International Journal of Respiratory and Pulmonary Medicine
Diffuse Alveolar Hemorrhage as a Initial Manifestation of Henoch-Schönlein Purpura in Adults
Carmen Carrasco Cubero1* and Chamizo Carmona2
1Service of Rheumatology, Hospital Infanta Cristina, Spain
2Service of Rheumatology, Hospital de Mérida, Spain
*Corresponding author: Carmen Carrasco Cubero, Service of Rheumatology, Hospital Infanta Cristina, Avenida Damián Téllez Lafuente, s/n. CP: 06010, Badajoz, Spain, E-mail: mcarrascocubero@gmail.com
Int J Respir Pulm Med, IJRPM-3-052, (Volume 3, Issue 3), Case Report; ISSN: 2378-3516
Received: May 17, 2016 | Accepted: August 19, 2016 | Published: August 22, 2016
Citation: Cubero CC, Carmona C (2016) Diffuse Alveolar Hemorrhage as a Initial Manifestation of Henoch-Schönlein Purpura in Adults. Int J Respir Pulm Med 3:052. 10.23937/2378-3516/1410052
Copyright: © 2016 Cubero CC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Summary
Henoch-Schönlein purpura (HSP) is a systemic vasculitis that affects small-sized vessels, characterized by non-thrombocytopenic purpura, along with other clinical manifestations such as arthritis, abdominal pain, gastrointestinal bleeding and renal disease. Although the presence of subclinical alterations of pulmonary function has been noticed, the appearance of diffuse alveolar hemorrhage (DAH) is a rather an unusual symptom. We report a case of HSP with DAH as initial manifestation.
Keywords
Henoch-Schönlein purpura (HSP), Diffuse alveolar hemorrhage (DAH)
Introduction
Henoch-Schönlein purpura (HSP) is a systemic vasculitis that affects small-sized vessels characterized by non-thrombocytopenic purpura [1]. The pathogenesis of this disease is related to IgA deposition in vessel walls [2,3], so since the last International Chapel Hill Consensus Conference Nomenclature of Vasculitides (2012) renamed "Ig A vasculitis".
It is the most common form of acute vasculitis in children, although it can be also observed in adults [4], where the incidence is approximately 1.2/million [5]. It has been recognized that men have a predisposition for it.
The most characteristic clinical manifestations include purpura (100%), arthritis (82%), abdominal pain (63%), gastrointestinal bleeding (33%) and renal disease (40%) [2].
DAH is an uncommon manifestation, although some cases have been reviewed literature [3,6-10]. Some authors have noticed the existence of pulmonary dysfunction in patients with HSP who do not show any pulmonary symptom [1,11].
We present a case in which the HSP in an adult appears with haemoptysis in a previously asymptomatic patient.
Case Report
A 54-year-old male, with a medical history of long-term controlled arterial hypertension, who had severe aortic valve failure which required surgery with mechanical prosthesis, and an ascending aorta plasty in 2003. He started with warfarin.
In August 2007 he had right occipital hematoma open to ventricles. During his hospital stay, he also had complications such as an episode of thrombosis on the mechanical valvular prosthesis, requiring emerging aortic thrombectomy and subsequently secondary septic shock, bilateral pneumonia and cardiac tamponade. In August 2008 he was admitted with endocarditis in mechanical aortic prosthesis, and was referred for surgical evaluation due to hemodynamic instability, requiring prosthesis extraction and replacement.
In March 2009 he was admitted to the Respiratory Unit with haemoptysis. Oral anticoagulation was stopped and replaced with low molecular weight heparin. The INR value during this episode of hemorrhage was 3.5.
The patient went on to occasionally show a small amount of hemoptoic sputum, after his discharge from the Respiratory Unit, adding the appearance of skin lesions in lower limbs, abdomen, and back in the last days, and he was admitted for a new evaluation.
He did not show signs of arthritis, nor dyspnea or fever, neither constitutional symptoms nor previous infection of upper respiratory tract. No oral or genital aphtha, or parestesia, myalgia or muscle weakness. He had no gastrointestinal disorders, abdominal pain or symptoms of renal disease.
