International Journal of Surgery Research and Practice
Gender Differences in Left Ventricular Assist Device (LVAD) Support
Sneha Raju, Morgan Leigh Zych, Jane MacIver, FilioBillia and Vivek Rao*
Toronto General Hospital, University Health Network, Canada
*Corresponding author:
Vivek Rao, Chief. Cardiovascular Surgery, 457-4PMB, Peter Munk Cardiac Centre, Toronto General Hospital, 200 Elizabeth Street, Toronto, Ontario, Canada, M5G 2C4, Tel: +416-340-3562, Fax: +416-340-3337, E-mail: vivek.rao@uhn.ca
Int J Surg Res Pract, IJSRP-4-053, (Volume 4, Issue 2), Case Report; ISSN: 2378-3397
Received: September 07, 2016 | Accepted: March 31, 2017 | Published: April 03, 2017
Citation: Sneha R, Zych ML, Jane M, Billia F, Rao V (2017) Gender Differences in Left Ventricular Assist Device (LVAD) Support. Int J Surg Res Pract 4:053. 10.23937/2378-3397/1410053
Copyright: © 2017 Sneha R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Studies have demonstrated gender differences in adverse events after cardiac surgery, with men experiencing more favorable outcomes. The scarce gender-focused literature in the left ventricular assist device (LVAD) population has failed to find consistent differences. It has also been suggested that women experience longer transplant wait times due to higher PRA values.
Methods: A retrospective chart review was conducted of 28 women and 75 men who received an LVAD from 2006-2015. Chi squares, T tests, nonparametric tests, regression and Kaplan-Meier analysis were used as appropriate to compare event rates between groups. Re-admissions were categorized using the INTERMACS Appendix.
Results: Before and after correction for gender size differences, women on LVAD support were shown to wait longer for transplant (p = 0.02, p = 0.006). On average, women waited for 562 ± 374 days versus 279 ± 207 days for men. During LVAD support, mortality rates were 25% in men and 39% in women (p > 0.05). There were no differences in outcomes, re-admissions, or time spent in hospital (p > 0.05). While women have higher PRA values than men (p = 0.04, p = 0.04), patients' PRA values at discharge did not relate to transplant wait time (p = 0.13).
Patients maximum PRAs throughout LVAD support showed a statistically significant but mild relationship to transplant wait time (p = 0.04, R2 = 0.07).
Conclusions: Since men and women spend similar amounts of time in-hospital, this suggests that the longer support times observed in women are not associated with increased adverse events. Gender differences in PRAs were not sufficient to explain women's longer transplant wait times.
Introduction
In Canada, 40,000 women are newly diagnosed with heart failure (HF) every year [1]. While the prevalence and incidence of HF is comparable among genders, women exhibit a unique risk factor profile [1,2]. While some reports claim that women with HF experience increased adverse events and higher mortality rates, [3-9] others have shown that women present with fewer comorbidities and preserved ejection fractions, reducing total mortality [10-13]. End-stage heart failure requires intervention, which could include medical therapy, mechanical circulatory support, or cardiac transplantation. Cardiac transplantation is still considered the ideal treatment for advanced HF, but donor heart shortages have led to increasing numbers of patients being bridged with left ventricular assist devices (LVAD).
Women were underrepresented in most multi-center, randomized controlled trials for mechanical circulatory support (MCS). Further, it has been shown that women recovering from cardiac surgeries, including coronary artery bypass grafting, aortic valve repair, and cardiac transplantation, have poorer outcomes then men [14,15]. The literature is scarce regarding gender-related LVAD outcomes and complications. Studies from the pulsatile era found that women experienced increased mortality and higher incidences of bleeding and neurological events [3,9,16]. Studies from the non-pulsatile era examining gender differences in LVAD support have generally found no differences in mortality [8,17-19].
When it comes to post-LVAD transplantation, women tend to receive fewer cardiac transplants and wait longer to receive them [8]. Lower rates of transplantation may be explained by higher titres of panel reactive antibodies (PRAs) in parous women. Additionally, women may be at higher risk of antibody-mediated rejection of donor allograft [19]. Studies have also shown an increase in sensitization post LVAD implantation [20]. The primary objective of the present study is to analyze sex-related outcomes post-LVAD implantation, with a special interest in transplant wait times, re-admissions, and PRA differences.
