International Journal of Respiratory and Pulmonary Medicine
Drainage using Chest Tubes Smaller than 20 French is Suitable for Patients with Thoracic Empyema
Ryo Matsunuma1*, Kazumasa Kase1, Nobuhiro Asai2, Satoshi Watanabe3, Yuko Waseda4, Norihiro Kaneko5, Masahiro Aoshima5, Masaki Fujimura6 and Kazuo Kasahara3
1Department of Respiratory Medicine, Komatsu Municipal Hospital, Japan
2Department of Respiratory Medicine, Aichi Medical University, Japan
3Department of Respiratory Medicine, Kanazawa University, Japan
4Department of Respiratory Medicine, Japan Community Health Care Organization Kanazawa Hospital, Japan
5Department of Respiratory Medicine, Kameda Medical Center, Japan
6Department of Respiratory Medicine, National Hospital Organization Nanao Hospital, Japan
*Corresponding author: Ryo Matsunuma, Department of Respiratory Medicine, Komatsu Municipal Hospital, Mukaimoto-orimachi, Komatsu city 60 ho, Ishikawa, Postal code: 923-8650, Japan, Fax: +81-761-21-7155, E-mail: manutsuma@gmail.com
Int J Respir Pulm Med, IJRPM-3-058, (Volume 3, Issue 4), Original Article; ISSN: 2378-3516
Received: July 29, 2016 | Accepted: September 28, 2016 | Published: October 01, 2016
Citation: Matsunuma R, Kase K, Asai N, Watanabe S, Waseda Y, et al. (2016) Drainage using Chest Tubes Smaller than 20 French is Suitable for Patients with Thoracic Empyema. Int J Respir Pulm Med 3:058. 10.23937/2378-3516/1410058
Copyright: © 2016 Matsunuma R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Patients with thoracic empyema occasionally require chest tubes for drainage. Several studies have reported that smaller chest tubes are effective and cause fewer complications. The aim of this study was to investigate the outcomes of patients with empyema who underwent drainage using a smaller-size chest tube.
Methods: We retrospectively reviewed all patients at 2 institutions with thoracic empyema. We performed both univariate and multivariate analysis to examine the relation between tube size and the incidence of complications.
Results: The mean chest-tube size was 22 ± 3.0 Fr. Six patients (12%) died within 90 days of tube insertion. Three patients (5.9%) required surgery. A total of 31 complications were recorded during the duration of drainage. On univariate analysis, a chest-tube size either greater than 20 Fr or greater than 22 Fr was significantly associated with the incidence of complications (OR, 4.90; 95% CI, 1.45-16.6; and OR, 4.75; 95% CI, 1.37-16.5, respectively). On multivariate analysis, a chest drain larger than 20 Fr (OR, 4.51; 95% CI, 1.27-16.3; P = 0.020) was significantly related to the incidence of adverse events during chest drainage. Treatment failure and death were not significantly increased in the population of patients with chest tubes smaller than 20 Fr, and the duration of drainage and of hospital admission was not extended in these patients.
Conclusions: Chest tubes smaller than 20 Fr may reduce complications included pain in patients with thoracic empyema. Furthermore, outcomes of chest drainage are similar for tubes larger than and smaller than 20 Fr. Further prospective studies examining various chest-tube sizes are required to validate our results.