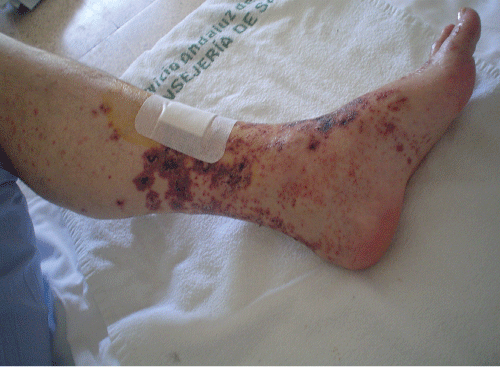
Physical examination showed the existence of mild edemas with fovea in tarsal and pretibial areas and non-painful purpuric lesions located in his lower limbs, abdomen, thorax and upper limbs, which did not disappear when pressure was applied (Figure 1). Bibasilar crackle heard on auscultation of the lungs. Examination findings were otherwise unremarkable.
Laboratory test results revealed the following: hemoglobin, 10.2 gr/dL; total leukocytes, 7770/nL; 349 × 109/L platelets, erythrocyte sedimentation rate, 44 mm/h; serum creatinine 1.27 mg/dl, urea 48 mg/dl, normal electrolyte levels. Proteinogram compatible with chronic inflammatory process, with a normal C3, C4 complement level and high Ig A.
The urine sediment contained microscopic hematuria and low level microalbuminuria. Antinuclear antibodies, antineutrophil cytoplasmic antibodies, cryoglobulins, anti glomerular basement membrane antibodies, circulating immune complexes, anticardiolipins and beta 2 microglobulins were all undetectable.
Serology for HCV, HBV, HIV and other viruses were negative; the bacilloscopy and sputum cytology were also negative.
The echocardiographic study showed aneurismatic dilatation of the aorta root with normal functioning prosthesis, together with dilatation of the right cavities with pulmonary hypertension pattern.
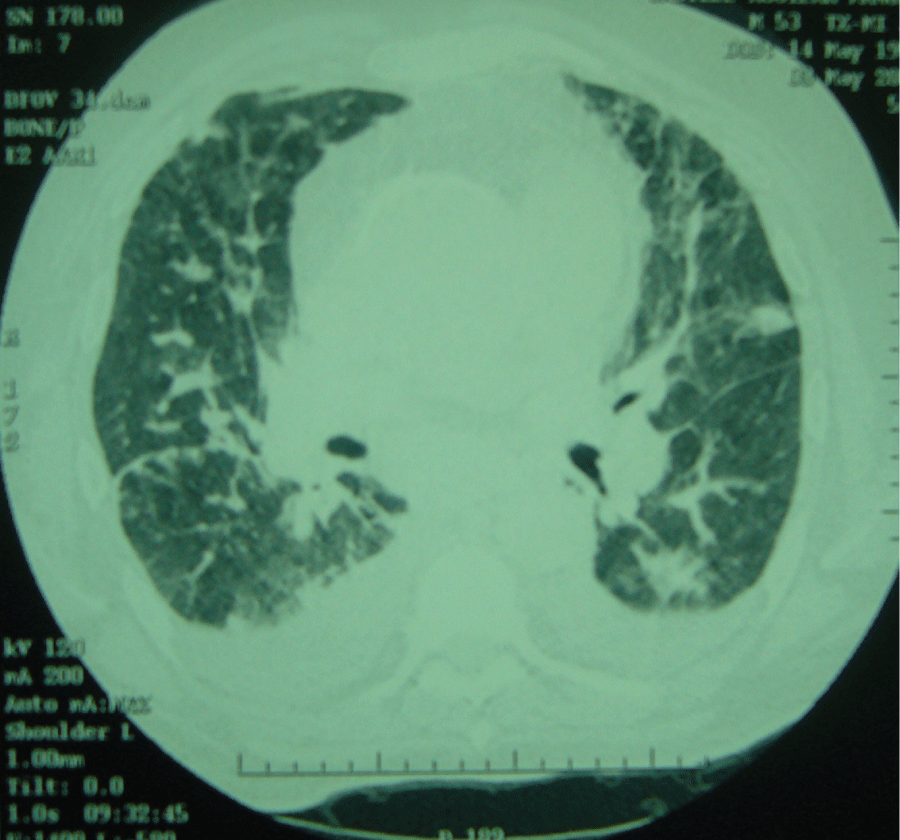
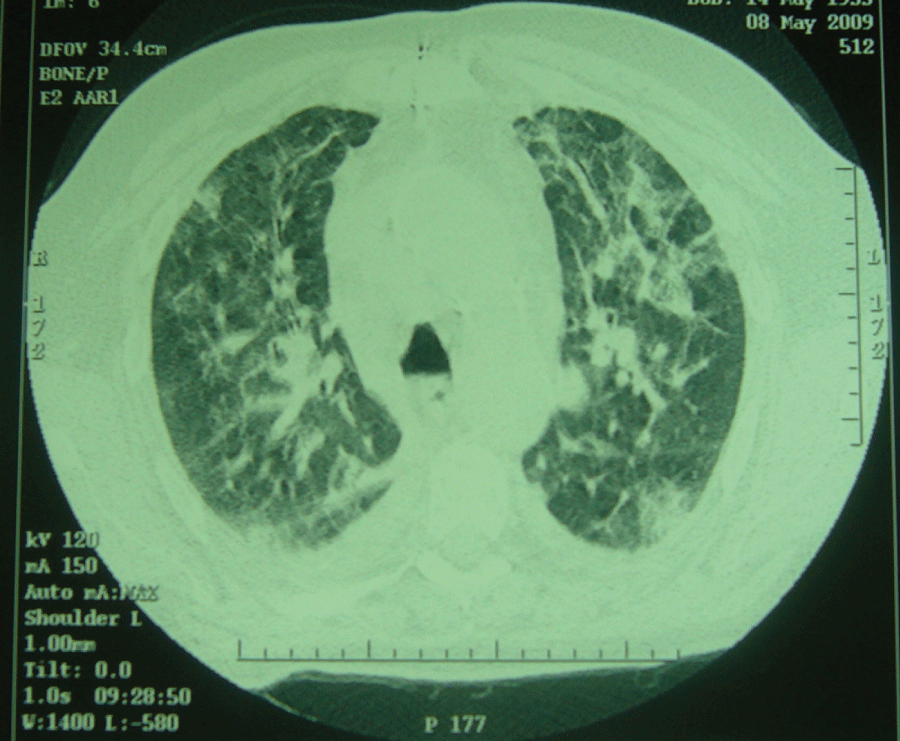
Chest radiography revealed a predominant pulmonary vascularisation associated with interstitial pattern. Computed tomography of the chest showed clear bilateral pleural effusion and diffuse infiltrate, consisting of patches of ground-glass areas, other peribronchial vascular areas, and septal enlargement in the upper lobes (Figure 2 and Figure 3).
.
Figure 2: Computed tomography of the chest reveals bilateral pleural effusion and diffuse reticular nodular densities associated with ground-glass attenuation.
View Figure 2
.
Figure 3: Chest computed tomography scan showing diffuse ground-glass opacity in the lung fields.
View Figure 3
Pulmonary function test were normal, including carbon monoxide diffusion test. In the bronchoscopy we found vasculitis like lesions in nasal mucosa and increasing vascularisation in bronchial mucosa. The bronchoalveolar lavage it was found 18% of macrophages with 60% of hemosiderophages. Cytopathology and transbronchial biopsy turned out to be normal.