Methods
The present study is a retrospective chart review of 103 patients who received continuous flow LVADs at Toronto General Hospital from June 2006 until May 2015. All patients who received a Heart Mate II, Heart Ware, Dura heart, and Heart Mate III device were included in the analysis. Re-admissions were used as a surrogate marker for overall patient morbidity. Patient wait-times were based on date of LVAD implant. Length and etiology of readmissions as categorized using INTERMACS 2015 Adverse Events Definitions were recorded. The institutional research ethics board approved the project.
Complete records were obtained of patients' panel reactive antibody testing history. Class I antibody values were collected and used for analyses as these values have the greatest bearing on transplant assessments at our centre. Test results collected included general PRA class I percentages as well as calculated PRAs (cPRAs) from single antigen specificity assays. For each member of the study population, two PRA values were obtained. These included the measurement after surgery, which was taken closest to time of discharge, and the highest value obtained during time on LVAD support. PRA levels were not assessed in those LVAD recipients who were not transplant eligible. However, in those patients who were eventually transplanted, PRA values were available in 100% of cases.
Statistical analysis was conducted using SAS 9.4 and SPSS software. An alpha level of p = 0.05 was set for all analyses and all tests conducted were two-sided. Categorical variables were analyzed using chi-squared tests. Continuous variables were assessed using unpaired T-tests. Variables that did not exhibit normal distributions were analysed using nonparametric tests. Linear regression analysis was conducted to assess the relationships between PRAs and patients' LVAD support time, the time spent outside of hospital, and waiting time for transplant.
Men in the study population weighed significantly more than women (85 ± 20 vs. 62 ± 10 kg, Table 1). Consequently, support times, time spent outside of hospital, and waiting time for transplant were also re-assessed after size adjustment with additional unpaired T-tests. Patients of a larger stature are expected to wait longer to have a donor heart that is able to adequately perfuse their body become available. This size adjustment excluded patients who had weights greater than 80 kg from subsequent T-tests to control for the effects of patient size on likelihood of transplantation. Survival comparisons were completed using the Kaplan-Meier method, censoring transplanted patients. Mantel-Cox log-rank test was used to compare differences in survival.
![]()
Table 1: Preoperative clinical characteristics.
View Table 1
Results
Seventy-five men and 28 women were included in the analysis (Table 1). There was a significant difference in heart failure etiology between the 2 groups with most men having an ischemic cardiomyopathy diagnosis and most women having a dilated cardiomyopathy diagnosis (p = 0.03, Table 1). Men had significantly higher preoperative weight, body mass index, and body surface area. These differences likely played a key role in the difference which was also observed in the type of device implanted, with the majority of men receiving a Heart Mate II and the majority of women receiving a Heart Ware HVAD (p = 0.005).
There were no significant differences in rates of transplantation and stroke, or in length of hospital stay and support times between the 2 groups (Table 2). Women waited for transplant for an average of 562 ± 374 days versus 279 ± 207 days for men (p = 0.02, Table 2). After size adjustment, these gender differences in wait times remained significant, with this cohort's wait times being 625 ± 345 days versus 273 ± 238 days for women and men, respectively (p = 0.006, Table 3).
![]()
Table 2: Postoperative outcomes.
View Table 2
![]()
Table 3: Postoperative outcomes, corrected for size*.
View Table 3
Women were found to have significantly higher class 1 PRA values throughout LVAD support (60% vs. 24% p = 0.04) and prior to discharge (49% vs. 17% p = 0.04) (Table 4). More specifically, women were more likely to be classified as highly sensitized (class 1 PRA > 90%) throughout LVAD support and at discharge (p = 0.0004 and p = 0.04, p = 0.0004, Table 5). Classification of patients as highly sensitized enables them to be listed for transplant as status 4S when applicable. A modest but statistically significant relationship was observed between patients' maximum PRAs on LVAD support and their transplant waiting time (p = 0.04, R2 = 0.07) (Table 5). Despite observed gender differences in each of these variables, patients' PRAs at discharge from hospital were not found to relate linearly to transplant wait times (p = 0.13, Table 5).
