International Journal of Respiratory and Pulmonary Medicine
The Differential Diagnosis of Asthma: Endobronchial Pulmonary Mesenchymoma
Mehmet Unlu* , Pinar Cimen, Nuran Katgi and Salih Zeki Guclu
Izmir Training and Research Hospital for Thoracic Medicine and Surgery, Pulmonary Division, Turkey
*Corresponding author: Mehmet UNLU, Izmir Training and Research Hospital for Thoracic Medicine and Surgery, Pulmonary Division,35210 Yenisehir, Izmir-Turkey, Tel: 90-232-4333333, Fax: 90-232-4587262, E-mail: lidokain21@hotmail.com
Int J Respir Pulm Med, IJRPM-1-005, (Volume 1, Issue 1), Case Report; ISSN: 2378-3516
Received: November 10, 2014 | Accepted: December 03, 2014 | Published: December 06, 2014
Citation: Unlu M, Cimen P, Katgi N, Guclu SZ (2014) The Differential Diagnosis of Asthma: Endobronchial Pulmonary Mesenchymoma. Int J Respir Pulm Med 1:005. 10.23937/2378-3516/1410005
Copyright: © 2014 Unlu M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A variety of airway tumours are reported to manifest with symptoms similar to those of asthma and may cause delays in exact diagnosis because of the increase in size at a slow rate. Pulmonary endobronchial mesenchymoma is one of the slow growing tumours which is associated with symptoms or signs of airway obstruction mimicking asthma. So, patients with chronic symptoms suggestive of asthma, poor response to asthma medications, and frequent exacerbations should be evaluated for endobronchial mesenchymoma.
Keywords
Asthma; Differential diagnosis; Pulmonary mesenchymoma
Introduction
Pulmonary mesenchymomas are the most common form of benign lung tumours which accounts for approximately 8% of all lung neoplasmsand 77% of all benign lung tumors [1,2]. The vast majority of pulmonary mesenchymomas are located peripherally within the lungs (>90%) and only up to 10% of such lesions represent with endobronchial location [3,4]. However, because of this rare endobronchial form of presentationpatients may be misdiagnosed as asthma and may be tried to be treatedmistakenly with bronchodilators for a long time.
Here we describe a case of pulmonary mesenchymoma mimicking features of asthma and treated with asthma medication for more than ten years before admission to our clinic.
Case Presentation
A 51-year-old female presented to the emergency department of our hospital with progressive dyspnea and dry cough. She had been diagnosed with asthma 12 years ago. Since that time, she had been prescribed bronchodilators, but her symptoms were only decreased at the beginning of the treatment. She had history of an increase especially in shortness of breath and cough affecting her especially since last four years. She was a housewife and had no work-life history. There was no past history of a chronic disease. Her smoking history was 4 packs/year and she had been quitted smoking immediately after diagnosis of asthma. She had never consumed illicit drugs and had no allergies.
On admission, the patient had a respiratory rate of 22 breaths/minute, a blood pressure of 110/70 mmHg, and a heart rate of about 140 beats/minute. Examination of the blood samples revealed only anemia (haemoglobin: 10.4g/dl). Number of leukocytes, thrombocytes, and results of blood chemistry were within normal limits. Erythrocyte sedimentation rate was 10 mm/h and serum level of C-reactive protein was 0.5 mg/dl. Measurement of arterial blood gas analysis on room air revealed pH: 7.44, PaCO2: 36mmHg, PaO2: 74mmHg, HCO3-. 24mmol/L and SaO2: 96% which was compatible with mild hypoxemia.
Her chest radiograph revealed a homogenous and regularly-shaped dense shadow expanding superiorly and laterally from the hilum of the right lung (Figure 1a). Computerized Tomography (CT) of the thorax demonstrated a mass lesion which was 40 mm in diameter and surrounding the right main bronchus and distal portion of the right main pulmonary artery (Figure 2a).