Skin biopsy was compatible with leukocytoclastic vasculitis in small vessels, with granular and subendothelial deposition of IgA and IgG in the immunofluorescence examination.
The patient was diagnosed as having HSP. Intravenous methylprednisolone, 1000 mg/d, was administered for five days, followed by prednisone, a dose of 1 mg/Kg weight/day; resulting in gradual recovery of the pulmonary symptoms, and skin alterations. The control HRCT turned out to be normal.
Discussion
HSP is a systemic necrotizing vasculitis that affects small vessels, characterized by vessels deposition of IgA-containing immune complexes. It is more common in children (50% of patients usually are less than 5 years old), than adults [1,2].
In both groups, clinical manifestations are similar, although adults with HSP are more likely than children to develop significant renal involvement, including end-stage renal disease [2].
The most frequent clinical manifestations in adults are purpura, arthritis, abdominal pain, gastrointestinal bleeding and renal disease. Purpura is present in 100% of cases, followed by arthritis which appears in 82%, abdominal pain in 63%, gastrointestinal bleeding in 33% and finally, glomerulonephritis in 40% of cases. There are other clinical manifestations that show up less often such as skin, musculoskeletal, neurologic, pulmonary, cardiac (carditis) and ocular (anterior uveitis, episcleritis) manifestations [1].
Pulmonary involvement in the HSP is extremely rare (2.4-5%) and includes mainly hemorrhage and/or interstitial disease [5].
DAH predominantly affects older male children and adults with HSP. The occurrence of DAH in HSP is rare and the reported prevalence ranged from 0.8% to 5%. DAH occurred variably after the diagnosis of HSP, ranging from 2 days to 18 years [7]. There are few cases described in which the HSP appears with pulmonary symptoms [2,8-14]. Hemoptysis (75%), drop in hemoglobin (74%), and chest infiltrates (94%) were the most common clinical findings in DAH cases [7]. Pleural effusions were also observed in 16.7% of patients with pulmonary hemorrhage, as happened in our patient.