Keywords
Complications, Chest pain, Drainage, Empyema, Tube size
Background
Pleural infection was first described by Hippocrates in 500 BC [1]. Worldwide, the management of patients with pleural infection varies widely and approaches differ among physicians [2]. The British Thoracic Society (BTS) recommends prompt pleural drainage for patients with frankly purulent or turbid/cloudy pleural fluid on sampling [1,3]. Sahn, et al. asserted that the need for pleural space drainage was due to clinical factors, including prolonged pneumonia symptoms, comorbidities, failure to respond to antibiotic therapy, and the presence of anaerobic organisms [4]. Moreover, there is no consensus regarding the optimal size of drainage tubes for patients with thoracic empyema [1,5]. Standard chest tubes (26-32 Fr) are often placed without ultrasound or CT guidance by thoracic surgeons for treating complicated parapneumonic effusion and empyema [6]. In addition, drainage tubes larger than 20 Fr have been recommended for patients presenting with thoracic empyema in Japan [7]. On the other hand, small-bore catheters (8-14 Fr), which are utilized more frequently today, can be placed and provided good outcome under ultrasound or CT guidance [8-13]. Moreover, Rahman, et al. suggested that in treating pleural infection, smaller, guide wire inserted chest tubes caused substantially less pain than larger, blunt dissection-inserted tubes without impairing clinical outcomes [5]. Furthermore, Light stated that the advantages of the smaller tube were that it was less painful to the patient and was easier to insert [14]. Therefore, smaller size of chest tube might be more appropriate than larger one, however, we do not determine which size should be used for patients with thoracic empyema. The aim of this study was to examine the relationship between the size of drainage tube, outcome, pain and complications in patients with thoracic empyema.
Methods
Patient characteristics
We retrospectively reviewed all patients with thoracic empyema who were admitted to the Kameda Medical Center, a 800-bed community hospital in Kamogawa City, Chiba, Japan, and the Komatsu Municipal Hospital, a 364-bed community hospital in Komatsu City, Ishikawa, Japan, from January 2009 to November 2014. Patients who received a chest tube for drainage were included. Patients with tuberculous pleuritis, malignant pleuritis, or unknown pleuritis were excluded. This study protocol was approved by the Institutional Review Board of Komatsu Municipal Hospital.
Definitions
Empyema was defined as pleural effusion exhibiting at least one of the following: 1) pleural fluid with a positive culture for bacterial infection; 2) macroscopic purulence; and 3) pH < 7.2 and clinical evidence of infection, including fever, cough, sputum, and elevated white blood cell (WBC) or C-reactive protein (CRP) [4]. Severity of empyema was categorized used by American College of Chest Physicians (ACCP) consensus statement [15]. Complications were recorded during drainage. Treatment failure was defined as death related to empyema or the need for reinsertion of a chest tube or surgical treatment. The success was defined as patients who removed drainage tube and discharged from the hospital. Deaths occurring within 90 days were also recorded.
Data collection
Clinical data were collected by review of electronic medical records. We collected the following patient data: chief complaint, especially included chest pain, age, gender, Eastern Cooperative Oncology Group (ECOG) Performance status (PS) [16], comorbidities, laboratory findings, characteristics of pleural effusion, including gross appearance, cell count, pH, glucose, protein, LDH (lactase dehydrogenase), gram stain, and culture findings. For assessing comorbidities, we employed the Charlson Comorbidity Index (CCI) [17], which has been validated in population-based studies for estimating mortality risk. Data related to tube thoracostomy were also recorded, including chest tube size, complications of drainage, duration of drainage, and the administration of intrapleural urokinase. The methods of insertion (Blunt dissection or Seldinger method) were researched. Chest computed tomography (CT) was researched the diameter of chest fluid and existence of multilocular fluid. Antibiotic therapy, including the type of antimicrobial and duration of treatment, was also documented.
Statistical analysis
Univariate analysis was performed the relationship between incidence of complications and various candidate predictors. Continuous variables of the factors were divided into two categories. White blood cell (WBC), hemoglobin (Hb) and hematocrit (Ht) set at the value that demarcated the normal and abnormal ranges, whereas albumin (Alb), serum lactase dehydrogenase (LDH), sodium (Na), C-reactive protein (CRP) and LDH of pleural effusion was based on the mean value. Drainage tube sizes, which were less than 14 Fr, 16 Fr, 18 Fr, 20 Fr, 22 Fr, 24Fr, 28 Fr and 30Fr, were also divided into two categories to examine the relationship with tube size and complications. Then, to identify independent factors the incidence of complications, multivariate logistic regression analysis with several factors included significantly different items by univariate analysis. Continuous variables were analyzed using the Student's t-test for normally distributed data or the Welch's test for nonparametric data. Nominal variables were analyzed using a chi-square test. Differences were assumed to be significant when the P-value was < 0.05. All analyses were performed using SPSS for Windows version 20.00 (SPSS Inc; Chicago, IL, US).