![]()
Table 4: Panel reactive antibodies (PRAs).
View Table 4
![]()
Table 5: Relationship of PRAs to support duration.
View Table 5
There were no differences in total number of readmissions or reason for admission between the 2 groups (p = 0.18, p > 0.05, Table 6). While there were no significant differences, women experienced double the incidence of bleeding in comparison to their male counterparts. Additionally, while the average number of readmissions between the genders did not reach significance, it is clear that women tended to be readmitted more and there was a wide variance in the group (SD = 4.6).
![]()
Table 6: Gender comparison of readmissions, mortality, and causes.
View Table 6
Figure 1 shows a Kaplan-Meier survival curve for men and women. There was no difference in perioperative, one year and 2-year mortality between the 2 groups (p = 0.611). Women exhibited higher mortality rates in each of the above-mentioned time points. While differences in overall mortality on LVAD support between genders did not reach significance, it is important to note that women experienced higher rates of mortality at 39.3% versus 25.3% for men (p = 0.17, Table 6). There were no significant gender differences in all-cause mortality (p = 0.32).
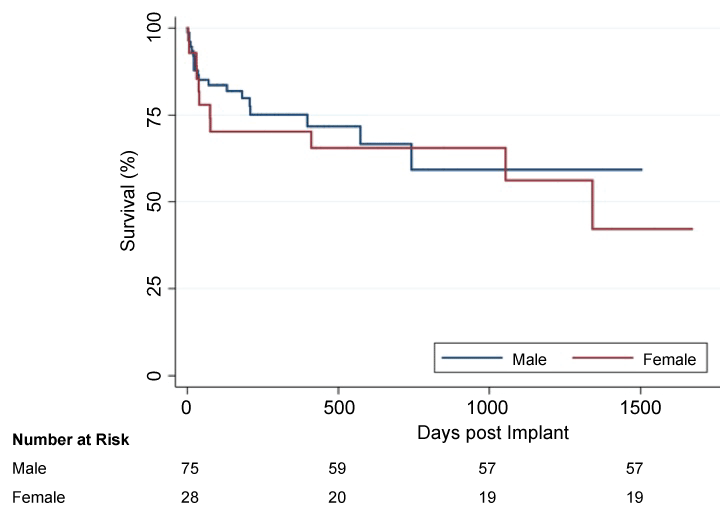
.
Figure 1: Kaplan-Meier survival for men and women during LVAD support. There were no differences in survival.
Log Rank (Mantel-Cox) = 0.611.
View Figure 1
Discussion
The findings from the present study have shown that on average, women have lower overall weight, BMI and BSA than their male counterparts. Further, men have significantly higher incidence of ischemic cardiomyopathy and are implanted with Heart Mate 2 versus their female counterparts who largely receive Heart Ware. Most importantly, women on LVAD support wait longer for transplant than men. Differences in transplant wait times were still present after correction for gender size differences, meaning that difficulties with size-matching donor hearts were not sufficient to explain observed wait times. Although women showed higher PRA values than men, it is not our opinion that these differences fully explained the gender discrepancy in transplant wait times either. A regression plotting patients' maximum PRAs with their transplant wait times did reach statistical significance, but with an R2 value of 0.07, this relationship can hardly be said to be linear. Although differences in mortality and readmissions between genders did not reach significance, the present study yielded observations which warrant further examination of these variables.
The finding that women on LVAD support have higher PRAs than their male counterparts is neither novel nor unexpected given the impact of pregnancies. The present study determined that the influence of these differences on transplant wait times may not be as straightforward as we expected. PRAs of women receiving LVAD support have consistently been shown to be higher than those of their male counterparts [21,22]. The relationship between PRAs and transplant wait times for people receiving LVAD support is less clear. A recent multi-center study which included earlier data from our own centre found that increased PRAs reduced odds of transplant, but had no significant impact on wait time [21]. When assessed categorically, authors have found that highly sensitized patients (class 1 or 2 > 25%) have longer wait times [22]. While the present study observed only a very mild relationship between PRAs and transplant waiting time, we cannot negate the impact which PRAs are known to have on transplantation decisions. It is now important that we consider which other variables could have influenced this pattern of data. We have two theories as to why this may have occurred. For women receiving LVAD support, the risks associated with transplantation may appear to outweigh the benefits more frequently, with the converse occurring more frequently for men. Our second theory for why women on LVAD support wait longer for transplant concerns the impact of clinicians' expectations of the impact of PRAs on transplant wait times. There is a chance that women are simply expected to wait longer due to their higher PRAs, so their care providers are not surprised or concerned when longer wait times are observed, hindering interventions to alter this trend.
