International Journal of Pathology and Clinical Research
Chronic Traumatic Encephalopathy-like Neuropathological Findings without a History of Trauma
Andrew F Gao1, David Ramsay2, Richelle Twose3, Ekaterina Rogaeva4, Charles Tator5,6 and Lili-Naz Hazrati1,6,7*
1Department of Laboratory Medicine and Pathobiology, University of Toronto, Toronto, ON, Canada
2Department of Pathology and Laboratory Medicine, University of Western Ontario, London, ON, Canada
3Department of Psychiatry, University Health Network, Toronto, ON, Canada
4Tanz Centre for Research in Neurodegenerative Disease, University of Toronto, Toronto, ON, Canada
5Department of Surgery, University of Toronto, Toronto, ON, Canada
6Canadian Concussion Centre, Toronto Western Hospital, Toronto, ON, Canada
7Department of Laboratory Medicine, Hospital for Sick Children, and the Canadian Concussion Centre, Toronto Western Hospital, Toronto, ON, Canada
*Corresponding author:
Dr. Lili-Naz Hazrati, Department of Laboratory Medicine, Hospital for Sick Children and the Canadian Concussion Centre, Black wing, 3rd floor, Room #3205, 555 University Avenue, M5G 1X8, Toronto, ON, Canada, Email: lilinaz.hazrati@utoronto.ca
Int J Pathol Clin Res, IJPCR-3-050, (Volume 3, Issue 1), Case Report; ISSN: 2469-5807
Received: September 27, 2016 | Accepted: January 09, 2017 | Published: January 12, 2017
Citation: Gao AF, Ramsay D, Twose R, Rogaeva E, Tator C, et al. (2017) Chronic Traumatic Encephalopathy-like Neuropathological Findings without a History of Trauma. Int J Pathol Clin Res 3:050. 10.23937/2469-5807/1510050
Copyright: © 2017 Gao AF, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Chronic traumatic encephalopathy (CTE) is a neurodegenerative disease characterized by neurocognitive decline and abnormal neuronal and glial deposition of hyperphosphorylated-tau in a pattern distinct from other tauopathies. These changes are hypothesized to occur following repeated mild traumatic brain injury. We report a case of CTE-like neuropathological changes, and features of motor neuron disease in a patient without history of either head trauma or participation in activities associated with increased risk of concussive or sub-concussive blows.
A 45-year-old man with a 7-year history of clinically diagnosed amyotrophic lateral sclerosis was discovered dead by his wife following a 4-hour unattended sleep. Microscopically, neurofibrillary tangles and neuropil threads were seen in the frontal and temporal cortices in the superficial cortical layers, in the depths of sulci, and surrounding blood vessels. Glial tau pathology included astrocytic tangles in a perivascular and subpial distribution. TDP-43 inclusions were seen in the spinal cord. No MAPT mutations or C9ORF72 repeat expansions were detected.
Although CTE has been found only in patients with a history of head trauma thus far, our case potentially highlights the complexity in the pathogenesis of this disorder.
Keywords
Chronic traumatic encephalopathy, Traumatic brain injury, Neurodegeneration, Motor neuron disease, Tau, TDP-43, Amyotrophic lateral sclerosis
Introduction
Chronic traumatic encephalopathy (CTE) is characterized by neurocognitive decline and abnormal neuronal and glial deposition of hyperphosphorylated-tau in a pattern distinct from other tauopathies [1]. These changes are hypothesized to occur following repeated mild traumatic brain injury [2]. We report the first case of CTE-like neuropathological changes, and features of motor neuron disease (MND) [3], in a patient without history of either head trauma or participation in activities associated with increased risk of concussive or sub-concussive blows.
CTE has recently been subject to intense media scrutiny following diagnosis in a number of high-profile professional athletes, raising broad societal and legal implications [4]. Despite this, it remains unclear if sports-related trauma is a definitive cause of CTE [5]. Since CTE requires postmortem examination, most studies to date have been based on patient/family referral, and thus may contain an inherent selection bias [6], and some authors have challenged the concept that head trauma leads to an increased risk of neurocognitive pathology in later life [5,7]. Interestingly, non-traumatic causes of tau accumulation have been reported in cases of epilepsy, which has been shown to be associated with abnormal tau accumulation [8] and can recapitulate some of the pathological features of CTE [9].