Results
Patient characteristics
Table 1 shows the characteristics of the patients. The study sample included 51 patients, of whom the majority (n = 41; 80%) were male. The mean (± standard deviation) age was 68 ± 15 years. The most frequent symptom was fever (41%), the other symptoms were chest pain (27%), dyspnea (16%) and cough (5.9%). Hypertension (51%), cerebrovascular disease (31%), diabetes mellitus (22%), and chronic heart failure (18%) were the most prevalent underlying conditions. The CCI was 1.4 ± 1.2. The severity of empyema was shown in table 2. There were no patients in Category 1 and 2. On the other hands, Category 3 and 4 were 11, 40 patients, respectively.
![]()
Table 1: Characteristics and comorbidities of patients with empyema.
View Table 1
Chest tube outcomes
Tube size, duration of drainage, and the frequency of death and complications related to chest drainage are described for all patients in table 2. The mean chest tube size was 22 ± 3.0 Fr. Six patients (12%) died within 90 days. Three patients (5.9%) needed surgery because medical treatment could not cure. Total 31 complications were recorded. Notably suffering from pain (51%) caused by the chest tube and tube decanulation (12%) mostly occurred. Tube obstruction occurred in only one patient (2.0%). We then analysed the relationship between the duration of hospital stay and tube size using Spearmann's rank-correlation coefficient. The coefficient of correlation and P value was 0.186, 0.191, respectively. All patients received blunt dissection method and only 1 patient received on the ventral side.
![]()
Table 2: Characteristics of and complications related to chest drainage.
View Table 2
Analysis of the relationship between various candidate predictors and incidence of complications
We performed univariate analysis of the relationship between incidence of complications and various candidate predictors (Table 3). Under 10 g/dL of hemoglobin and under 35% of hematocrit were significantly reduced the incidence of adverse events in patients who received chest drainage. (Odds ratio (OR): 95% confidence interval (CI); 0.26: 0.078-0.84, 0.22: 0.056-0.88, respectively) Furthermore, more than 20 Fr and 22 Fr chest tube significantly associated adverse events. (OR: 95% CI; 4.90: 1.45- 16.6 and 4.75: 1.37-16.5, respectively) Less 1 of performance status (PS) were not significantly but tend to be caused complications. The examination of pleural effusion and the analysis of chest computed tomography (CT) were not related the incidence of complications.
![]()
Table 3: Univariate analysis of factors related to complications.
View Table 3
Multivariate logistic regression analysis were then performed using Hb ≤ 10 g/dL, chest drain ≥ 22 Fr and chest drain ≥ 20 Fr. Chest drain ≥ 20 Fr (OR = 4.51, 95% CI: 1.27- 16.3, p = 0.020) was significantly related to complications caused by chest drain (Table 4).
![]()
Table 4: Multivariate logistic regression analysis of factors related to complications.
View Table 4
We then discussed outcome of using smaller than 20 Fr chest tube. The outcome was defined as treatment failure. Treatment failure and death was not significantly increased. Moreover, duration of drainage and hospital admission was not extended using smaller than 20 Fr chest tube (Table 5). Similarly, the proportion alive and not requiring surgery was described used by Kaplan- Mayer method in figure 1. Using smaller than 20 Fr and more than 22 Fr chest tube was not significantly difference the duration of incidence of treatment failure (γ2 = 0.24, p = 0.63).