In the general heart failure population, women have been shown to have worse outcomes waiting for transplant. In Morris, et al. examination of all adults listed for cardiac transplant in the United States from 2000-2014, women were shown to in fact spend fewer days on the transplant list, but have an increased mortality risk while waiting [23]. Furthermore, women were less likely to receive LVAD support than men [23]. Women were also at increased risk for removal from the transplant list for death or deterioration in The Waiting for a New Heart Study [24]. Although significant survival differences were not observed in our study population, these observations still provide incentive to further examine causes for increased transplant wait times their effects.
While the majority of baseline characteristics examined in the present study did not display gender differences, differences were observed in size, device, diagnosis, and presence of hypertension. Women in the present study were found to be significantly smaller than men with respect to weight, BMI, and BSA. Not unexpectedly, this has been true of other examinations of patients on LVAD support [8,25]. These size differences impact LVAD patients' courses of care through numerous avenues. Smaller patients are more frequently implanted with a Heart Ware HVAD due to its compact design, and larger patients are more frequently implanted with a Heart Mate device due to its power capabilities. It is also likelier that transplanted individuals receive hearts from oversized donors than undersized donors to accommodate for any residual pulmonary hypertension in the recipient. This makes our finding of increased transplant wait times for women all the more significant, since one might expect women to in fact receive acceptable hearts faster due to their smaller sizes. Our study also observed higher incidence of hypertension and ischemic etiology in men. Other authors have observed comparable gender differences for these variables in their LVAD patient populations [8,25] although this is not always the case [18].
The present study found no difference in all-cause mortality between women and men on continuous flow LVAD support. The literature examining gender differences in mortality post LVAD implantation is conflicting. Large, multicenter trials including one reporting findings from the INTERMACS database found no gender differences in LVAD patients' mortality [8,17,18,25]. Smaller studies did observe increased risk in women for mortality and right heart failure [3,26]. We are happy to note that the present study is in agreement with INTERMACS findings. Of note, most of the mortality observed in the present study was contained within the postoperative time point of index admission (19 patients, 63% of deaths) which is agreement with literature [8,18]. The sixth INTERMACS Annual Report described 1 and 2 year LVAD mortality rates at 20% and 31%, respectively. We observed comparable mortality rates of 23% at 1-year and 26% at 2-years. Interestingly, while INTERMACS reported a 30-day mortality of 5%, our center experienced rates nearing 18% [27]. This difference in mortality rates warrants further analysis at our center and might reflect center-specific practices such as discharge to external care facilities [18,25].
With an increasing population of longitudinally supported LVAD patients, it is important to monitor and reduce re-admissions to increase patients' quality of life and lower the financial burden on our healthcare system. While there were no statistically significant gender differences between the average numbers of readmissions, women had a higher number on average with a large variance. Of note, the patients that had the three highest numbers of readmissions [13,14,17] were all women. Unlike other centers [25], time spent in the hospital was not found to differ significantly between genders at our center. However, women spent more time hospitalized on average, with high variances. Interestingly, there were no significant differences in etiology for readmissions between the genders, which is promising considering women have been shown to be at higher risk of bleeding episodes post implantation. There does seem to be increased incidence of bleeding events in women, supporting findings at other centers [28,29]. While increased incidence of bleeding presents a concern independently, these adverse events were not associated with increased PRA values (p > 0.05 for all analyses).
The bulk of the present study's limitations center on its retrospective nature. More specifically, baseline characteristics that were not controlled for at the outset of the study may have impacted its results. Our sample size was relatively small, and we were not able to correct for repeated analysis on this sample. The present study also may not be widely generalizable due to the fact that it was a single-centre study. This being said, our centre serves a wide catchment area and is the largest LVAD center in the country.
