
Ng AC, Sindone AP, Wong HS, Freedman SB (2008) Differences in management and outcome of ischemic and non-ischemic cardiomyopathy. Int J Cardiol 129: 198-204.
-
Follath F, Cleland JG, Klein W, Murphy R (1998) Etiology and response to drug treatment in heart failure. J Am Coll Cardiol 32: 1167-1172.
-
Deng MC, Brisse B, Erren M, Khurana C, Breithardt G, et al. (1997) Ischemic versus idiopathic cardiomyopathy: differing neurohumoral profiles despite comparable peak oxygen uptake. Int J Cardiol 61: 261-268.
-
Ho KK, Pinsky JL, Kannel WB, Levy D (1993) The epidemiology of heart failure: the Framingham Study. J Am Coll Cardiol 22: 6-13.
-
Dickstein K, Cohen-Solal A, Filippatos G, McMurray JJ, Ponikowski P, et al. (2008) ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur Heart J 29: 2388-2442.
-
Hunt SA, Abraham WT, Chin MH, Feldman AM, Francis GS, et al. (2009) 2009 Focused Update Incorporated Into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults - A Report of the American College of Cardiology Foundation/American Heart Association. J Am Coll Cardiol 53: e1-90.
-
Abraham WT, Singh B (1999) Ischemic and nonischemic heart failure do not require different treatment strategies. J Cardiovasc Pharmacol 33 3: S1-7.
-
Lourenço C, Saraiva F, Martins H, Baptista R, Costa S, et al. (2011) Ischemic versus non-ischemic cardiomyopathy--are there differences in prognosis? Experience of an advanced heart failure center. Rev Port Cardiol 30: 181-197.
-
Velavan P, Khan NK, Goode K, Rigby AS, Loh PH, et al. (2010) Predictors of short term mortality in heart failure - insights from the Euro Heart Failure survey. Int J Cardiol 138: 63-69.
-
Mullens W, Abrahams Z, Skouri HN, Taylor DO, Starling RC, et al. (2008) Prognostic evaluation of ambulatory patients with advanced heart failure. Am J Cardiol 101: 1297-1302.