Case Presentation
We report a 45-year-old man with CTE-like neuropathology and MND without an antecedent history of trauma. The patient had a 7-year history of progressive motor wasting and a clinical diagnosis of amyotrophic lateral sclerosis (ALS). He exhibited marked upper limb wasting and weakness, but was still mobile. The patient's course was complicated by recurrent pneumonia and respiratory involvement, necessitating the use of BiPAP during sleep. The patient had been a high functioning professional prior to illness and was married with one child. There was no history of smoking, alcohol, or illicit drug use. Importantly, there was no history of participation in contact sports or of head injury of any severity per the patient and his wife. There was also no history of cognitive or psychiatric illness at any time throughout life. There was no family history of neurological illness. The patient was discovered dead by his wife following a 4-hour unattended sleep, lying on the floor with vomitus in his BiPAP mask that was still in place.
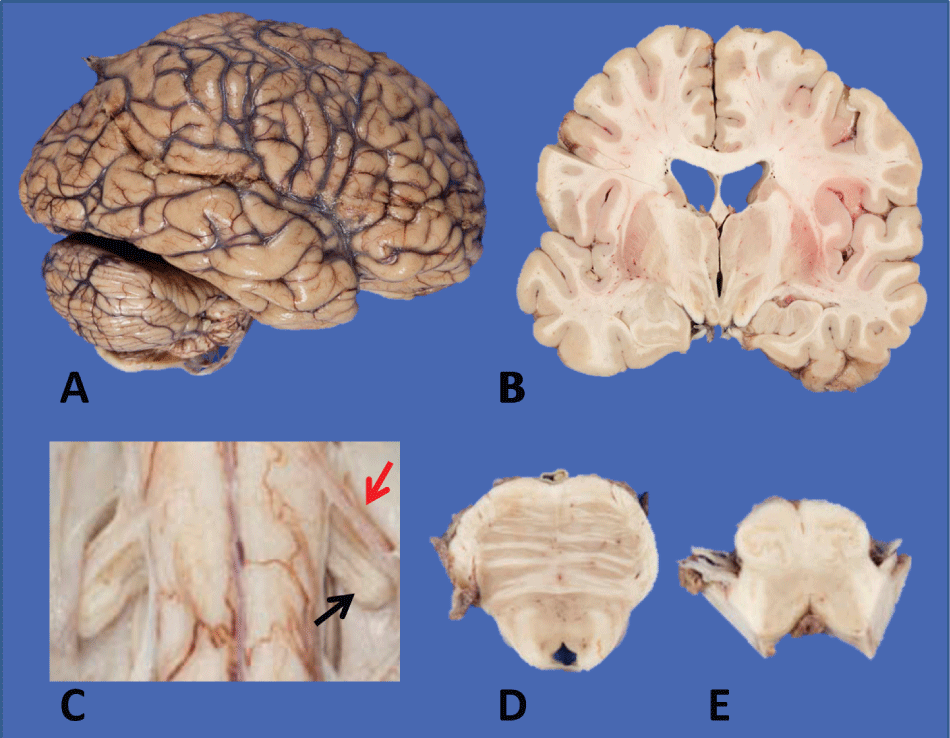
Postmortem examination confirmed the cause of death as asphyxiation due to aspiration in the setting of ALS and the general autopsy was unremarkable. Neuropathological examination demonstrated a grossly normal brain (Figure 1a and Figure 1b) without signs of cortical atrophy. The spinal cord revealed marked atrophy of the anterior nerve roots, particularly prominent in the cervical region (Figure 1c). The brainstem was showed decreased volume of the corticospinal tract in the basis pontis and medullary pyramids (Figure 1d and Figure 1e).
.
