International Journal of Pathology and Clinical Research
A Review of Carcinoma Ex-Pleomorphic Adenoma of the Salivary Glands
Saeedeh Khalesi*
Department of Oral and Maxillofacial Pathology, School of Dentistry, Isfahan University of Medical Sciences, Isfahan, Iran
*Corresponding author:
Saeedeh Khalesi, Department of Oral and Maxillofacial Pathology, School of Dentistry, Isfahan University of Medical Sciences, Isfahan, Iran, E-mail: s_khalesi@dnt.mui.ac.ir
Int J Pathol Clin Res, IJPCR-2-043, (Volume 2, Issue 3), Review Article; ISSN: 2469-5807
Received: July 17, 2016 | Accepted: August 27, 2016 | Published: August 31, 2016
Citation: Khalesi S (2016) A Review of Carcinoma Ex-Pleomorphic Adenoma of the Salivary Glands. Int J Pathol Clin Res 2:043. 10.23937/2469-5807/1510043
Copyright: © 2016 Khalesi S. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Abstract: One of the malignant forms of pleomorphic adenomas (PA) is carcinoma ex pleomorphic adenoma (CA-ex-PA). Carcinoma ex pleomorphic adenoma is an uncommon malignant tumor with high aggressive behavior and poor prognosis. However, there are a few studies in this topic in the recent literature. Furthermore, the lesions are uncommon and often have a difficult diagnosis by clinicians and pathologists, because the residual small PA component and various malignancy subtypes may be present. In this study, we reviewed the recent studies in about clinical, radiographical, histopathological and molecular features of CA-ex-PA.
Methods: In this review article, the studies were identified by researches on three databases- Pubmed, MEDLINE, and Googlescholar- from 2005 through 2015 in the English language for the carcinoma ex pleomorphic adenoma. We used the following search terms: « carcinoma ex pleomorphic adenoma », « salivary gland tumors ». We included original studies and review articles.
Results: Recently, many researchers have been performed to study, a review of development and progression of carcinoma ex pleomorphic adenoma which have been identified already.
Conclusion: CA-ex-PA is an uncommon malignant tumor with the particular biological behavior in contrast to the other salivary gland neoplasm. It is important to be aware of the tumor and early detection of pleomorphic adenoma is important for the prevention of Ca-ex-PA.
Keywords
Salivary gland tumors, Carcinoma ex pleomorphic adenoma, Histopathology
Introduction
The malignant form of pleomorphic adenoma (PA) is including malignant mixed tumor, carcinosarcoma and metastasizing PA. They are malignant tumor arising from a primary (de novo) or recurrent benign pleomorphic adenoma [1]. Carcinosarcoma include the malignant epithelial and mesenchymal components and often called "true malignant mixed tumor". However, metastasizing PA is characterized by the presence of one or more location of metastatic lesion that in histopathological features without any malignant view [2]. The most common malignant form of PA is carcinoma ex pleomorphic adenoma (CA-ex-PA). Carcinoma ex pleomorphic adenoma is an uncommon malignant salivary gland tumor with high aggressive behavior and poor prognosis [3]. There are a few studies in this topic in the recent literature. These lesions often have a difficult diagnosis by clinicians and pathologists, because the residual small PA component and various malignancy subtypes may be present [4]. In this study, we reviewed the recent studies in about clinical, radiographical, histopathological and molecular features of CA-ex-PA.
Epidemiology
According the most studies, CA-ex-PA constitutes approximately 3.6% of all salivary gland tumors, 6.2% of all mixed tumors and 11.6% of all malignant salivary gland tumors [5,6]. CA-ex-PA is a rare malignant salivary gland tumor with a prevalence rate of 5.6% cases per 100,000 malignant tumors and yearly incidence rate of 0.17% tumors per 1 million persons [5]. However, geographic differences in the prevalence of this lesion have been observed. In the study of United Kingdom noted that CA-ex-PA forms about 25% of all primary parotid malignant tumors. Furthermore, the Byrne's study showed a prevalence of 12% among primary parotid malignant neoplasms in the United States. In 2008, the Zbaren, et al. showed the 14% of all primary parotid malignant neoplasms over a 20 year period in Switzerland. As well as, the prevalence of CA-ex-PA transformation from PA has been reported as 1.5% to 13.8% [4,5,7]. The study in the Isfahan city, CA-ex-PA was only 2.1% of all salivary gland tumors in this population [6].
Clinical Features
CA-ex-PA is a malignant transformation within a primary (de novo) or recurrent pleomorphic adenoma. Lasting untreated pleomorphic adenoma has an increased risk of carcinomatous change [5]. But, some studies showed the patients with CA-ex-PA had a previously treated PA [1,4]. In the most patients, malignant transformation of PAs have not any significant clinical sign and symptoms [4,8]. The incidence of malignant transformation increased from 1.6% for the tumors with a clinical duration of < 5 years to 9.6% over 15 years [9].
