International Journal of Sports and Exercise Medicine
Effect of Flare Reaction to Intra-Articular Hyaluronan Injection on Cartilage Boundary Lubricating Ability of Human Synovial Fluid
Taryn E Ludwig1, Jenelle R McAllister2, Victor Lun2, J Preston Wiley2,3 and Tannin A Schmidt1,3*
1Biomedical Engineering Graduate Program, University of Calgary, Canada
2Sport Medicine Centre, University of Calgary, Canada
3Faculty of Kinesiology, University of Calgary, Canada
*Corresponding author: Dr. Tannin A. Schmidt, KNB 426, Faculty of Kinesiology, University of Calgary, Biomedical Engineering, Calgary, AB, Canada, Tel: 403-220-7028, Fax: 403-284-3553, E-mail: tschmidt@ucalgary.ca
Int J Sports Exerc Med, IJSEM-2-042, (Volume 2, Issue 3), Original Article; ISSN: 2469-5718
Received: December 04, 2015 | Accepted: May 16, 2016 | Published: July 01, 2016
Citation: Ludwig TE, McAllister JR, Lun V, Wiley JP, Schmidt TA (2016) Effect of Flare Reaction to Intra-Articular Hyaluronan Injection on Cartilage Boundary Lubricating Ability of Human Synovial Fluid. Int J Sports Exerc Med 2:042. 10.23937/2469-5718/1510042
Copyright: © 2016 Ludwig TE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: The objectives of this study were to 1) quantify proteoglycan 4 (PRG4) and hyaluronan (HA) content in osteoarthritic (OA) synovial fluid (SF) after a flare reaction to intra-articular (IA) HA injection and 2) assess the cartilage boundary lubricating ability of PRG4-deficient flare-SF, with and without supplementation with PRG4 ± HA.
Methods: Seven SF samples from 5 OA patients who returned to the clinic within 11 days of initial IA HA were included in this study. PRG4 and HA concentration were measured by sandwich enzyme linked immunosorbent assay, and HA molecular weight (MW) was measured by 1% agarose gel electrophoresis. Five flare samples that were identified as having low PRG4 concentration were selected for cartilage-cartilage boundary lubricating ability tests.
Results: PRG4 and HA concentrations in flare-SF ranged from below normal to super-physiological. HA MW in these samples was shifted towards the lower range in the 3.1 - 6.1 and 0.5 - 1.1 MDa ranges (p = 0.02, 0.005) only. The kinetic coefficient of friction in PRG4-deficient flare-SF was not altered compared to normal SF, and no changes were observed with PRG4 or PRG4+HA supplementation (p = 0.70 - 1.0).
Conclusion: SF can exhibit altered boundary lubricant composition after a flare reaction to IA HA. Despite a decrease in PRG4 concentration in some samples, normal cartilage boundary lubricating ability was retained, possibly due to sufficient high MW HA content. This suggests that maintaining normal composition of both PRG4 and HA through biotherapeutic treatment may preserve SF lubricating function and therefore contribute to joint preservation and health.
Keywords
Cartilage, Osteoarthritis, Hyaluronan, Proteoglycan 4, Lubricin
Introduction
Lubrication of articular cartilage is achieved by a combination of lubrication mechanisms. Fluid film lubrication occurs at high speeds and low loads when cartilage surfaces are separated by a layer of synovial fluid (SF). Boundary lubrication occurs at low speeds and high loads when cartilage surfaces are in contact and lubrication is provided through molecular interactions at the surface [1]. This surface-to-surface contact is thought to occur over approximately 10% of the cartilage area, exposing these contact areas to high friction [2]. The hydrostatic pressure that supports load during fluid film lubrication dissipates over time with loading, causing the cartilage surfaces to bear more load [3]. Whole SF has been shown to effectively reduce friction in the boundary mode [4], and this boundary lubricating ability is provided primarily by its constituents proteoglycan 4 (PRG4) and hyaluronan (HA) [5]. SF boundary lubricant composition and function are of clinical interest, as friction has been observed to be associated with wear at the articular surface [6].
The PRG4 [7] gene encodes for mucin-like O-linked glycosylated proteins, collectively referred to as PRG4. PRG4, also known as lubricin, is synthesized by cells within articular joints and is present in SF [8] and at the articular cartilage surface [9]. PRG4 is important for normal cartilage boundary lubricating function; SF from patients with a genetic autosomal recessive disorder caused by mutations in the PRG4 gene, camptodactyly-arthropathy-coxa vara-pericarditis (CACP) syndrome [10], is void of PRG4 and fails to lubricate compared to normal SF [6]. Furthermore, PRG4 knock-out mice demonstrate earlier cartilage wear and higher total joint friction [6], and in human SF PRG4 concentrations are decreased for approximately 1 year after ACL tear [11]. While PRG4 is necessary for normal joint function, other SF constituents and their interactions with PRG4 are also required to maintain normal boundary lubricating ability.