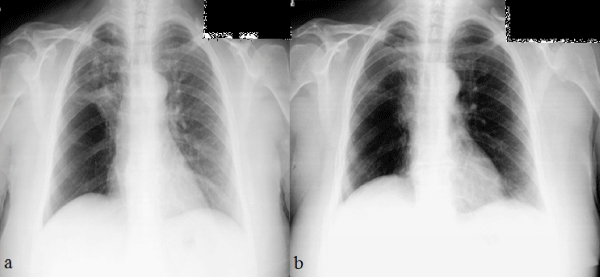
Figure 1: Chest radiographs of the patient. (a) On admission (b) A year after discharge.
View Figure 1
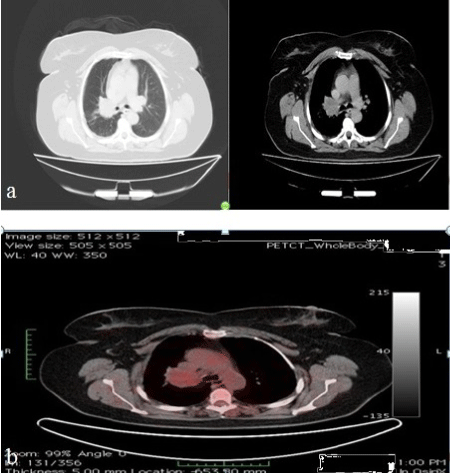
Figure 2: Thorax-CT (a) and PET-CT (b) images of the patient on admission.
View Figure 2
The patient underwent flexible bronchoscopy and near total occlusion of the right main bronchus by a white, luminous and polypoid lesion was observed. During the bronchoscopy procedure the right main bronchus appeared inflamed and contained increased secretions. Gram stain and cultures of the bronchial washings did not reveal any infectious organism. Pathology report of the forceps biopsy was describing a mesenchymal tumour with a polypoid growth pattern and eosinophilic chondroid matrix surrounded by respiratory epithelium. The tumorous lesion had no infiltrative nature although it was displaying cellular atypia. Immunohistochemical studies showed positivity to Epithelial Membrane Antigen (EMA), vimentin and S-100 protein, but staining was negative for CD34 and Neuron Specific Enolase (NSE) (Figure 3). Positron Emission Tomography-Computerized Tomography (PET-CT) revealed 18-Fluorodeoxyglucose (18-FDG) uptake in the right hilar mass lesion equal to theadjacent blood pool (Figure 2b). Based on the clinical, radiological and histopathological features, the patient was diagnosed as endobronchial pulmonary mesenchymoma.
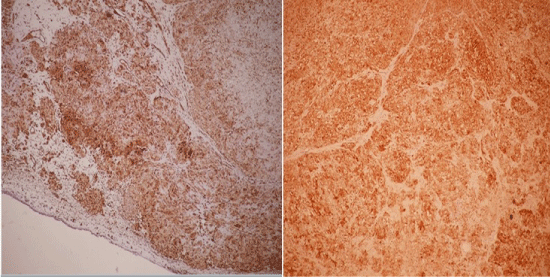
Figure 3: Pathological view of the tumour cells. Positive staining with vimentin (left) and S-100 antibodies (right) (100x).
View Figure 3
Right upper lobectomy via video-assisted thoracoscopic surgery was performed and gross examination of the specimen disclosed a lobular, well-circumscribed, and yellowish-white mass about 45?mm in diameter (Figure 4). Histologically, a lobular mass of mature hyaline cartilage was intimately admixed with fibromyxoid stroma and there was no sign of a malignancy. In addition, surgical margins were clear and there was no invasion of lymph nodes.