CA-ex-PA predominantly involved the major salivary glands specially in the parotid, the minor salivary gland tumors, lacrimal glands, breast, trachea, and nasal cavity may be affected [10,11]. Hard and soft palate is the most locations of involved minor salivary gland [5]. The most clinical features of CA-ex-PA are a firm mass in the parotid gland that is palpable in extraoral examination [1,8]. Generally, the cancers at minor salivary gland have smaller size than the major salivary gland tumors [12].
CA-ex-PA can be asymptomatic and quite resemble to benign PA [4,5,8]. Some clinical features such as pain may be observed with locally invasive of neoplasm into adjacent structures [4,8]. Facial nerve paresis or palsy has been detected in approximately one-third of the patients with facial nerve involvement [8]. Other clinical manifestation such as skin ulceration, skin fixation, palpable lymphadenopathy and dysphasia reported [1,8]. Jaw swelling, dental pain and loss of tooth vitality may be observed in the cases with bone involvement [13]. Also, lacrimal mass has been seen with extension of cancer into the canaliculi and nasolacrimal dust with or without chronic epiphora and recurrent dacryocystitis [14]. Observation of rapid enlargement of the mass, pain, or other warning clinical symptoms can help to early diagnosis. According to the studies, rare patients with malignant changes of PA may carry a slow growing for over 40 years before coming to clinical attention [8]. In Gepps's study, the cancer had a mean lead up time of 23-3 years with the half the cases aware of the swelling for about 2-3 years [15]. General clinical parameters such as stage (size, soft and hard tissue or skin involvement, and lymph node status), age and margin status are important for prognosis detection of salivary gland carcinomas [16].
Pathological Properties
Macroscopic features
The macroscopic features depend to rate of benign and malignant components of tumor. The tumors with dominant PA component are greyish-blue and transport to yellowish-white. This appearance may be related to hyalinization and calcification in the stroma of PA. When the malignant component is dominant, the tumor is widely infiltrative, ill-defined and has foci of necrosis and hemorrhage. Furthermore, the size of the tumors have ranging from 1 to 25 cm [8,15,17].
Microscopic features
The histopathological features of CA-ex-PA is includes both benign PA and malignant components. This lesion is high grade malignancy and have histological feature similar to their de novo counterparts [11,18]. The carcinoma component may be show different types of malignant tumors and have an aggressive clinical behavior [19]. According to Lewis study, more than 50% of the cancer is the carcinomatous component in 84% of the patients [17]. In histopathological features of some CA-ex-PAs, the carcinoma counterpart is without PA counterpart. In these cases, the PA component can be detected by past medical history, previous biopsy answer, clinical feature and additional sectioning of the tissue samples. Also, CA-ex-PA may be has predominantly the PA component and few areas of malignant transformation with nuclear pleomorphism, atypical mitotic figures, hemorrhage and necrosis. In these cases, correct diagnosis is difficult for pathologist and need to more evaluation. Observation of extensive stromal hyalinization, hypercellularity, cellular atypism, increase mitotic activity, necrosis, and cellular infiltration to capsular zone are commonly related to malignant transformation in PA [5].
The CA-ex-PA is mainly classified into 2 subtypes by the morphological and immunohistochemical features. The first group (75% of all of CA-ex-PA) comprises carcinomas with luminal (ductal epithelium) differentiation [20]. In this subtype, the malignant changes of ductal epithelial cells may allow a stepwise sequence and manifest as 1) intraductal carcinoma or carcinoma in situ, which the carcinomatous cells located to the inner layer of ducts, but myoepithelial layer is intact; 2) intracapsular carcinoma, which the carcinomatous cells break through the myoepithelial layer with the invasion to the matrix of PA without capsular invasion; 3) extracapsular carcinoma, which the invasion of carcinoma cells observed beyond the fibrous capsule to the extracapsular area [21-23]. The most intraductal ductal-type CA-ex-PAs are same to the intraductal component of salivary duct carcinoma (SDC), and the intracapsular or extracapsular invasive ductal-type CA-ex-PAs can be subclassified in to the salivary duct carcinomas and adenocarcinoma not otherwise specified (ADC-NOS) type [18,22,24,25]. The Demasi's study showed that CA-ex-PA with only epithelial differentiation (CA-ex-PA-LD) was limited to the major salivary glands [26].
The second subtype of CA-ex-PA comprises carcinomas with non-luminal (myoepithelial) differentiation (CA-ex-PA-NLD). Non-luminal types of CA-ex-PAs histopathologically correspond to myoepithelial carcinomas, which depend to extent of cellular invasion, can be classified as intracapsular or extracapsular. Furthermore, this type of CA-ex-PA can be further divided in to those that exhibit both epithelial and myoepithelial malignancy and those with exclusive myoepithelial malignancy. Those with exclusive myoepithelial malignancy are quite rare. The malignant component of CA-ex-PA-NLD may be myoepithelial carcinoma (ME), epimyoepithelial carcinoma (EMC), and adenoid cystic carcinoma (ADCC). CA-ex-PA-NLD was usually frankly invasive and found in all salivary glands [5,20,23,25,26].