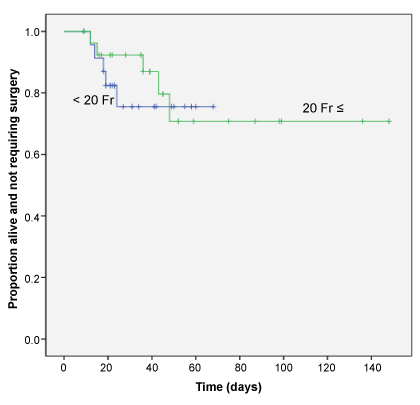
.
Figure 1 The proportion of patients with empyema who were alive and not requiring surgery on any given admission day is described using the Kaplan-Meier method. Patients are divided into those with chest tubes smaller than 20 Fr and those with tubes larger than 20 Fr. There was no significant difference in the duration of treatment or the incidence of treatment failure (γ2 = 0.24; P = 0.63).
View Figure 1
![]()
Table 5: Treatment failure, death, and duration of admission.
View Table 5
Discussion
The present study showed that tubes more than 22 Fr caused complications included pain in patients with thoracic empyema. Furthermore, outcomes of chest drainage were similar both more than 22 Fr and smaller than 20 Fr chest tube. These results indicate that physician and surgeon should use tubes smaller than 20 Fr for the patients with thoracic empyema who receive drainage. This finding is consistent with the results of several studies. Sahn, et al. reported higher overall pain scores in patients treated with larger-size chest tubes. The authors concluded that the major advantage of small-bore (8-14 Fr) catheters was improved patient tolerance and the avoidance of major complications [4]. Similarly, Cafarotti, et al. suggested that with regard to pain and tolerability, the small-bore chest drains (12 Fr) were generally considered more comfortable by patients and induced less pain [9]. The BTS has also recommended the use of flexible small bore catheters (10-14 Fr) which appear less traumatic for insertion and more comfortable for the patient [18]. On the other hand, Inaba, et al. found that patients with trauma who were treated with small (28-32 Fr) and large (36-40 Fr) chest tubes reported similar mean VAS scores [19]. However, the lower boundary of size for the small chest tube (28- 32 Fr) in this study was larger than 20 Fr. Therefore, chest tubes less 20 Fr may have reduce adverse events and less pain.
Further, the treatment success rate was 78% less than 20 Fr chest tubes in this study. Several studies have cited success rates ranging from 63% to 82% in patients with empyem [20-22]. Mandal and Thadepalli reported a 93% cure rate for patients treated by chest tube drainage alone [23]. On the other hand, the success rate was only 53% in patients treated with 24-28 Fr chest tubes in a study by Huang, et al. [24]. The success rate was low because causes of pleural effusion, examined by Huang, et al., may have included trauma, surgical intervention, esophageal diseases, and malignant diseases. Moreover, published studies have reported rates of mortality due to empyema ranging from 1% to 61% [23,25-27]. In this study, the mortality rate at 90 days in the patients who received less than 20 Fr chest tubes was 13%, which was consistent with the findings from prior studies.
Concerning safety, the life-threatening complications of large-bore chest tubes have been widely documented with incidence rates ranging from 0.2% to 6% [1,28]. Complications include organ penetration leading to lung laceration, heart and great vessel puncture, diaphragmatic perforation, and spleen/liver injury [28]. Liu, et al. suggested that the most common complication was iatrogenic pneumothorax, which has been reported to occur at a frequency of 2.5% to 5.4% [20]. Fortunately, none of the patients in the present studies experienced these life-threatening complications. However, decannulation occurred when the chest tube was unexpectedly removed in patients who still needed drainage. In our study, decannulation was the 2nd most frequent complication (12%). A study by Horsley, et al. found that decannulation occurred in 6% of chest tube cases [21]. Patients experiencing decannulation require reinsertion of another chest tube, which increases the risk of additional life-threatening complications. Therefore, patients undergoing chest tube drainage should be monitored frequently for chest tube placement and secure pinning of the chest tube is required.
