International Journal of Respiratory and Pulmonary Medicine
An Unusual Case of Left-Sided Massive Biliothorax
María José Soler-Sempere1*, Nuria Vicente-Ibarra2 and Laura Heredia-Oliva3
1Respiratory Medicine, Hospital General Universitario de Elche, Spain
2Section of Cardiology, Hospital General Universitario de Elche, Spain
3Section of Pathological Anatomy, Hospital General Universitario de Elche, Spain
*Corresponding author: María José Soler-Sempere, Respiratory Medicine, Hospital General Universitario de Elche, Camí de L'Almassera 03203 Elche, Spain, Tel/Fax: 966616708, E-mail: majosoler1@hotmail.com
Int J Respir Pulm Med, IJRPM-2-034, (Volume 2, Issue 4), Case Report; ISSN: 2378-3516
Received: June 17, 2015 | Accepted: October 28, 2015 | Published: October 31, 2015
Citation: Soler-Sempere M, Vicente-Ibarra N, Heredia-Oliva L. (2015) An Unusual Case of Left-Sided Massive Biliothorax. Int J Respir Pulm Med 2:034. 10.23937/2378-3516/1410034
Copyright: © 2015 Soler-Sempere M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Biliothorax and pleurobiliary fistula are rare complications secondary to biliary tree diseases. We report the case of a patient with left massive pleural effusion secondary to biliothorax without fistula or ascites. To our knowledge, this is the first case to be described with left side biliothorax.
Keywords
Biliothorax, Pleurobiliary fistula, ERCP, Ampullary tumor
Introduction
Biliothorax is a rare clinical entity associated with biliary tree diseases. Biliary contamination in the thorax can affect the pleural space secondary to pleurobiliary fistula (PBF), the pulmonary parenchyma or the bronchial tree (bronchobiliary fistula) [1] and the diagnosis is based on the demonstration of bile in plerural fluid [2]. We report a clinical case of massive left biliothorax without ascites or evidence of fistula in the imaging studies. To our knowledge, this is the first case to be described with left side biliothorax.
Case history
An 83-year-old woman with a past medical history of chronic ischaemic heart disease, cerebrovascular disease, intrathoracic goitier and cholecystectomy for symptomatic cholelithiasis 8 years ago, was admitted to our hospital because of progressive dyspnea and left hemithorax pain. At hospital admission, pertinent physical findings demonstrated a dyspneic woman with a general health deterioration, a blood pressure of 145/87 mm Hg, pulse of 99 beats/min and temperature of 37.9°C. The left inferior hemithorax was dull to percussion and no breath sounds were appreciated; abdominal examination did not showed ascites or masses.
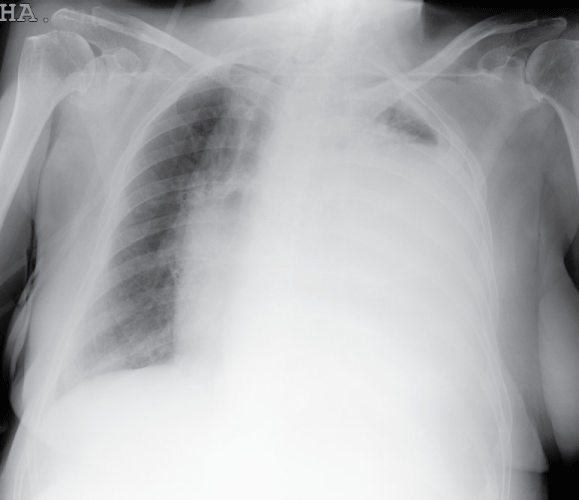
Chest radiograph showed the presence of a left pleural effusion occupying two thirds of the hemithorax (Figure1). A thoracocentesis was performed and 800 cc of green-colored pleural fluid was drained. Biochemical analysis of pleural fluid showed a pH level of 7.28, WBC count of 130/mm3 with 65% of neutrophils, glucose of 144 mg/dL, total protein of 5.8 g/dL (total protein level in serum of 6.2 g/dL), LDH of 2551 U/L (LDH level in serum of 443 U/L), bilirubin 5.8 mg /dl (plasma 0.4 mg/dl) and amylase 47 U/L. The bilirubin ratio pleural fluid/serum was 14.5.