HA, a linear polymer of repeating disaccharides composed of D-glucuronic acid and D-N-acetlyglucosamine [12], is another contributor to cartilage boundary lubrication in SF [5]. It appears that both PRG4 and HA are critical to the boundary lubricating function of SF, as PRG4 acts synergistically with HA to reduce friction to levels approaching that of whole SF [5]. Decreases in HA concentration and/or molecular weight [13] in post-injury equine SF and PRG4 [14] concentration in chronic osteoarthritis (OA) human SF have been observed to be linked to decreased boundary lubricating ability at a cartilage-cartilage interface in vitro; lubricating ability could respectively be restored with HA or PRG4 supplementation. While the mechanism of the PRG4-HA interaction is currently unknown, both are required to approach the boundary lubricating ability of SF in in vitro cartilage-cartilage boundary lubrication tests.
HA of various molecular weight (MW) is routinely used, over long terms, as an intra-articular (IA) viscosupplement for treatment of OA [15]. It has been shown to provide pain relief for up to 6 months [16] despite a comparatively short residence time in the joint (8.8 days for hylan G-F20, a high MW ~ 6MDa cross-linked product [17]). An inflammatory or flare reaction is an adverse event associated with all IA injections [18], including visco supplementation with HA [19]. There appears to be several types of flare reactions that can occur in response to IA injections. First, some flare reactions are associated with injection site pain and swelling [20] 24-72 hours after injection [21]; these reactions are usually mild, do not recur, and subsequent injections can be performed [20]. Secondly, injection of cross-linked HA products may be associated with infrequent severe acute inflammatory reactions, or pseudosepsis; these reactions often require clinical intervention and tend to occur after exposure to more than one injection [20]. Some flares occurring after hylan G-F20 injection are thought to be cell-mediated hypersensitivity reactions [22].
Through the natural course of disease, OA patients can experience "flare-ups" in symptoms; these flare-ups are characterized by sudden aggravation of knee pain, with an identifiable onset, that causes nocturnal awakening, and evidence of effusion [23]. SF from flare-up patients has been observed to have lower HA concentration (with higher MW), increased protein size and concentration, and decreased viscosity compared to OA patients without flare-up [23]; decreased viscosity could have an effect on the fluid film lubricating function of SF.
This study focuses on SF from patients that have had a flare reaction to IA HA injection, as the effects of a flare reaction to IA injection on SF boundary lubricant composition and function are unknown. The objectives of this study were to: 1) quantify PRG4 and HA content in OA SF after flare reaction to IA HA injection and 2) assess the cartilage boundary lubricating ability of PRG4-deficient flare-SF, with and without supplementation with PRG4 ± HA. These results were then compared to SF from non-arthritic knees.
Materials and Methods
SF samples
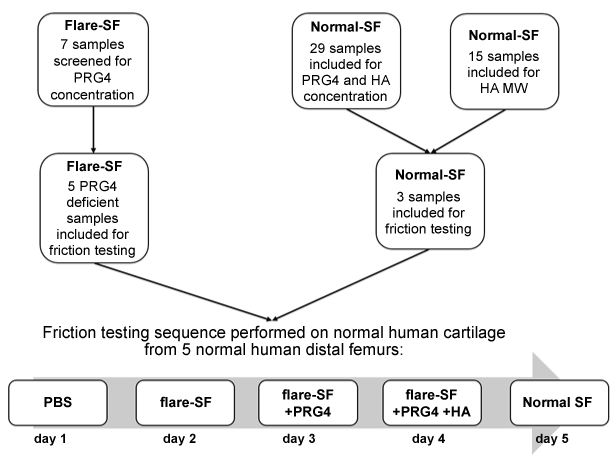
Collection of all human tissues and fluids was approved by the Conjoint Health Research Ethics Board at the University of Calgary. In a larger ongoing study, SF was aspirated from patients with symptomatic chronic knee OA requiring aspiration prior to therapeutic IA HA or corticosteroid injection. Patients were diagnosed with knee OA by 2 sport medicine physicians (co-authors VL and PW) after physical examination and review of patient symptoms and plain-film radiographs. For this study, a subset of patients who returned to the clinic for subsequent treatment within 2 weeks of receiving an IA HA injection (hylan G-F20) and whose knee was aspirated again at this time were included. This flare-SF was aspirated using standard sterile knee aspiration technique, and as much fluid as possible was aspirated with each attempt. The normal and flare-SF were clarified by centrifugation [11,24] prior to storage at -80°C with protease inhibitors, as well as without protease inhibitors for HA MW analysis when sufficient was volume available.