The present study supports existing literature on gender differences in transplant wait times while questioning our assumptions regarding the reasons for these differences. Contrary to commonly held beliefs that patients' PRAs independently predict their time to transplant, we have not observed this relationship in our patient population. We are happy to report that there were no significant gender differences for actuarial mortality or readmissions.
Disclosures
Vivek Rao MD is a consultant to Terumo Corp (Tokyo), Thoratec Corp (Plesanton, Calif), and Heart Ware International Inc (Framingham, Mass). All other authors have nothing to disclose.
References
-
Lee DS, Johansen H, Gong Y, Hall RE, Tu JV, et al. (2004) Regional outcomes of heart failure in Canada. Can J Cardiol 20: 599-607.
-
Lloyd-Jones D, Adams R, Carnethon M, De Simone G, Ferguson TB, et al. (2009) Heart disease and stroke statistics--2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 119: e21-e181.
-
Morgan JA, Weinberg AD, Hollingsworth KW, Flannery MR, Oz MC, et al. (2004) Effect of gender on bridging to transplantation and post transplantation survival in patients with left ventricular assist devices. J Thorac Cardiovasc Surg 127: 1193-1195.
-
Weymann A, Patil NP, Sabashnikov A, Mohite PN, García Sáez D, et al. (2015) Gender Differences in Continuous-Flow Left Ventricular Assist Device Therapy as a Bridge to Transplantation: A Risk-Adjusted Comparison Using a Propensity Score-Matching Analysis. Artif Organs 39: 212-219.
-
Bourassa MG, Gurné O, Bangdiwala SI, Ghali JK, Young JB, et al. (1993) Natural history and patterns of current practice in heart failure. J Am Coll Cardiol 22: 14-19.
-
Boyle AJ, Jorde UP, Sun B, Park SJ, Milano CA, et al. (2014) Pre-operative risk factors of bleeding and stroke during left ventricular assist device support: An analysis of more than 900 HeartMate II outpatients. J Am Coll Cardiol 63: 880-888.
-
Reitsma B, Mosterd A, de Craen AJM, Koster RW, van Capelle FJL, et al. (1996) Increase in hospital admission rates for heart failure in the Netherlands, 1980-1993. Heart 76: 388-392.
-
Bogaev RC, Pamboukian SV, Moore SA, Chen L, John R, et al. (2011) Comparison of outcomes in women versus men using a continuous-flow left ventricular assist device as a bridge to transplantation. J Heart Lung Transpl 30: 515-522.
-
Najjar SS (2014) Late bleeding and neurological sequelae after HeartMate II left ventricular assist device: Risk factors from the prequel. J Am Coll Cardiol 63: 889-890.
-
O'Meara E, Clayton T, McEntegart MB, McMurray JJ, Piña IL, et al. (2007) Sex differences in clinical characteristics and prognosis in a broad spectrum of patients with heart failure - Results of the Candesartan in Heart failure: Assessment of Reduction in Mortality and morbidity (CHARM) program. Circulation 115: 3111-3120.
-
Galvao M, Kalman J, Demarco T, Fonarow GC, Galvin C, et al. (2006) Gender differences in in-hospital management and outcomes in patients with decompensated heart failure: Analysis from the acute decompensated heart failure national registry (ADHERE). J Card Fail 12: 100-117.
-
Nieminen MS, Harjola VP, Hochadel M, Drexler H, Komajda M, et al. (2008) Gender related differences in patients presenting with acute heart failure. Results from EuroHeart Failure Survey II. Eur J Heart Fail 10: 140-148.
-
Schocken D, Arrietta M, Leaverton P, Ross E (1992) Prevalence and mortality rate of congestive heart failure in the United States. J Am Coll Cardiol 20: 301-306.
-
Blankstein R, Ward RP, Arnsdorf M, Jones B, Lou YB, et al. (2005) Female gender is an independent predictor of operative mortality after coronary artery bypass graft surgery: Contemporary analysis of 31 midwestern hospitals. Circulation 112: 323-327.