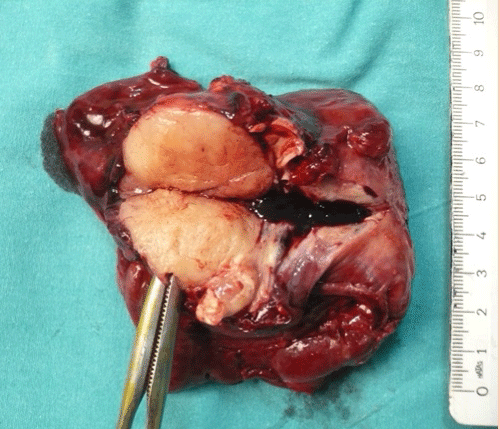
Figure 4: Gross appearance of the endobronchial mesenchymoma.
View Figure 4
After surgery, all symptoms of the patient disappeared and she had no more need for bronchodilators. Last contact with her was a polyclinic visit for control one year after discharge and she was free of respiratory symptoms and radiological relapse (Figure 1b)
Discussion
Pulmonary mesenchymomas are the most common form of benign lung tumours with an incidence of between 0.035%�0.32% according to different necropsy studies [5]. Although it is very popular to call these tumours as "hamartoma", this term does not correspond to the pathological features of these tumours. The word "hamartoma� describes amalformation made up of an abnormal mixture of cells and tissues without a progression in size and it is considered as a developmental error. However, these tumours are consisting of various connective tissue elements and have a capability of growth which leads to the conclusion that the tumour is a benign neoplasm rather than a hamartoma. Therefore, the name "mesenchymoma" seems more appropriate than hamartoma[6]. In addition, some authors refer these tumours as "fibrolipochondroma" based on the composition of them consisting cartilage, muscle, fat, and bone [7]. The histology of the these lesions usually reveals a predominant chondroid differentiation (80%), with fibroblastic (12%), fatty (5%) and osseous (3%) differentiation making the rest [5].
The peak incidence is between the fifth and sixth decades with a male predominance[8]. Most patients with mesenchymomas are smokers and in one series only 14% of the patients had no smoking history[9]. Conversely, the patient in this case was a female and had no history of a heavy-smoking.
Most patients remain asymptomatic for a long time and usually the lesions are diagnosed incidentally. Majority of the mesenchymomas are intraparenchymal lesions (90%) and endobronchial lesions making up only about 10%. They are typically well-circumscribed nodules or small masses with either smooth or lobulated margins. The most common radiological presentation is a solitary pulmonary nodule which accounts for 6% of the pulmonary nodules [10]. Mostly they are less than 2.5-4 cm in diameter, although they can be larger than 10 cm in some cases [3,11]. The presence of fat density and pop-corn like calcification are essentially pathognomonic of a pulmonary mesenchymoma. Presence of fat density in a well circumscribed solitary pulmonary nodule which does not demonstrate significant growth is essentially pathognomonic of a pulmonary mesenchymoma and no further investigations are required [12]. Fat can be recognized by comparing it to subcutaneous fat, and will typically have a Hounsfield measurement of -40 to -120 units[3]. However, approximately 50% of the cases have fat density and only approximately 20-30% of cases have pop-corn like calcification [12,13]. Cavitation is not seen. PET-CT is useful for the differentiation of these lesions from malignant lung tumours, but some reports on mesenchymomas describe difficulty with differentiation from malignant tumours when slight abnormal accumulation of 18-FDG is observed [14]. This patient presented with an endobronchial lesion without popcorn calcification and without presence of fat density in CT scan. Luckily, it was easy to distinguish this tumorous lesion from malignant tumours with the help of equal uptake of 18-FDG to the adjacent blood pool.
Pulmonary mesenchymomas continue to increase in size at a slow rate with a typical volume doubling time of over 400 days [15]. Therefore, main approach to asymptomathic pulmonary mesenchymomas is close medical follow-up to exclude growth if the diagnosis could be obtained without surgical intervention. Conversely, although malignant transformation is exceedingly rare, there are some presented case reports of mesenchymoma with malignant alteration [16]. So, surgery is recommended for those patients presented with rapid growth of the tumour, patients who have respiratory symptoms and if the exact diagnosis could not be obtained without surgery [2,17]. Enucleation, wedge resection, segmentectomy, lobectomy, sleeve lobectomy and pneumonectomy are possible surgical procedures for pulmonary mesenchymomas. For endobronchial mesenchymomas, bronchoscopic procedure is another option.Unless there is accompanying parenchymal damage, lung resection should be avoided and parenchyma-protecting surgical procedures should be choosen the first place [18]. The patient in this case had progressive respiratory symptoms and occlusion of the right main bronchus for which right upper lobectomy was performed.