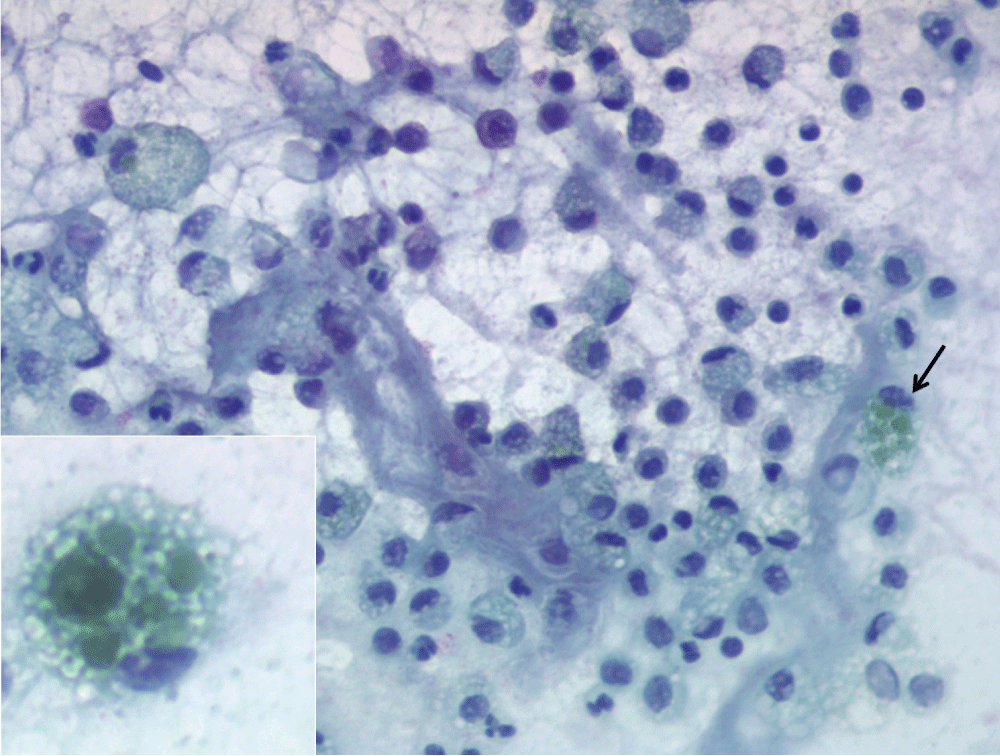
Aerobic and anaerobic cultures of pleural fluid had no growth, and cytology showed an inflammatory exudate with pigmented foamy histiocytes, neutrophils and absence of malignant cells (Figure 2).
.
Figure 2: Pap staining (PAP) (20X). Cytological showing numerous lymphocytes and foamy histiocytes, some of them loaded with pigment (arrow). Lower left corner. PAP (40X) Detail of a green pigment laden histiocyte.
View Figure 2
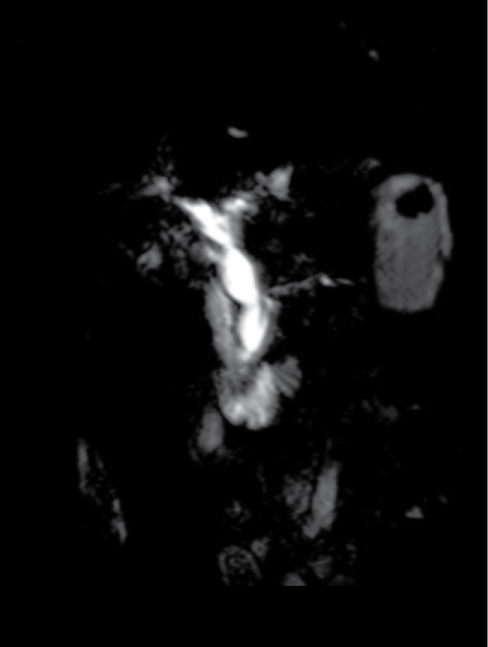
Biochemical parameters in blood test were: AST: 17 U/L, ALT: 20 U/L, gamma-GT: 260 U/L, alcaline phosphatase: 209 U/L, reactive protein C: 125.4 mg/L. A CT scan and MRI cholangiography showed a dilatation of intrahepatic and extrahepatic bile ducts with a maximum bile duct diameter of 20 mm. The extrahepatic bile duct had a progressive stenosis in the distal portion. A hypodense nodule of 8 mm of diameter was shown in the duodenum wall near the papilla of Vater, suggestive of intraluminal neoproliferative process (Figure 3).
.
Figure 3: MRI cholangiography: A dilatation of intrahepatic and extrahepatic bile ducts with a bile duct diameter of 20 mm.
View Figure 3
An endoscopic retrograde cholangiopancreatography (ERCP) was then performed to rule out a pleurobiliary fistula. ERCP showed a bulky papillary structure and common bile duct dilatation without filling defects inside. No fistula was located by cholangiography.
Taking into account these findings and the high comorbidity of the patient an endoscopic sphincterotomy and biliary stent placement was performed to improve biliary drainage. However, despite the measures taken, the patient had an unfavourable outcome and died few days after.
Discussion
Biliothorax is a rare complication of biliary tract disease. The diagnosis is based on the demonstration of bile in pleural fluid with a ratio between the pleural bilirubin and the serum was greater than 1 [2].