Figure 1: Macroscopic features of brain and spinal cord: a,b) Largely unremarkable brain with no evidence of cortical atrophy on external examination or coronal sections; mild ventricular dilatation; c) Marked decrease in caliber of anterior spinal rootlet (red arrow) compared to the dorsal rootlet (black arrow); d) Pons with a well-pigmented locus ceruleus and slight decrease in caliber of descending tracts; e) Medulla with mild flattening of pyramids.
View Figure 1
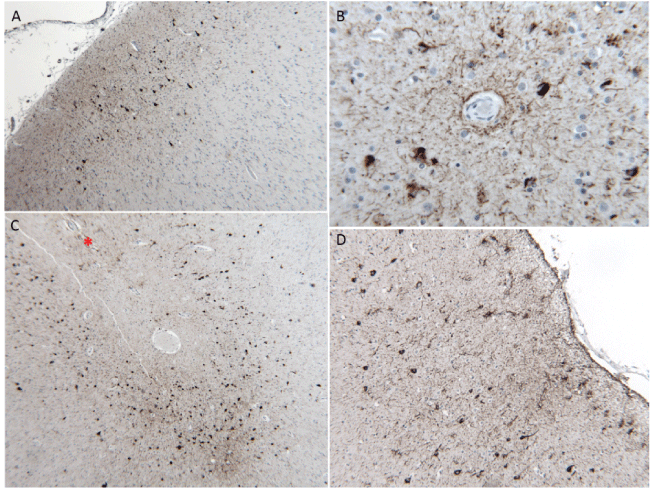
Routine microscopic examination of the frontal and temporal cortices showed scattered dense neurons but was otherwise unremarkable. Sections through the motor cortex showed mild loss of Betz cells. Phosphorylated-tau immunohistochemistry (AT8) highlighted mild to moderate neurofibrillary tangles (NFTs) and neuropil threads in the frontal and temporal cortices that were more frequent in the superficial cortical layers (Figure 2a), surrounding blood vessels (Figure 2b), and in the depths of sulci (Figure 2c). Glial tau pathology included astrocytic tangles in a perivascular and subpial distribution (Figure 2D). In the motor cortex, TDP-43-positive neuronal and glial cytoplasmic inclusions were seen. Beta-amyloid immunohistochemistry showed sparse faint-staining diffuse plaques.
.
Figure 2: Features of chronic traumatic encephalopathy-like neuropathology: a) Neurofibrillary tangles preferentially located in the superficial cortical layers, 10X; b) Foci of phosphorylated-tau inclusions in astrocytes and neurons in a perivascular pattern, 40X; c) Predilection for phosphorylated-tau pathology in the depths of sulci (marked by *), 10X and; d) Subpial clusters of astrocytic phosphorylated-tau pathology, 20X. All panels show AT8 immunohistochemistry.
View Figure 2
Ammon's horn had a normal morphology and was well populated with pyramidal neurons. Tau immunostained sections demonstrated isolated NFTs in CA4 and CA1 and dense accumulation in both superficial and deep layers of the entorhinal cortex. TDP-43 and beta-amyloid immunohistochemistry was negative. The amygdala was unremarkable.
Sections through the basal ganglia showed a prominence of the perivascular spaces but the putamen, caudate, globus pallidus, nucleus basalis of Meynert, and thalamic nuclei were otherwise unremarkable. The upper brainstem and cerebellum were unremarkable, including a well-populated substantia nigra and locus ceruleus. Rare AT8-positive inclusions were identified in the substantia nigra pars compacta (Figure 3e). The medulla showed myelin pallor of the pyramids and neuronal loss and central chromatolysis in the hypoglossal nuclei; scattered p62 positive glial inclusions.
.