Seven flare-SF samples from 5 knee OA patients requiring aspiration ≤ 11 days after HA injection were included in this study. One patient had both the left and right knees aspirated after a flare reaction to IA HA in both knees, and one patient was aspirated on day 5 and day 7 after the initial injection (biochemical data for aspiration at day 5 and 7 are presented for completeness, however only the SF aspirated at day 7 was examined for lubricating ability). Samples were first screened for PRG4 concentration to select samples of interest for lubricating ability testing. Five samples from 4 patients were identified as having PRG4 concentration lower than the average normal SF concentration (cadaveric, N = 29) and were selected for boundary lubricating ability tests. The number of PRG4-deficient samples is not intended to reflect a proportion of flare population with low PRG4. PRG4 deficient samples were selected to allow characterization of the contribution of HA to SF lubricating ability.
Normal human cartilage samples
Normal SF and normal human distal femurs were obtained through the Joint Transplantation Program at the University of Calgary and were harvested within 4 hours of donor death. The normal human femurs were stored at -80°C until use, and were used as the friction testing substrate for all experiments. Use of frozen tissues allows isolation of the desired boundary lubricants, as it prevents production of PRG4 from viable chondrocytes.
Biochemical characterization of flare-SF
Total protein concentration in SF was measured by bicinchoninic acid assay. HA and PRG4 concentration were measured using sandwich enzyme linked immunosorbent assays (ELISA) as previously described [14]. Briefly, PRG4 concentration was measured in triplicate using a custom sandwich ELISA [14] with antibody LPN used to capture the C-terminal of full-length PRG4 [25] and peanut agglutinin-horseradish peroxidase to detect glycosylations in the mucin domain of PRG4 [26]. HA concentration was measured in triplicate using a commercially available kit from R&D Systems®. HA MW distribution was determined using 1% agarose gel electrophoresis [27] followed by staining with Stains-All and densitometric analysis using ImageJ (NIH, Bethesda, MD). HA MW distribution in each SF sample was quantified as relative abundance (%) in each of 5 MW ranges (based on size of control HA markers) spanning very high to low MW: > 6.1, 3.1 - 6.1, 1.1 - 3.1, 0.5 - 1.1, and < 0.5 MDa. HA MW was only characterized in 15 of the normal SF samples due to limited available volumes.
Cartilage-cartilage boundary lubricating ability
Human cartilage boundary lubricating ability of flare-SF deficient in PRG4, supplemented flare-SF, and normal SF was measured in a previously characterized in vitro cartilage-cartilage boundary mode friction test on a Bose ELF 3200 [4]. Annulus and core shaped osteochondral samples were harvested from the patellofemoral groove of 5 macroscopically normal human distal femurs (age 57 - 82 years). Samples were shaken overnight in phosphate buffered saline (PBS) at 4°C to remove any residual SF from the cartilage surface, and then soaked overnight at 4°C in the subsequent test lubricant prior to testing [14]. The cartilage surfaces of the annulus and core were opposed against each other, compressed to 18% of the total cartilage thickness, and allowed to stress-relax for 40 minutes. Without removing compression, samples were then rotated at 0.3 mm/second +2 and -2 revolutions with pre-sliding duration (Tps) of 120, 12, and 1.2 seconds; Tps is the duration of time the samples are opposed and stationary prior to rotation. The test was then repeated in the opposite direction of rotation. Static (μstatic,Neq, resistance to the onset of motion) coefficients of friction were calculated using the peak torque within 10° of the start of rotation. Torque from the second revolution was averaged to calculate the kinetic (<μkinetic,Neq>, resistance to steady motion) friction coefficients.
Each osteochondral pair was tested sequentially over 5 days in each of the 5 test lubricants. Each flare-SF deficient in PRG4 selected for friction testing (N = 5) was tested in triplicate (n = 3, total n = 15) in the following sequence: 1) PBS (negative control), 2) flare-SF deficient in PRG4, 3) flare-SF+PRG4, 4) flare-SF+PRG4+HA, 5) normal SF (positive control). Three normal SF samples were used as positive controls for boundary lubricating ability tests (age 56 - 59, PRG4 concentration 136 - 373 μg/mL). Flare-SF was supplemented with normal concentrations (based on ELISA measurements in normal SF, N = 29) of PRG4 and PRG4+HA. PRG4 at 450 μg/mL (obtained from bovine cartilage explants culture as described previously [5]) and 1.5 MDa HA at 1.0 mg/mL (obtained from Life core Biomedical, LLC) were used for supplementation. PRG4 and HA concentrations for supplementation were based on preliminary ELISA measurements in normal SF, and to be consistent with previously published experiments using the same methodology (Figure 1).
Statistical analysis
Data are presented as mean with 95% confidence interval (lower limit, upper limit) unless otherwise noted. Differences in PRG4 and HA composition were assessed by ANOVA. Arcsine square root transformation was used to improve uniformity of the variance for the proportional distribution of HA MW [28]. Repeated measures ANOVA was used to assess the effects of lubricant and Tps (as a repeated factor) on μstatic,Neq and <μkinetic,Neq>. The effect of lubricant on μstatic,Neq at each Tps and <μkinetic,Neq> at Tps = 1.2 s was assessed by ANOVA with Tukey post-hoc testing. Statistical analysis was performed with Systat 12.