The main cause of biliothorax, described in the few reported cases in the literature, is the presence of PBF secondary to choledocolithiasis [3], trauma [4] or iatrogenic procedures [5]. Other rare causes described in the literature are hepatic parasitic disease [6], radiofrequency ablation of liver tumors [7] and biliary stent migration [8].
A high index of suspicion, prompt diagnosis and treatment is crucial for the management of PBF. In our case, the presence of green-colored pleural fluid with elevated levels of bilirubin confirmed the diagnosis of biliothorax. The MRcholangiopancreatography showed an important dilatation of the intrahepatic and extrahepatic bile ducts and a hypodense nodule in the Vater papilla sugessting the presence of ampulloma.
A key differential aspect in our case was the unusual presentation with left side pleural effusion; furthermore the absence of a recent invasive procedure in the bile duct or a history of trauma in our patient. Most biliothorax described in the literature are mainly located in the right side due to a transdiaphragmatic passage of bile into pleural cavity either directly by the presence of a fistula or bile passage from peritoneal to pleural cavity secondary to the presence of right hemidiaphragmatic defects [2]. The absence of ascites in our case and the presence of a dilated intrahepatic bile duct lead us to think that there was a BPF from the left bile duct to left pleural cavity, despite a BPF was not shown in the ERCP. Moreover, we have not found references in the literatre to spontaneous biliothorax in the context of neoplastic processes of the bile duct (cholangiocarcinoma and ampullary adenoma).
In our case, biliothorax was the first manifestation of a bililary neoplasm, what it could be considered as unusual clinical presentation of an ampullary tumor. However, in the literature some cases of biliothorax of idiopathic etiology had been published [9].
Initial management of biliothorax consists of a combination of pleural fluid drainage, location of the PBF and biliary decompression to facilitate BPF closure by endoscopic procedures such as ERCP or percutaneous cholangiography. If endoscopic and/or radiological procedures are not effective, thoracoscopic and pleural decortication is a reasonable surgical [10]. Given the high surgical risk for the patient, a conservative management was done. Biliary decompression by endoscopic retrograde cannulation of the common bile duct with an endoprosthesis has been proven to be an effective treatment compared with surgery in some cases [11]. The advanced age and high comorbidity of the patient limited the other therapeutic efforts in our case.
In summary, biliothorax remains a rare and unusually left entity, not always evidenced the presence of a fistula biliopleural and may be the first manifestation of a neoplastic process. No guidelines exist for the treatment of biliothorax; therefore, management should be tailored to individual patient needs. Endoscopic therapies to relieve biliary obstruction and achieve drainage could be indicate in high-risk surgical patients.
References
-
Saraya T, Light RW, Takizawa H, Goto H (2013) Black pleural effusion. Am J Med 126: 641.
-
Cordoba Lopez A, MonterrubioVillar J, Bueno Alvarez-Arenas I (2008) Biliothorax unrelated to fistula: a rare complication in gallbladder disease. Arch Bronconeumol 44: 396-397.
-
Cunningham LW, Grobman M, Paz HL, Hanlon CA, Promisloff RA (1990) Cholecystopleural fistula with cholelithiasis presenting as a right pleural effusion. Chest 97: 751-752.
-
Bhattacharya A, Gupta R, Mittal BR (2006) Traumatic biliopleural communication detected by hepatobiliary scintigraphy. Clin Nucl Med 31: 170-171.
-
Al-Qahtani HH (2011) Biliopleural fistula with cholethorax. A rare complication of percutaneous transhepatic biliary drainage. Saudi Med J 32: 1189-1192.
-
Amir-Jahed AK, Sadrieh M, Farpour A, Azar H, Namdaran F (1972) Thoracobilia: a surgical complication of hepatic echinococcosis and amebiasis. Ann Thorac Surg 14: 198-205.
-
Lewis JR, Te HS, Gehlbach B, Oto A, Chennat J, et al. (2009) A case of biliopleural fistula in a patient with hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 6: 248-251.
-
Dasmahapatra HK, Pepper JR (1988) Bronchopleurobiliary fistula. A complication of intrahepatic biliary stent migration. Chest 94: 874-875.
-
Seong MK, Kim SM, Yoo SH, Park WR, An JY, et al. (2011) A Case of Cholethorax Developed by Unknown Cause. Tuberc Respir Dis 70: 261-265.
-
Crnjac A, Pivec V, Ivanecz A (2013) Thoracobiliary fistulas: literature review and a case report of fistula closure with omentummajus. Radiol Oncol 47: 77-85.
-
Singh B, Moodley J, Sheik-Gafoor MH, Dhooma N, Reddi A (2002) Conservative management of thoracobiliary fistula. Ann Thorac Surg 73: 1088-1091.