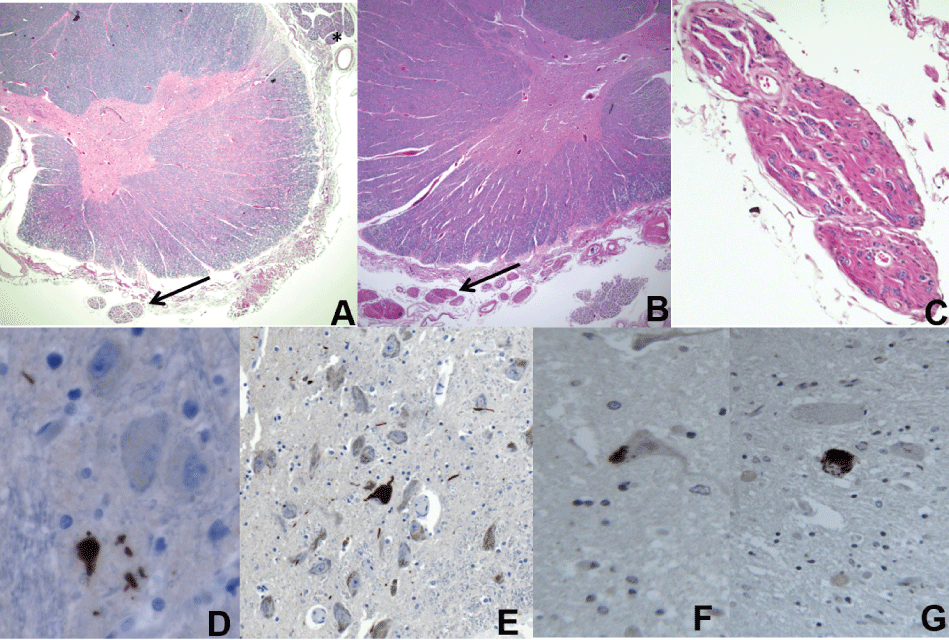
Figure 3: a) Thoracic spinal cord with neuronal loss and gliosis in the anterior horn of the spinal cord and pallor of the corticospinal tract. Note the diminished size of the ventral nerve roots (arrow) compared to the dorsal roots (*), 5X; b) Cervical cord with similar findings. Note the thinned ventral rootlets (arrow); c) Higher magnification of the anterior spinal rootlet, 10X; d) AT8-positive inclusions in the anterior horn (d,40X); e) Substantia nigra pars compacta (e,20X); f) TDP-43 immunohistochemistry shows positive neuronal cytoplasmic inclusions in the lumbosacral; g) Cervical cords, respectively; 40X.
View Figure 3
Sections through all levels of the spinal cord showed marked loss of anterior horn motor neurons (Figure 3a and Figure 3b), with some of the remaining neurons showing central chromatolysis. The anterior horns were gliotic with scattered axonal swellings. The lateral corticospinal tracts were asymmetrically pale with occasional foamy macrophages and vacuolation present on myelin staining (Figure 3a). Ventral rootlets were markedly thinned compared to dorsal rootlets (Figure 3a and Figure 3c). No Bunina bodies were seen. AT8-positivie inclusions were seen in the ventral horn (Figure 3d). Numerous TDP-43 positive cytoplasmic threads and, more rarely, round cytoplasmic inclusions were present in some of the remaining neurons (Figure 3f and Figure 3g). Glial cytoplasmic inclusions were also seen on TDP-43 staining.
Mutations in the MAPT gene, or an expansion in C9orf72 (genotype 2/2) were not detected in this patient.
Overall, the features of this case are of a tauopathy with MND. The clinical and neuropathological findings -- tau deposition consistent with the pattern seen in CTE, TDP-43+ MND, and an antemortem clinical diagnosis of ALS -- are similar to the newly described entity of chronic traumatic encephalomyelopathy [3,10,11]. The distribution of tau pathology corresponds to McKee stage II to III CTE [11]. However, a history of traumatic brain injury is lacking in this case.
Discussion
Traumatic brain injury can result in long-term neurological deficits and CTE has been seen in a wide array of athletes (boxing, American football, soccer, wrestling, hockey, and rugby) and in soldiers who have experienced blast injuries [12]. However, one must still be cautious in assigning trauma as the inciting factor for the pathological changes seen in CTE [7]. Case histories are often retrospective accounts from the patient's family or friends and studies are mostly composed of samples of convenience [5]. Additionally, because of the long interval between the injury and development of symptoms, it is difficult to control for additional potential insults including drug and alcohol use that are often concomitant [2].