Results
Biochemical characterization of flare-SF
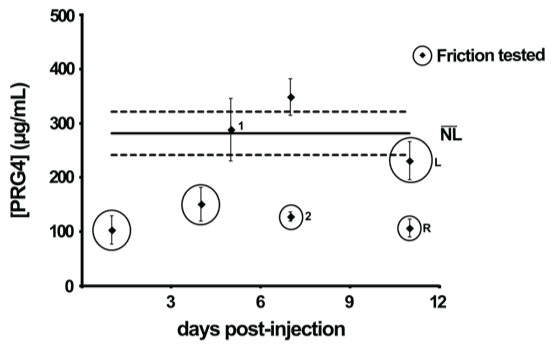
Flare-SF samples contained PRG4 and HA at concentrations ranging from below normal to super-physiological. PRG4 concentration in normal SF averaged 281.4 (241.6, 321.3) μg/mL (Figure 2). PRG4 concentration in flare-SF samples selected for friction testing were below the average normal concentration and ranged from 102.8 to 231.0 μg/mL; PRG4 concentrations in all flare samples ranged from 102.8 to 348.7 μg/mL.
.
Figure 2: PRG4 concentration in flare-SF after IA HA. PRG4 concentration in normal ((NL) ̅) SF (N = 29) shown by black line, ± 95% CI shown in dashed lines. PRG4-deficient samples selected for friction testing are circled. "L" and "R" denotes SF that was obtained from the left and right knee of the same patient. "1" and "2" denotes the 1st and 2nd aspiration of the same knee after a flare reaction to HA.
View Figure 2
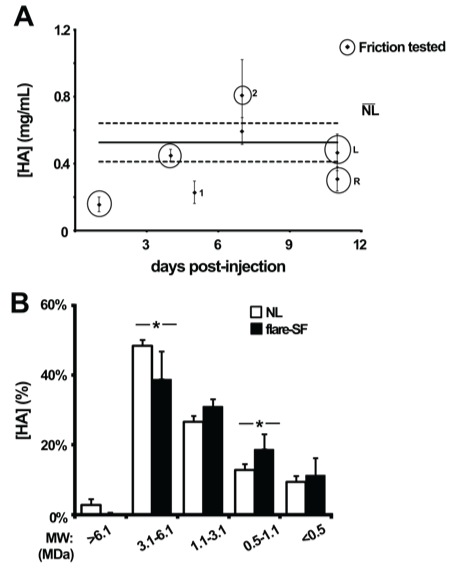
Table 1 summarizes the characteristics of the PRG4-deficient flare-SF selected for friction testing as well as the normal donors. HA concentration in normal SF averaged 0.53 (0.41, 0.64) mg/mL (Figure 3A). HA concentration in all flare-SF samples ranged from 0.16 to 0.68 mg/mL. Relative HA concentration (as a percentage of total concentration) in PRG4-deficient flare SF was significantly lower in the 3.1 - 6.1 MDa (p = 0.02) and 0.5 - 1.1 MDa (p = 0.005) ranges, and not significantly different from normal in the > 6.1 MDa (p = 0.095), 1.1 - 3.1 MDa (p = 0.16), and < 0.5 MDa (p = 0.20) ranges (Figure 3B).
.
Figure 3: (A) HA concentration in flare-SF after IA HA. HA concentration in normal ((NL) ̅) SF (N = 29) shown by black lines, ± 95% CI shown in dashed lines. PRG4-deficient samples selected for friction testing are circled. "L" and "R" denotes SF that was obtained from the left and right knee of the same patient. "1" and "2" denotes the 1st and 2nd aspiration of the same knee after a flare reaction to HA. (B) HA MW distribution in N = 5 PRG4-deficient flare-SF samples selected for friction testing and N = 15 normal (NL) SF (* represents p < 0.05).Values are mean ± 95% CI.
View Figure 3
![]()
Table 1: Characteristics of flare patients whose SF was identified as PRG4-deficient and were selected for lubrication testing, and normal (NL) SF from cadaveric donors. * = significantly higher (p < 0.001) compared to normal.
View Table 1
Cartilage-cartilage boundary lubricating ability
Friction was modulated by test lubricant and Tps. In each test lubricant, μstatic,Neq decreased with decreasing Tps and appeared to approach <μkinetic,Neq> as Tps decreased from 120 seconds towards 0 seconds. Values of μstatic,Neq were consistently highest in PBS; values were lower and similar for flare, supplemented, and normal SF. In all test lubricants, values of <μkinetic,Neq> increased only slightly with increasing Tps, with mean values at Tps = 1.2 seconds being on average within 10 ± 1 % (mean ± SD) of values at Tps = 120 seconds. Therefore, as presented previously and for brevity and clarity, <μkinetic,Neq> data are shown at Tps = 1.2 seconds only. Average equilibrium stress for all tests was 0.165 (0.151, 0.178) MPa.