Chest tube obstruction occurred in one patient (4.3%) who had a 12 Fr chest tube in this study. In several studies, chest drain obstruction occurred in 7.5% to 12.9% of patients drained by smaller chest tubes (10-14 Fr) [9,21,28]. This may indicate that chest tubes smaller than 14 Fr more frequently cause obstruction as compared to larger tubes. Therefore, the BTS recommends regular flushing with 30 ml of saline every 6 hours via a three-way tap [1]. While Horsley, et al. and Cafarotti, et al. suggested that small-bore wire-guided drains (< 20F) should not be routinely used for treating empyema because small-bore wire-guided drains are less effective for resolving empyema and have limited success rates and a high propensity for blockage in other studies [9,20]. However, chest tubes ≤ 20 Fr in our study caused less pain. Further, the success rate in this group was similar to that in the large-size chest tube group as well as the success rates cited in other studies. Therefore, we conclude that chest tubes smaller than 20 Fr are acceptable for patients with empyema.
Our study has a few limitations. First, it was a retrospective analysis of a very small population. Second, we did not use a visual analogue scale (VAS) to evaluate pain, which may have provided a more appropriate method for measuring the degree of pain. However, we could not discuss the degree of pain because use of a VAS was not indicated in our medical records. Third, several studies as well as the BTS guidelines have recommended the use of 10-14 Fr chest tubes in patients with empyema, whereas the chest tube sizes examined in our study were comparatively larger. To validate our findings, further prospective large-scale studies are required in order to determine the most appropriate chest tube size.
Conclusions
Chest tubes smaller than 20 Fr may reduce complications included pain in patients with thoracic empyema. Furthermore, outcomes of chest drainage were similar both more than 20 Fr and less 20 Fr chest tube. These results indicate that physician and surgeon might use tubes less 20 Fr for the patients with thoracic empyema who receive drainage. However, this study was a retrospective analysis of a very small population. Therefore, further prospective studies examining various chest tube sizes are required.
Acknowledgements
We are also grateful for the diligent and thorough critical reading of our manuscript by Mr. John Wocher, Executive Vice President and Director, International Affairs/International Patient Services, Kameda Medical Center (Japan) and Yoshihido Ohkuni, department of respirology, Kameda Medical Center (Japan).
Conflicts of Interest
There are no conflicts of interest.
References
-
Davies CW, Gleeson FV, Davies RJ (2003) BTS guidelines for the management of pleural infection. Thorax 58: 18-28.
-
Berger HA, Morganroth ML (1990) Immediate drainage is not required for all patients with complicated parapneumonic effusions. Chest 97: 731-735.
-
Hooper C, Lee YC, Maskell N; BTS Pleural Guideline Group (2010) Investigation of a unilateral pleural effusion in adults: British Thoracic Society Pleural Disease Guideline 2010. Thorax 65: 4-17.
-
Sahn SA (2007) Diagnosis and management of parapneumonic effusions and empyema. Clin Infect Dis 45: 1480-1486.
-
Rahman NM, Maskell NA, Davies CW, Hedley EL, Nunn AJ, et al. (2010) The relationship between chest tube size and clinical outcome in pleural infection. Chest 137: 536-543.
-
Ferguson AD, Prescott RJ, Selkon JB, Watson D, Swinburn CR (1996) The clinical course and management of thoracic empyema. QJM 89: 285-289.
-
Tezuka K, Yamamoto S, Endo S (2011) The surgical treatment based on pathological condition in patients with empyema. Kokyu 30: 230-236.
-
Chen CH, Chen W, Chen HJ, Yu YH, Lin YC, et al. (2009) Transthoracic ultrasonography in predicting the outcome of small-bore catheter drainage in empyemas or complicated parapneumonic effusions. Ultrasound Med Biol 35: 1468-1474.
