International Journal of Respiratory and Pulmonary Medicine
Cross-Antigenicity of Paragonimus Westermani Egg Yolk Cells with Antibody against Surfactant Protein-D (SP-D) in Pulmonary Necrotizing Granuloma in a Native Japanese Woman
Yuji Ohtsuki1*, Shinji Fujioka2, Masashi Uomoto2, Yasuki Hachisuka2, Masataka Korenaga3, Atsushi Kurabayashi4, Ichiro Murakami4 and Mutsuo Furihata4
1Department of Diagnostic Pathology, Matsuyama-shimin Hospital, Japan
2Department of Thoracic Surgery, Matsuyama-shimin Hospital, Japan
3Department of Parasitology, Kochi University, Japan
4Department of Pathology, Kochi University, Japan
*Corresponding author: Yuji Ohtsuki, MD, PhD, Department of Diagnostic Pathology, Matsuyama-shimin Hospital, Matsuyama, Ehime 790-0067, Japan, Tel: 81-89-943-1151, Fax: 81-89-947-0026, E-mail: y.ohtsuki@matsuyama-shimin-hsp.or.jp
Int J Respir Pulm Med, IJRPM-3-057, (Volume 3, Issue 3), Case Report; ISSN: 2378-3516
Received: September 01, 2016 | Accepted: September 15, 2016 | Published: September 19, 2016
Citation: Ohtsuki Y, Fujioka S, Uomoto M, Hachisuka Y, Korenaga M, et al. (2016) Cross-Antigenicity of Paragonimus Westermani Egg Yolk Cells with Antibody against Surfactant Protein-D (SP-D) in Pulmonary Necrotizing Granuloma in a Native Japanese Woman. Int J Respir Pulm Med 3:057. 10.23937/2378-3516/1410057
Copyright: © 2016 Ohtsuki Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A native Japanese woman in her 60s complained of hemosputum and had an irregular nodular shadow in her chest radiograph. The nodule was surgically removed, and the histopathological findings of the pulmonary lesion showed a necrotizing granuloma with central irregular geographic necrosis associated with parasite eggs. The results of the enzyme-linked immunosorbent assay of her serum confirmed Paragonimus westermani (Pw) infection as the cause, owing to her history of ingesting uncooked wild boar meat. Interestingly, immunohistochemical results demonstrated that the Pw egg yolk cells could react with antibody against surfactant protein D (SP-D). Antibody against SP-D can bind to fucose residues of Schistosoma mansoni. Therefore, this common antigenicity of Pw eggs with antibody against SP-D was very interesting in terms of fucose residue binding. This is the first report to demonstrate the cross-antigenicity of Pw eggs, probably yolk cells, with antibody against SP-D in pulmonary necrotizing granuloma. Further investigation is needed to clarify this cross-antigenicity more precisely.
Keywords
Necrotizing granuloma, Paragonimus westermani, Egg yolk cells, Surfactant protein D, Immunohistochemistry
Introduction
Pulmonary paragonimiasis, a sequela of Paragonimus westermani (Pw) infection, has been identified not only in native Japanese individuals, but also in immigrants from China, Thailand, and Korea, in Japan [1]. Total 322 cases were identified in Japan among the native Japanese individuals from 2001 to 2012. Histogenic analysis of 22 out of the 67 female patients identified ingestion of uncooked wild boar meat as the cause [1]. Pw eggs are usually difficult to detect in pulmonary granulomas, as demonstrated by the stated 11.7% detection rate (52 out of 443 cases) [1]. Additionally, its clinical differentiation from lung cancer is difficult, because fluorodeoxyglucose-positron emission tomography/computed tomography scans yield false-positive results for these parasite granulomas [2]. In another case, its differentiation from tuberculosis was difficult [3]. Furthermore, an antibody against surfactant protein D (SP-D) has been found to react with the fucose residues of Schistosoma mansoni [4] and with Helicobacter pylori-infected foveolar epithelial cells of the stomach [5].
In the present study, antibody against SP-D was found to immunohistochemically react with Pw eggs, especially egg yolk cells, in a case of pulmonary necrotizing granuloma.