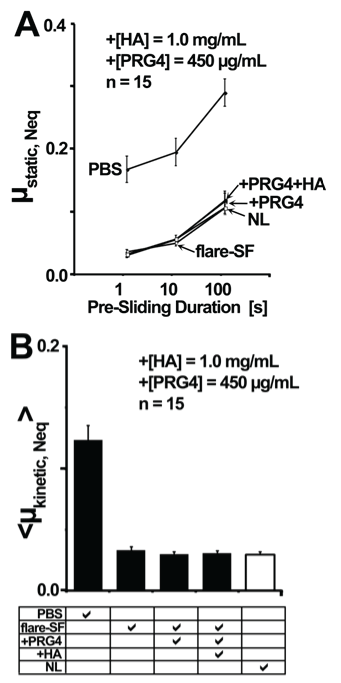
Lubricating ability of flare-SF deficient in PRG4 did not differ from that of normal SF. μstatic,Neqvaried with test lubricant and Tps, with an interaction effect (all p < 0.001) (Figure 4A). Values of μstatic,Neq were similar in flare and normal SF at all Tps (p = 0.83 - 1).<μkinetic,Neq> at Tps = 1.2 seconds also varied with test lubricant (p < 0.001) (Figure 4B). <μkinetic,Neq> for flare-SF was not different than normal SF (0.033 (0.027, 0.039) vs. 0.030(0.025, 0.034), p = 0.70).
.
Figure 4: Effect of HA and PRG4 supplementation on the cartilage boundary lubricating ability of PRG4-deficient flare-SF samples, as determined by in vitro cartilage-on-cartilage friction testing. Two friction coefficients, static (μstatic,Neq) (A) and kinetic (<μkinetic,Neq>; at Tps = 1.2 s) (B) were calculated in PBS (negative control lubricant), PRG4-deficient flare-SF alone, flare-SF plus PRG4, flare-SF plus PRG4 and HA, and normal SF (NL; positive control lubricant). Values are mean ± 95% CI.
View Figure 4
Friction coefficients in flare-SF samples were not altered with PRG4 or PRG4+HA supplementation. Values of μstatic,Neq were similar in flare-SF, flare-SF+PRG4, and flare-SF+PRG4+HA at all Tps (p = 0.72 - 1, Figure 4A). <μkinetic,Neq>at Tps = 1.2 seconds was also similar in flare-SF, flare-SF+PRG4, flare-SF+PRG4+HA, and normal SF (p = 0.76 - 1.0, Figure 4B).
Discussion
These results demonstrate that OA SF can exhibit altered lubricant composition after flare reaction to IA HA injection, but also retain normal cartilage boundary lubricating ability. The range of PRG4 concentrations observed in this study is consistent with previous observations that PRG4 concentrations can vary considerably within both normal donors and chronic OA patients [14]. The normal concentration and partially altered MW of HA observed also agree with previous work [29]. These results provide insight into the molecular basis of cartilage boundary lubricating ability, and suggest that maintenance of HA at physiologically normal concentration and structure (MW) is important for interaction with PRG4, even at diminished levels, and normal joint lubrication.
The PRG4 concentration measured in flare-SF ranged from below normal to super-physiological. There did not appear to be a consistent response in PRG4 concentration to flare reaction, which may suggest that other factors in addition to the flare response are affecting SF PRG4 composition. Joint loading/exercise [30] and inflammation [23,31] are known to affect PRG4 composition and glycosylation in SF, and could have varied between the patients included in this study. PRG4 concentrations have been reported to both increase [32,33] and decrease [14] in chronic OA, suggesting individual baseline levels and responses to external stimuli may vary. In addition, there did not appear to be a consistent response in HA concentrations or HA MW distribution. While HA concentrations measured here are somewhat lower than previous measurements in human SF, the approximately normal concentration and partial shift to lower MW are consistent with previous observations [29]. The small sample size used in this study is a limitation given the known variation in boundary lubricant content in human SF.
The normal human cartilage-cartilage boundary lubrication test used in this study has previously been used for bovine cartilage and SF [4], bovine cartilage and ovine SF [34], and normal human cartilage and SF [14]. The normal human cartilage used in this study was harvested from macroscopically normal areas of distal femurs from donors who were not taking anti-inflammatory medications at time of death. The total protein concentration and volumes of the normal SF used here (Table 1) are within previously reported ranges for normal SF [35], and both aspirated volumes and total protein concentration for flare SF were significantly higher as expected [35]. Coefficients of friction obtained here for normal human SF on normal human cartilage (<μkinetic,Neq> = 0.030) are consistent with previous measurements of bovine cartilage and bovine (<μkinetic,Neq> = 0.025 [4]) and ovine (<μkinetic,Neq> = 0.034 - 0.041 [34]) SF, suggesting that the boundary lubricating function of the human cartilage used here is representative of normal cartilage.