Regrettably, large-scale controlled studies examining the prevalence of CTE-like pathologies in the general population are generally lacking and data has mainly come from several smaller studies. Omalu, et al. [2] examined 14 professional athletes and 3 high school athletes after unexpected death and found CTE pathology in 10 (71%) and 1, respectively. Drug-related deaths were overrepresented in this cohort. In a series of 6 consecutively referred brains of former professional athletes with presumptive clinical diagnoses of CTE, 3 revealed the co-existence of CTE with an additional neurodegenerative disease (Alzheimer disease [AD], diffuse Lewy body disease [DLBD], and multiple infarcts), and 3 had an absence of CTE with findings of a different neurodegenerative disorder (ALS, AD, and Parkinson disease [PD]) [13]. In the largest series to date of 85 brains from former athletes, military veterans, or civilians with a history repetitive head injury, McKee, et al. [11] found that 17 (20%) showed no CTE neuropathology while 68 (80%) did. However, out of the latter group, only 43 (51%) demonstrated CTE pathology alone; whereas 8 (9%) cases were CTE-MND and 17 (20%) were CTE plus another neurodegenerative disease (AD, PD, DLBD, frontotemporal lobar degeneration, progressive supranuclear palsy, Pick's disease). The individuals without a head injury (18 controls) showed no CTE pathology. Among the athletes without military service (60 cases), 15 (25%) did not have CTE neuropathology, 27 (45%) had CTE only, 7 (12%) CTE-MND, and 11 (18%) CTE plus another type of neurodegeneration. An investigation of CTE pathology in a large brain bank for neurodegenerative disorders found 21 of 66 former athletes had pathology consistent with CTE [14]. In 198 control patients without exposure to contact sports, including those with single-incident traumatic injuries, there was no evidence of CTE pathology.
To our knowledge, this is the first description of a patient with neuropathological features of CTE-MND in the absence of a history traumatic brain injury. Interestingly, despite cortical tau pathology, our patient never exhibited cognitive impairment, which speculatively could be explained by the relative sparing of the nucleus basalis of Meynert. This highlights the uncertainty surrounding the pathogenesis and pathophysiology of CTE and underscores the need for further detailed studies to elucidate the causative role of trauma. Nevertheless, our case report has several important limitations. For example, the lack of a trauma history comes only from the recollection of the patient's wife and early life subconcussive blows cannot be entirely excluded. There are also inherent limitations to drawing conclusions from the results of a single patient.
To date, repetitive traumatic brain injury has been shown to be associated with no neuropathological changes, with CTE alone, with CTE and another neurodegenerative disease, or with non-CTE neurodegeneration. Since CTE is a postmortem diagnosis, the majority of samples have come from brains of symptomatic individuals referred by family; these individuals are more likely to demonstrate some type of neuropathology thus introducing a selection bias. Our case adds to this complexity given the observation that CTE-like changes can occur in the absence of any known head injury; casting doubt that trauma is always the inciting etiological factor. Future studies should assess whether CTE-like pathology is prevalent in non-concussed patient populations.
Methods
The formalin-fixed brain underwent systematic and standardized sampling and neuropathological examination by a neuropathologist (LNH). 6-μm paraffin-embedded sections were stained for hematoxylin and eosin/luxol fast blue (H&E/LFB) and processed for immunohistochemistry. Following de-paraffinization and heat-induced antigen retrieval where necessary, immunohistochemistry was performed with the following antibodies: anti-tau AT8 pS202/T205 (mouse monoclonal, 1:1000, ThermoFisher Scientific, USA), anti-phospho-TDP-43 pS409/10 (mouse monoclonal, 1:3000, Cosmo Bio Co., Ltd., USA), anti-α-synuclein LB509 (mouse monoclonal, 1:100, BioLegend Inc, USA), anti-p62 (mouse monoclonal, 1:200, BD Transduction Laboratories, USA), anti-beta-amyloid 6F/3D (mouse monoclonal, 1:50, Dako, Denmark).