Case Report
A native Japanese woman in her 60s complained of hemosputum, with a chest radiograph indicating an irregular nodular shadow. Her peripheral blood cell count was increased, and eosinophils accounted for more than 20% of this population, indicating a high serum IgE level. Chest computed tomography scan revealed an irregular, somewhat nodular shadow on the left lower lobe, which was extirpated. After surgery, her abnormal laboratory data gradually normalized. An enzyme-linked immunosorbent assay-based serum analysis supported Pw infection, probably caused by the consumption of Pw eggs through ingestion of uncooked wild boar meat.
Histopathology
The cut surface of the resected left lower lobe revealed a parasitic granuloma of a maximum size of 26 mm that was associated with a cystic cavity (Figure 1a, arrow). Histopathological findings of the granuloma included a central irregular geographic necrosis (Figure 1b) and an epithelioid cell rim (Figure 1c) with interspersed multinucleated giant cells. Parasite eggs were also scattered throughout the granuloma (Figure 1c, arrows). Within the granuloma, the macrophages had phagocytized eggs to form foreign body-type multinucleated giant cells (Figure 1c, inset), although no phagocytosis was observed in the necrotic areas. Various types of eggs were found, including those harboring empty, amorphous eosinophilic materials and many egg yolk cells (Figure 1c).
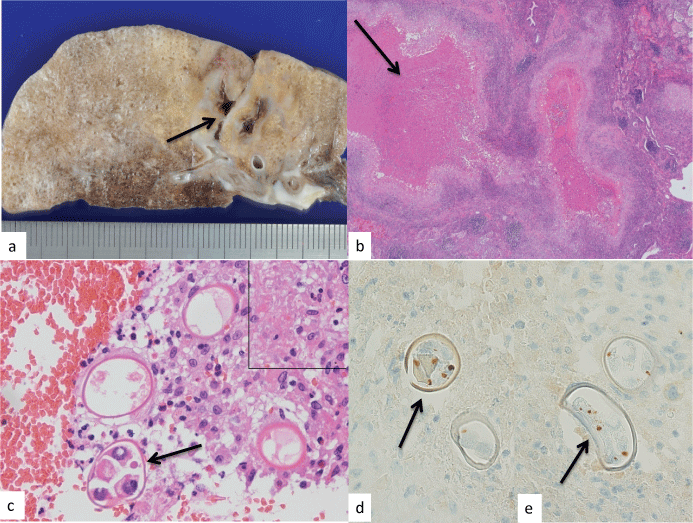
.
Figure 1 (a) The cut surface of the resected lung lesion revealed a cavity (arrow); (b) Geographic central necrotic areas were observed in the granuloma using hematoxylin and eosin (H & E) staining, magnification × 40; (c) The granuloma also had an epithelioid rim and scattered parasite eggs containing several yolk cells (H&E, magnification × 400) that had been phagocytized by macrophages (inset, magnification × 400); (d,e) Positive intracellular SP-D antibody staining revealed its punctate distribution within the egg yolk cells (arrows; SP-D immunostaining, magnification × 400).
View Figure 1
In the bronchioles, many neutrophils and eosinophils were packed in their cavities, intermingled with parasite eggs. However, sputum examination did not reveal the parasite eggs. Besides granulomas, many eosinophils had densely infiltrated the surrounding peribronchiolar and adjoining alveolar spaces, to cause eosinophilic pneumonia, associated with Charcot-Leyden crystals on occasions, as previously stated [2]. Organizing pneumonia and obstructive pneumonia patterns were also focally detected [6].
Immunohistochemistry
An immunohistochemical study was also performed. For this analysis antibodies against surfactant protein-A (SP-A) (clone PE10, 1 : 50 dilution with no pretreatment; DAKO, Kyoto, Japan) and SP-D (clone 10H11 [7], 1 : 1600 dilution after pretreatment with 0.01 M citric buffer at 121°C for 15 min; Yamasa, Chousi, Japan) were used. Immunohistochemical staining was performed using a Ventana BenchMark GX device (Roche Diagnostic Co. Ltd., Tokyo, Japan), according to the manufacturer’s protocol.
Antibody against SP-D was purified through the immunosorbent assay of human SP-D; then, antibody against SP-D producing clone 10H11 [7] was obtained.