Previous in vitro studies have demonstrated that if composition of either PRG4 or HA is altered it can affect the lubricating ability of whole SF, possibly through alterations in PRG4+HA synergism. Decreased boundary lubricating ability has been observed in equine SF with decreased HA concentration, low HA MW, and increased PRG4 concentration after acute injury; boundary lubricating ability was restored with supplementation with high MW HA (4 MDa) [13]. In human chronic OA SF, decreased boundary lubricating ability was observed in SF with decreased PRG4 concentration, normal HA concentration, and HA MW shifted towards the lower range in all MW ranges from 0.5-6 MDa; boundary lubricating ability was restored with addition of PRG4, and subsequent addition of HA had no additional effect [14]. The flare-SF samples tested here had decreased PRG4, HA concentration similar to normal, and HA MW significantlyshifted towards lower sizes in only two MW ranges: 3.1-6.1 and 0.5-1.1 MDa, suggesting that the flare-SF presented here had sufficient amounts of adequately high MW HA to either provide cartilage lubricating ability, or interact with PRG4 to provide lubricating ability.
The flare-SF used in this study was obtained between 1 - 11 days after injection, which is slightly longer than the a forementioned flare timeline of 24 - 72 hours after injection. The mechanisms of the flare reactions included in this study are unknown, and large SF aspirations, which may indicate underlying inflammatory processes, were not excluded in this study as has been done previously [36]. An acute exercise model in murine knee joints demonstrated that PRG4 concentrations in SF can change very quickly (on the order of minutes) after joint loading [30], and previous modeling work has predicted that PRG4 and HA concentrations would return to equilibrium within hours to days (respectively) after joint lavage [37].
Given that PRG4 and HA are critical contributors to the cartilage boundary lubricating ability of SF, and that PRG4 and HA concentrations in SF seem to be variable, the potential application of PRG4 combined with HA as a biotherapeutic treatment for restoration of altered SF lubricant content is intriguing. While IA HA is generally an effective, well-tolerated treatment that can provide pain relief for OA patients, it does not appear to protect the cartilage surface or act as a disease-modifying agent [38]. Furthermore, recent in vitro evidence has suggested that diminished boundary lubrication may be associated with increased chondrocyte apoptosis, and that hylan G-F20 alone is unable to lubricate and prevent apoptosis as well as SF [39]. PRG4 has been used as an IA therapeutic in rat ACL-transection models of OA and has been observed to reduce cartilage degeneration [40], and also to counteract the additional cartilage damage caused by forced joint exercise [41]. Furthermore, over-expression of PRG4 in mouse articular cartilage protects against development of both age-related and post-traumatic OA [42]. Given that IA PRG4 may stimulate endogenous production of PRG4 [43], its potential as a cartilage-preserving biotherapeutic is promising, especially in conjunction with the pain relief already provided by HA.
In this study, quantification of PRG4 and HA content revealed that post-IA HA flare SF can exhibit decreased PRG4 content after flare reaction to IA injection. Possibly due to 1) sufficient amounts of high MW HA being retained or 2) maintenance of the PRG4+HA interaction, this SF was able to retain normal cartilage boundary lubricating ability. Supplementation with PRG4 and PRG4+HA had no additional effect on lubricating ability. These findings support and extend the concept that the concentration and structure of both PRG4 and HA in SF are important to maintain normal joint lubrication. Previous studies have demonstrated that SF PRG4 and HA can be altered in acute and chronic conditions, and that in vitro restoration of lubricant levels can negate the deleterious effects of lubricant alterations [13,14]. Collectively, this and previous studies suggest that maintaining normal composition of both PRG4 and HA through biotherapeutic treatment may preserve SF lubricating function and therefore contribute to joint preservation and health.
Acknowledgements
The authors thank the University of Calgary Joint Transplantation Program for access to the normal human tissue, Mrs. Sue Miller and Dr. Roman Krawetz for assistance with collection of normal human tissue, the Sports Medicine Centre at the University of Calgary for collection of OA hSF, and the McCaig Institute for Bone and Joint Health. This work was supported by funding from the National Science and Engineering Research Council of Canada, Canadian Arthritis Network, Alberta Innovates Technology Futures, Alberta Innovates Health Solutions OA Team Grant, Faculty of Kinesiology and Schulich School of Engineering’s Center for Bioengineering Research and Education at the University of Calgary.
Ethical Statement
The authors have no conflicts of interest to disclose.
Funding Sources
National Science and Engineering Research Council of Canada, Canadian Arthritis Network, Alberta Innovates Technology Futures, Alberta Innovates Health Solutions, Faculty of Kinesiology and Schulich School of Engineering's Center for Bioengineering Research and Education at the University of Calgary.