-
Doenst T, Ivanov J, Borger MA, David TE, Brister SJ, et al. (2006) Sex-Specific Long-Term Outcomes After Combined Valve and Coronary Artery Surgery. Ann Thorac Surg 81: 1632-1636.
-
Rose EA, Gelijns AC, Moskowitz AJ, Heitjan DF, Stevenson LW, et al. (2001) Long-Term Use of a Left Ventricular Assist Device For End-Stage Heart Failure. N Engl J Med 345: 1435-1443.
-
Hsich EM, Naftel DC, Myers SL, Gorodeski EZ, Grady KL, et al. (2012) Should women receive left ventricular assist device support? Findings from INTERMACS. Circ Hear Fail 5: 234-240.
-
Tsiouris A, Morgan JA, Nemeh HW, Hodari A, Brewer RJ, et al. (2014) Sex-specific outcomes in patients receiving continuous-flow left ventricular devices as a bridge to transplantation or destination therapy. ASAIO J 60: 199-206.
-
Chih S, Chruscinski A, Ross HJ, Tinckam K, Butany J, et al. (2012) Antibody-Mediated Rejection: An Evolving Entity in Heart Transplantation. J Transplant 2012: 210210.
-
Pak S, Uriel N, Burke E, Cappleman SA, Sutton EM, et al. (2010) Abstract 20395: High Incidence of Elevated Panel Reactive Antibodies in Patients Supported with HeartMate II Left Ventricular Assist Devices. Circulation 122: A20395.
-
Alba AC, Tinckam K, Foroutan F, Nelson LM, Gustafsson F, et al. (2015) Factors associated with anti-human leukocyte antigen antibodies in patients supported with continuous-flow devices and effect on probability of transplant and post-transplant outcomes. J Heart Lung Transpl 34: 685-692.
-
Arnaoutakis GJ, George TJ, Kilic A, Weiss ES, Russell SD, et al. (2011) Effect of sensitization in US heart transplant recipients bridged with a ventricular assist device: Update in a modern cohort. J Thorac Cardiovasc Surg 142: 1236-1245.
-
Morris AA, Cole RT, Laskar SR, Kalogeropoulos A, Vega JD, et al. (2015) Improved outcomes for women on the heart transplant wait list in the modern era. J Card Fail 21: 555-560.
-
Weidner G, Zahn D, Mendell NR, Smits JM, Deng MC, et al. (2011) Patients' sex and emotional support as predictors of death and clinical deterioration in the waiting for a new heart study: Results from the 1-year follow-up. Prog Transplant 21: 106-114.
-
Birks EJ, McGee EC, Aaronson KD, Boyce S, Cotts WG, et al. (2015) An examination of survival by sex and race in the HeartWare ventricular assist device for the treatment of advanced heart failure (ADVANCE) bridge to transplant (BTT) and continued access protocol trials. J Heart Lung Transpl 346: 815-824.
-
Ochiai Y, McCarthy PM, Smedira NG, Banbury MK, Navia JL, et al. (2002) Predictors of severe right ventricular failure after implantable left ventricular assist device insertion: analysis of 245 patients. Circulation 106: 198-202.
-
Kirklin JK, Naftel DC, Pagani FD, Kormos RL, Stevenson LW, et al. (2014) Sixth INTERMACS annual report: A 10,000-patient database. J Heart Lung Transplant 31: 555-564.
-
Kato TS, Schulze PC, Yang J, Chan E, Shahzad K, et al. (2012) Pre-operative and post-operative risk factors associated with neurologic complications in patients with advanced heart failure supported by a left ventricular assist device. J Heart Lung Transpl 31: 1-8.
-
Tsiouris A, Paone G, Nemeh HW, Brewer RJ, Morgan JA, et al. (2014) Factors determining post-operative readmissions after left ventricular assist device implantation. J Heart Lung Transplant 33: 1041-1047.
-
Forest SJ, Bello R, Friedmann P, Casazza D, Nucci C, et al. (2013) Readmissions after ventricular assist device: Etiologies, patterns, and days out of hospital. Ann Thorac Surg 95: 1276-1281.