As this case describes, endobronchial lesions including pulmonary mesenchymomas may present with the symptoms of asthma and may be treated mistakenly for a long time with bronchodilators. Patients with chronic symptoms suggestive of asthma, poor response to asthma medications, and frequent exacerbations should be evaluated for endobronchial lesions.
References
-
Gaerte SC, Meyer CA, Winer-Muram HT, Tarver RD, Conces DJ Jr (2002) Fat-containing lesions of the chest. Radiographics 22: S61-78.
-
Shields TW, Robinson PG (2000) Benign tumors of the lung. General Thoracic Surgery (5th edn). Lippincott Williams & Wilkins, Philadelphia.
-
Collins J, Stern EJ (2007) Chest radiology: the essentials. Lippincott Williams & Wilkins.
-
Cos�o BG, Villena V, Echave-Sustaeta J, de Miguel E, Alfaro J, et al. (2002) Endobronchialhamartoma. Chest 122: 202-205.
-
Human pathology (2012) Pulmonary parenchymal hamartoma.
-
van den Bosch JM, Wagenaar SS, Corrin B, Elbers JR, Knaepen PJ, et al. (1987) Mesenchymoma of the lung (so called hamartoma): a review of 154 parenchymal and endobronchial cases. Thorax 42: 790-793.
-
Zheng RH, Chou DH, Shi MX (1994) [Hamartoma of the lung: an analysis of 24 cases]. ZhonghuaJie He He Hu Xi ZaZhi 17: 86-87, 126.
-
Hansen CP, Holtveg H, Francis D, Rasch L, Bertelsen S (1992) Pulmonary hamartoma. J Thorac Cardiovasc Surg 104: 674-678.
-
Gjevre JA, Myers JL, Prakash UB (1996) Pulmonary hamartomas. Mayo Clin Proc 71: 14-20.
-
Thomas JW, Staerkel GA, Whitman GJ (1999) Pulmonary hamartoma. AJR Am J Roentgenol 172: 1643.
-
Siegelman SS, Khouri NF, Scott WW Jr, Leo FP, Hamper UM, et al. (1986) Pulmonary hamartoma: CT findings. Radiology 160: 313-317.
-
Klein JS, Braff S (2008) Imaging evaluation of the solitary pulmonary nodule. Clin Chest Med 29: 15-38, v.
-
Chai JL, Patz EF Jr (1994) CT of the lung: patterns of calcification and other high-attenuation abnormalities. AJR Am J Roentgenol 162: 1063-1066.
-
Himpe U, Deroose CM, Leyn PD, Verbeken E, Vansteenkiste J (2009) Unexpected slight fluorodeoxyglucose-uptake on positron emission tomography in a pulmonary hamartoma. J Thorac Oncol 4: 107-108.
-
Naidich DP, Srichai MB, Krinsky GA (2001) Computed tomography and magnetic resonance of the thorax. Thorax 56: 898.
-
Basile A, Gregoris A, Antoci B, Romanelli M (1989) Malignant change in a benign pulmonary hamartoma. Thorax 44: 232-233.
-
deRooij PD, Meijer S, Calame J, Golding RP, van Mourik JC, et al. (1988) Solitary hamartoma of the lung: is thoracotomy still mandatory? Neth J Surg 40: 145-148.
-
Guo W, Zhao YP, Jiang YG, Wang RW, Ma Z (2008) Surgical treatment and outcome of pulmonary hamartoma: a retrospective study of 20-year experience. J Exp Clin Cancer Res 27: 8.