References
-
Ateshian GA, Mow VC (2005) Friction, lubrication, and wear of articular cartilage and diarthrodial joints. In: Mow VC, Huiskes R Basic Orthopaedic Biomechanics and Mechano-Biology. (3rd edn) Philadelphia: Lippincott Williams &Wilkins 447-494.
-
Morrell KC, Hodge WA, Krebs DE, Mann RW (2005) Corroboration of in vivo cartilage pressures with implications for synovial joint tribology and osteoarthritis causation. Proc Natl Acad Sci USA 102: 14819-14824.
-
Mcutchen CW (1966) Boundary lubrication by synovial fluid: demonstration and possible osmotic explanation. Fed Proc 25: 1061-1068.
-
Schmidt TA, Sah RL (2007) Effect of synovial fluid on boundary lubrication of articular cartilage. Osteoarthritis Cartilage 15: 35-47.
-
Schmidt TA, Gastelum NS, Nguyen QT, Schumacher BL, Sah RL (2007) Boundary lubrication of articular cartilage: role of synovial fluid constituents. Arthritis Rheum 56: 882-891.
-
Jay GD, Torres JR, Rhee DK, Helminen HJ, Hytinnen MM, et al. (2007) Association between friction and wear in diarthrodial joints lacking lubricin. Arthritis Rheum 56: 3662-3669.
-
Ikegawa S, Sano M, Koshizuka Y, Nakamura Y (2000) Isolation, characterization and mapping of the mouse and human PRG4 (proteoglycan 4) genes. Cytogenet Cell Genet 90: 291-297.
-
Swann DA, Silver FH, Slayter HS, Stafford W, Shore E (1985) The molecular structure and lubricating activity of lubricin isolated from bovine and human synovial fluids. Biochem J 225: 195-201.
-
Schumacher BL, Hughes CE, Kuettner KE, Caterson B, Aydelotte MB (1999) Immunodetection and partial cDNA sequence of the proteoglycan, superficial zone protein, synthesized by cells lining synovial joints. J Orthop Res 17: 110-120.
-
Marcelino J, Carpten JD, Suwairi WM, Gutierrez OM, Schwartz S, et al. (1999) CACP, encoding a secreted proteoglycan, is mutated in camptodactyly-arthropathy-coxa vara-pericarditis syndrome. Nat Genet 23: 319-322.
-
Elsaid KA, Fleming BC, Oksendahl HL, Machan JT, Fadale PD, et al. (2008) Decreased lubricin concentrations and markers of joint inflammation in the synovial fluid of patients with anterior cruciate ligament injury. Arthritis Rheum 58:1707-1715.
-
Fam H, Bryant JT, Kontopoulou M (2007) Rheological properties of synovial fluids. Biorheology 44: 59-74.
-
Antonacci JM, Schmidt TA, Serventi LA, Cai MZ, Shu YL, et al. (2012) Effects of equine joint injury on boundary lubrication of articular cartilage by synovial fluid: role of hyaluronan. Arthritis Rheum 64: 2917-2926.
-
Ludwig TE, McAllister JR, Lun V, Wiley JP, Schmidt TA (2012) Diminished cartilage lubricating ability of human osteoarthritic synovial fluid deficient in proteoglycan 4: Restoration through proteoglycan 4 supplementation. Arthritis Rheum 64: 3963-3971.
-
Conrozier T, Chevalier X (2008) Long-term experience with hylan GF-20 in the treatment of knee osteoarthritis. Expert OpinPharmacother 9: 1797-1804.
-
Waddell DD (2007) Viscosupplementation with hyaluronans for osteoarthritis of the knee: clinical efficacy and economic implications. Drugs Aging 24: 629-642.
-
Brandt KD, Smith GN Jr, Simon LS (2000) Intraarticular injection of hyaluronan as treatment for knee osteoarthritis: what is the evidence? Arthritis Rheum 43: 1192-1203.
-
Owen DS Jr (2001) Diagnostic Tests and PRocedures: Aspiration and Injection of Joints and Soft Tissues. In: Ruddy S, Harris ED, Sledge CB Kelley's Textbook of Rheumatology. (6th Edition) Philadelphia: WB Saunders.
-
Chevalier X, Jerosch J, Goupille P, van Dijk N, Luyten FP, et al. (2010) Single, intra-articular treatment with 6 ml hylan G-F 20 in patients with symptomatic primary osteoarthritis of the knee: a randomised, multicentre, double-blind, placebo controlled trial. Ann Rheum Dis 69: 113-119.
-
Goldberg VM, Coutts RD (2004) Pseudoseptic reactions to hylanviscosupplementation: diagnosis and treatment. ClinOrthopRelatRes : 130-137.
-
Rutjes AW, Jüni P, da Costa BR, Trelle S, Nüesch E, et al. (2012) Viscosupplementation for osteoarthritis of the knee: a systematic review and meta-analysis. Ann Intern Med 157: 180-191.