In X-rays bilateral infiltrates or pulmonary opacities usually appear, and these findings are confirmed with the computed axial tomography scan.
In this setting, the findings of IgA deposits in skin biopsy, together with the absence of other clinical, serologic or histologic features of other more common potencial causes, (drugs, ANCA-vasculitis, lupus) secured the diagnosis of Henoch-Schönlein purpura in a patient with DAH.
DAH was frequently severe and 50% of the patients required mechanical ventilation. DAH has been traditionally associated with a high mortality rate (27.6%) and morbidity [7]. Usually treatment of patients with diffuse alveolar hemorrhage includes high steroids regimen and immunosuppressives drugs. Cyclophosphamide and pulse methylprednisolone for DAH was associated with better outcomes, particularly in patients who were already receiving steroids at the time of DAH. Systemic recurrences (27.7%) and recurrences of DAH (8.3%) were frequent [7].
Our patient had a clinical and radiological recurrery with the use of methylprednisolone bolus followed by a dose of 1 mg/kg/day prednisone, with no need to use azathioprine or cyclophosphamide.
Conclusion
HSP should be part of the differential diagnosis of DAH in adults. Early diagnosis and treatment can be vital for better patient prognosis.
Current protocols use pulse methylprednisolone and cyclophosphamide.
References
-
Blanco R, Martínez-Taboada VM, Rodríguez-Valverde V, García-Fuentes M, González-Gay MA (1997) Henoch- Schölein purpura in adulthood and childhood: two different expresions of the same syndrome. Arthritis Rheum 40: 859-864.
-
Goel SS, Langford CA (2009) A 72-year-old man with a purpuric rash. Cleve Clin J Med 76: 353-360.
-
Cream JJ, Gumpel JM, Peachey RD (1970) Schönlein-Henoch purpura in the adult. A study of 77 adults with anaphylactoid or Schönlein-Henoch purpura. Q J Med 39: 461-484.
-
Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, et al. (2013) 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum 65: 1-11.
-
Nadrous HF, Yu AC, Specks U, Ryu JH (2004) Pulmonary involvement in Henoch-Schönlein purpura. Mayo Clin Proc 79: 1151-1157.
-
Carreras L, Poveda R, Bas J, Mestre M, Rama I, et al. (2002) Goodpasture syndrome during the course of a Schönlein-Henoch purpura. Am J Kidney Dis 39: E21.
-
Rajagopale S, Shobha V, Devaraj U, D'Souza G, Garg I (2013) Pulmonary Hemorrhage in Henoch-Schonlein Purpura: Case Report and Systematic Review of the English Literature. Semin Arthritis Rheum 42: 391-400.
-
Agraharkar M, Gokhale S, Le L, Rajaraman S, Campbell GA (2000) Cardiopulmonary manifestations of Henoch-Schönlein purpura. Am J Kidney Dis 35: 319-322.
-
Usui K, Ochiai T, Muto R, Abe I, Aikawa M, et al. (2007) Diffuse pulmonary hemorrhage as a fatal complication of Schönlein-Henoch purpura. J Dermatol 34: 705-708.
-
Ros S, Martínez Ara J, García de Miguel MA, Olea T, de Alvaro F (2004) Familial Henoch-Schönlein purpura manifested with lung hemorrhage. Nefrologia 24: 499-502.
-
de la Prada Alvarez FJ, Prados Gallardo AM, Tugores Vázquez A, Uriol Rivera M, Saus Sarrias C, et al. (2005) Shönlein-Henoch nephritis complicated with pulmonary renal syndrome. An Med Interna 22: 441-444.
-
Stienstra Y, Fijen JW, Tervaert JW, Dankbaar H, Zijlstra JG, et al. (2000) Pulmonary hemorrhage with respiratory insufficiency in Henoch- Schoenlein purpura. Ned Tijdschr Geneeskd 144: 617-622.
-
Nadrous HF, Yu AC, Specks U, Ryu JH (2004) Pulmonary involvement in Henoch-Schönlein purpura. Mayo Clin Proc 79: 1151-1157.
-
Vats KR, Vats A, Kim Y, Dassenko D, Sinaiko AR (1999) Henoch-Schönlein purpura and pulmonary hemorrhage: a report and literature review. Pediatr Nephrol 13: 530-534.