-
Cafarotti S, Dall'Armi V, Cusumano G, Margaritora S, Meacci E, et al. (2011) Small-bore wire-guided chest drains: safety, tolerability, and effectiveness in pneumothorax, malignant effusions, and pleural empyema. J Thorac Cardiovasc Surg 141: 683-687.
-
Keeling AN, Leong S, Logan PM, Lee MJ (2008) Empyema and effusion: outcome of image-guided small-bore catheter drainage. Cardiovasc Intervent Radiol 31: 135-141.
-
Merriam MA, Cronan JJ, Dorfman GS, Lambiase RE, Haas RA (1988) Radiographically guided percutaneous catheter drainage of pleural fluid collections. AJR Am J Roentgenol 151: 1113-1116.
-
vanSonnenberg E, Nakamoto SK, Mueller PR, Casola G, Neff CC, et al. (1984) CT- and ultrasound-guided catheter drainage of empyemas after chest-tube failure. Radiology 151: 349-353.
-
Crouch JD, Keagy BA, Delany DJ (1987) "Pigtail" catheter drainage in thoracic surgery. Am Rev Respir Dis 136: 174-175.
-
Light RW (2006) Parapneumonic effusions and empyema. Proc Am Thorac Soc 3: 75-80.
-
Colice GL, Curtis A, Deslauriers J, Heffner J, Light R, et al. (2000) Medical and surgical treatment of parapneumonic effusions : An evidence-based guideline. Chest 118: 1158-1171.
-
Zubrod C, Sheiderman M, Frei E, Brindley C, Lennard Gold G, et al. (1960) Appraisal of methods for the study of chemotherapy in man: Comparative therapeutic trial of nitrogen mustard and triethylene thiophosphoramide. J Chronic Dis 11: 7-33.
-
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40: 373-383.
-
Davies HE, Davies RJ, Davies CW; BTS Pleural Disease Guideline Group (2010) Management of pleural infection in adults: British Thoracic Society Pleural Disease Guideline 2010. Thorax 65: 41-53.
-
Inaba K, Lustenberger T, Recinos G, Georgiou C, Velmahos GC, et al. (2012) Does size matter? A prospective analysis of 28-32 versus 36-40 French chest tube size in trauma. J Trauma Acute Care Surg 72: 422-427.
-
Liu YH, Lin YC, Liang SJ, Tu CY, Chen CH, et al. (2010) Ultrasound-guided pigtail catheters for drainage of various pleural diseases. Am J Emerg Med 28: 915-921.
-
Horsley A, Jones L, White J, Henry M (2006) Efficacy and complications of small-bore, wire-guided chest drains. Chest 130: 1857-1863.
-
Bar I, Stav D, Fink G, Peer A, Lazarovitch T, et al. (2010) Thoracic empyema in high-risk patients: conservative management or surgery? Asian Cardiovasc Thorac Ann 18: 337-343.
-
Mandal AK, Thadepalli H (1987) Treatment of spontaneous bacterial empyema thoracis. J Thorac Cardiovasc Surg 94: 414-418.
-
Huang HC, Chang HY, Chen CW, Lee CH, Hsiue TR (1999) Predicting factors for outcome of tube thoracostomy in complicated parapneumonic effusion for empyema. Chest 115: 751-756.
-
Lee-Chiong TL Jr, Matthay RA (1996) Current diagnostic methods and medical management of thoracic empyemas. Chest Surg Clin N Am 6: 419-438.
-
Bartlett JG, Finegold SM (1974) Anaerobic infections of the lung and pleural space. Am Rev Respir Dis 110: 56-77.
-
Jess P, Brynitz S, Friis Møller A (1984) Mortality in thoracic empyema. Scand J Thorac Cardiovasc Surg 18: 85-87.
-
Baldt MM, Bankier AA, Germann PS, Pöschl GP, Skrbensky GT, et al. (1995) Complications after emergency tube thoracostomy: Assessment with CT. Radiology 195: 539-543.