-
Marino AA, Waddell DD, Kolomytkin OV, Pruett S, Sadasivan KK, et al. (2006) Assessment of immunologic mechanisms for flare reactions to Synvisc. ClinOrthopRelat Res 442: 187-194.
-
Conrozier T, Mathieu P, Vignon E, Piperno M, Rinaudo M (2012) Differences in the osteoarthritic synovial fluid composition and rheology between patients with or without flare-up: a pilot study. Clin Exp Rheumatol 30: 729-734.
-
Jay GD, Elsaid KA, Zack J, Robinson K, Trespalacios F, et al. (2004) Lubricating ability of aspirated synovial fluid from emergency department patients with knee joint synovitis. J Rheumatol 31: 557-564.
-
Schmidt TA, Plaas AH, Sandy JD (2009) Disulfide-bonded multimers of proteoglycan 4 PRG4 are present in normal synovial fluids. Biochim Biophys Acta 1790: 375-384.
-
Estrella RP, Whitelock JM, Packer NH, Karlsson NG (2010) The glycosylation of human synovial lubricin: implications for its role in inflammation. Biochem J 429: 359-367.
-
Lee HG, Cowman MK (1994) An agarose gel electrophoretic method for analysis of hyaluronan molecular weight distribution. Anal Biochem 219: 278-287.
-
McDonald J (2009) Handbook of Biologial Statistics (2nd ed). Baltimore, Maryland: Sparky House Publishing.
-
Dunn S, Kolomytkin OV, Marino AA (2009) Pathophysiology of osteoarthritis: evidence against the viscoelastic theory. Pathobiology 76: 322-328.
-
Abusara Z, Krawetz R, Steele B, DuVall M, Schmidt T, et al. (2013) Muscular loading of joints triggers cellular secretion of PRG4 into the joint fluid. J Biomech 46: 1225-1230.
-
Ali L, Jin C, Karlsson NG (2012) Glycoproteomics of Lubricin-Implication of Important Biological Glyco- and Peptide-Epitopes in Synovial Fluid. In: Lemmey AB Rheumatoid Arthritis - Etiology, Consequences and Co-Morbidities.
-
Neu CP, Reddi AH, Komvopoulos K, Schmid TM, Di Cesare PE (2010) Increased friction coefficient and superficial zone protein expression in patients with advanced osteoarthritis. Arthritis Rheum 62: 2680-2687.
-
Ballard BL, Antonacci JM, Temple-Wong MM, Hui AY, Schumacher BL, et al. (2012) Effect of tibial plateau fracture on lubrication function and composition of synovial fluid. J Bone Joint Surg Am 94: e64.
-
Barton KI, Ludwig TE, Achari Y, Shrive NG, Frank CB, et al. (2013) Characterization of proteoglycan 4 and hyaluronan composition and lubrication function of ovine synovial fluid following knee surgery. J Orthop Res 31: 1549-1554.
-
Koopman WJ, Moreland LW, eds. (2005) Arthritis and allied conditions: a textbook of rheumatology 15th ed: Lippincott Williams &Wilkins.
-
Bagga H, Burkhardt D, Sambrook P, March L (2006) Longterm effects of intraarticular hyaluronan on synovial fluid in osteoarthritis of the knee. J Rheumatol 33: 946-950.
-
Blewis ME, Nugent-Derfus GE, Schmidt TA, Schumacher BL, Sah RL (2007) A model of synovial fluid lubricant composition in normal and injured joints. Eur Cell Mater 13: 26-39.
-
Balazs EA (2009) Therapeutic uses of hyaluronan. Structural Chemistry 20: 341-349.
-
Waller KA, Zhang LX, Fleming BC, Jay GD (2012) Preventing friction-induced chondrocyte apoptosis: comparison of human synovial fluid and hylan G-F 20. J Rheumatol 39: 1473-1480.
-
Flannery CR, Zollner R, Corcoran C, Jones AR, Root A, et al. (2009) Prevention of cartilage degeneration in a rat model of osteoarthritis by intraarticular treatment with recombinant lubricin. Arthritis Rheum 60: 840-847.
-
Elsaid KA, Zhang L, Waller K,Tofte J, Teeple E, et al. (2012) The impact of forced joint exercise on lubricin biosynthesis from articular cartilage following ACL transection and intra-articular lubricin's effect in exercised joints following ACL transection. Osteoarthritis Cartilage 20: 940-948.
-
Ruan MZ, Erez A, Guse K, Dawson B, Bertin T, et al. (2013) Proteoglycan 4 expression protects against the development of osteoarthritis. Sci Transl Med 5: 176.
-
Jay GD, Fleming BC, Watkins BA, McHugh KA, Anderson SC, et al. (2010) Prevention of Cartilage Degeneration and Restoration of Chondroprotection by Lubricin Tribo supplementation in the Rat Following Anterior Cruciate Ligament Transection. Arthritis Rheum 62:2382-2391.