Interestingly, the eggs reacted with antibody against SP-D (Figure 1d, Figure 1e, arrows), but not with antibody against SP-A. The positive reaction pattern showed two to four punctate positive signals in the egg yolk cells (Figure 1d, Figure 1e, arrows). In addition, some alveolar epithelial cells were also positive for antibodies against to both SP-A and SP-D.
Discussion
Regarding the distribution of SP-D in the human body, many epithelial cells have been previously reported to exhibit a positive response to a rabbit anti-mouse SP-D antibody [8]. However, the antibody used herein reacts only with type II alveolar cells [9]. No other examined tissues or cells have yielded positive results so far [9].
The positive signals obtained from Pw eggs in the present study seemingly corresponded to the yolk egg cells, based on the number of signals and their location within the eggs. Not all eggs may have reacted positively owing to the degeneration of the eggs in the necrotic areas. In fact, empty eggs were often detected and internal structures of many eggs were obscure.
Recently, SP-D has been found to bind to terminal α (1,3)-linked fucose residues on S. mansoni [5]. In S. mansoni, exposed fucose-rich glycoconjugate structures exist on the surface of cercariae [10] and appear to play a role in the immune responses associated with schistosomiasis [11,12]. Moreover, exposed SP-D has been observed on the surface of gastric mucosal epithelial cells during H. pylori infection [5].
In conclusion, this cross-antigenicity of Pw eggs with antibody against SP-D was interesting. Further evaluation, particularly of Pw egg yolk cells, is needed. This is the first report in the literature demonstrating the cross-antigenicity of PW egg yolk cells with antibody against SP-D in a pulmonary necrotizing granuloma.
Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
-
Nagayasu E, Yoshida A, Hombu A, Horii Y, Maruyama H (2015) Paragonimiasis in Japan: a twelve-year retrospective case review (2001-2012). Intern Med 54: 179-186.
-
Boland JM, Vaszar LT, Jones JL, Mathison BA, Rovzar MA, et al. (2011) Pleuropulmonary infection by Paragonimus westermani in the United States:rare cause of eosinophilic pneumonia after ingestion of live crabs. Am J Surg Pathol 35: 707-713.
-
Osaki T, Takama T, Nakagawa M, Oyama T (2007) Pulmonary paragonimus westermani with false positive fluorodeoxyglucose positron emission tomography mimicking primary lung cancer. Gen Thorac Cardiovasc Surg 55: 470-472.
-
Lall M, Sahni AK, Rajput AK (2013) Pleuropulmonary paragonimiasis: mimicker of tuberculosis. Pathog Glob Health 107: 40-42.
-
van de Wetering JK, van Remoortere A, Vaandrager AB, Batenburg JJ, van Golde LM, et al. (2004) Surfactant protein D binding to terminal alpha1-3-linked fucose residues and to Schistosoma mansoni. Am J Respir Cell Mol Biol 31: 565-572.
-
Murray E, Khamri W, Walker MM, Eggleton P, Moran AP, et al. (2002) Expression of surfactant protein D in the human gastric mucosa and during Helicobacter pylori infection. Infec Immun 70: 1481-1487.
-
Inoue T, Matsuura E, Nagata A, Ogasawara Y, Hattori A, et al. (1994) Enzyme-linked immunosorbent assay for human pulmonary surfactant protein D. J Immunol Methods 173: 157-164.
-
Stahlman MT, Gray ME, Hull WM, Whitsett JA (2002) Immunolocalization of surfactant protein-D (SP-D) in human fetal, newborn, and adult tissues. J Histochem Cytochem 50: 651-660.
-
Ohtsuki Y, Nakanishi N, Fujita J, Yoshinouchi T, Kobayashi M, et al. (2007) Immunohistochemical distribution of SP-D, compared with that of SP-A and KL-6, in interstitial pneumonias. Med Mol Morphol 40: 163-167.
-
Jang-Lee J, Curwen RS, Ashton PD, Tissot B, Mathieson W, et al. (2007) Glycomics analysis of Schistosoma mansoni egg and cercarial secretions. Mol Cell Proteomics 6: 1485-1499.
-
Hokke CH, Deelder AM (2001) Schistosome glycoconjugates in host-parasite interplay. Glycoconj J 18: 573-587.
-
Cummings RD, Nyame AK (1999) Schistosome glysoconjugates. Biochim Biophys Acta 1455: 363-374.