Genomic DNA was obtained from peripheral blood using a QIAGEN kit. Mutation analysis of the MAPT gene, coding for tau protein, and the presence of a G4C2-repeat expansion in C9orf72 were done as described previously [15,16].
Acknowledgements
The authors thank Kelvin, So for assisting with immunohistochemistry.
Ethics Statement
Not applicable.
References
-
McKee AC, Cantu RC, Nowinski CJ, Hedley-Whyte ET, Gavett BE, et al. (2009) Chronic traumatic encephalopathy in athletes: progressive tauopathy after repetitive head injury. Neuropathol Exp Neurol J 68: 709-735.
-
Omalu B, Bailes J, Hamilton RL, Kamboh MI, Hammers J, et al. (2011) Emerging histomorphologic phenotypes of chronic traumatic encephalopathy in American athletes. Neurosurgery 69: 173-183.
-
McKee AC, Gavett BE, Stern RA, Nowinski CJ, Cantu RC, et al. (2010) TDP-43 proteinopathy and motor neuron disease in chronic traumatic encephalopathy. Neuropathol Exp Neurol J 69: 918-929.
-
Korngold C, Farrell HM, Fozdar M (2013) The National Football League and chronic traumatic encephalopathy: legal implications. Am Acad Psychiatry Law J 41: 430-436.
-
Karantzoulis S, Randolph C (2013) Modern chronic traumatic encephalopathy in retired athletes: what is the evidence? Neuropsychol Rev 23: 350-360.
-
Tartaglia MC, Hazrati LN, Davis KD, Green RE, Wennberg R, et al. (2014) Chronic traumatic encephalopathy and other neurodegenerative proteinopathies. Front Hum Neurosci J 8:30.
-
Gardner A, Iverson GL, McCrory P (2014) Chronic traumatic encephalopathy in sport: a systematic review. Br J Sports Med.
-
Thom M, Liu JY, Thompson P, Phadke R, Narkiewicz M, et al. (2011) Neurofibrillary tangle pathology and Braak staging in chronic epilepsy in relation to traumatic brain injury and hippocampal sclerosis: a post-mortem study. Brain 134: 2969-2981.
-
Puvenna V, Engeler M, Banjara M, Brennan C, Schreiber P, et al. (2016) Is phosphorylated tau unique to chronic traumatic encephalopathy? Phosphorylated tau in epileptic brain and chronic traumatic encephalopathy. Brain Res 1630: 225-240.
-
Stern RA, Riley DO, Daneshvar DH, Nowinski CJ, Cantu RC (2011) Long-term consequences of repetitive brain trauma: chronic traumatic encephalopathy. PM R.
-
McKee AC, Stern RA, Nowinski CJ, Stein TD, Alvarez VE, et al. (2013)The spectrum of disease in chronic traumatic encephalopathy. Brain 136: 43-64.
-
Mez J, Stern RA, McKee AC (2013) Chronic traumatic encephalopathy: where are we and where are we going? Curr Neurol Neurosci Rep 13: 407.
-
Hazrati LN, Tartaglia MC, Diamandis P, Davis KD, Green RE, et al. (2013) Absence of chronic traumatic encephalopathy in retired football players with multiple concussions and neurological symptomatology. Front Hum Neurosci 7: 222.
-
Bieniek KF, Ross OA, Cormier KA, Walton RL, Soto-Ortolaza A, et al. (2015) Chronic traumatic encephalopathy pathology in a neurodegenerative disorders brain bank. Acta Neuropathol 130: 877-889.
-
Curcio SA, Kawarai T, Paterson AD, Maletta RG, Puccio G, et al. (2002) A large Calabrian kindred segregating frontotemporal dementia. Journal of neurology 249: 911-922.
-
Xi Z, Zinman L, Grinberg Y, Moreno D, Sato C, et al. (2012) Investigation of c9orf72 in 4 neurodegenerative disorders. Arch Neurol 69: 1583-1590.