The most common malignant component of CA-ex-PA is adenocarcinoma not otherwise specified. But, the other malignant components are including adenoid cystic carcinoma, myoepithelial carcinoma or salivary duct carcinoma. Rarely, acinic cell carcinoma, epithelial-myoepithelial carcinoma, basal cell carcinoma, myoepithelial carcinoma, squamous cell carcinoma and clear cell carcinoma may be observed (Figure 1) [4,17,27-29]. However a mixture of subtypes may be observed [5,30].
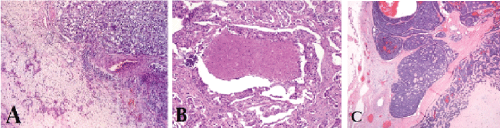
.
Figure 1: (A) Microscopic view of undifferentiated carcinoma is seen infiltrating into the benign pleomorphic adenoma component; (B) Salivary duct carcinoma ex pleomorphic adenoma; (C) Adenoid cystic carcinoma ex pleomorphic adenoma [29].
View Figure 1
In the recent World Health Organization (WHO) classified on the basis of the degree of invasion of the carcinomatous component outside the fibrous capsule, CA-ex-PA divided to non-invasive, minimally invasive, and invasive subtypes which have direct relation with the tumor prognosis [5,20,31].
The non-invasive CA-ex-PA is also known in the literature as intracapsular CA-ex-PA and carcinoma in situ [26,32]. In this lesion, the carcinoma component is confined within the well-defined fibrous capsule of the PA. Non-invasive CA-ex-PA marks the early stages of the cellular malignant transformation and have resemble behavior of PA [5,10,33]. If the malignant penetration of the extracapsular tissue less than 1.5 mm, it is classified as minimally invasive [34]. It is similar to intra-capsular CA-ex-PA with different proportions of PA area, intraductal carcinoma and carcinoma areas [4,26].
Invasive type of CA-ex-PA is characterized by the invasion of malignant component greater than 1.5 mm from the tumor capsule into adjacent tissue [20,34]. The benign PA is composed of nodules of hyalinized tissue with sparse, scattered ductal structures as the carcinoma areas increase in proportion. In invasive CA-ex-PAs have high tendency for malignant cells to decrease in size and migrate away from the origin [20].
Molecular Pathology
In recent studies, molecular analysis of CA-ex-PA has been investigated [5]. These investigations are including the data from chromosomal disorders and genetic alterations. These studies may provide important information on the events associated with development and progression of CA-ex-PA [5,19].
There are some studies about the multi-step model of carcinogenesis demonstrating progressive loss of heterozygosity (LOH) at chromosomal arm 8q, 12q, and 17p, respectively that the main role to development of CA-ex-PA [5,19,35]. There are specific candidate genes in these regions that are related to progression of this cancer. Early alternations of chromosomal arm 8q involve PLAG1 (pleomorphic adenoma gene1), a developmentally regulated zinc-finger proto-oncogene, are common abnormalities in PA and CA-ex-PA. Furthermore, HMGA2 gene rearrangements with or without amplifications, and MYC (8q22.1), a known oncogene over expressions have been described in PA and CA-ex-PA [11,36]. Moreover, the molecular subclassification of CA-ex-PA based on the HER2 gene status may be helpful for prognostic prediction regarding the choic of therapeutic strategy [11,36]. However, the malignant change of PA to CA-ex-PA can be attributed to the 12q genes, HMGIC, HMGA2, and MDM2 [36,37].
According to some studies, loss of P53 gene is common in CA-ex-PA. The P53 gene is a tumor suppressor gene that implicated in the development and progression of many cancers [38]. Moreover, the mutation of P53 gene is important cause of malignant transformation of PA [18,39].
Furthermore, over expression of CyclinD1, P16 and P21 proteins in the malignant epithelial component of CA-ex-PA found in the some immunohistochemical studies [40,41]. These genes are involved in cell cycle regulation [5].
Cox2 is expressed in response to tumor necrosis factor, epithermal growth factor that is common deregulation in many cancers including head and neck cancers [41,42]. This gene is associated with increased angiogenesis and cellular proliferation [5]. The studies showed the increased Cox2 expression in CA-ex-PA compared to PA [43]. Fibroblast growth factors (FGF) have a role in anti-apoptosis tumor growth, differentiation, and angiogenesis [44]. Over expression of FGF2 protein in myoepithelial cells of CA-ex-PA have been described [45]. On the other hand, FGFR1 is expressed in PAs and FGF2 negative resulting in this tumor observed, but is strongly expressed in CA-ex-PA [46].
Transforming growth factor beta 1 (TGFB1) is a tumor suppressor gene in the early stages of malignancy. In advanced stages of cancer, it plays a role in the survival, progression, and metastasis, angiogenesis [5]. According to studies, over expression of TGFB1 in the high grade carcinoma of CA-ex-PA has been observed [46]. In addition, HGFA (scatter factor), C-Met (a proto-oncogene) were strongly expressed in CA-ex-PA [46]. Epithermal growth factor receptor (EGFR) is important in invasive progression of CA-ex-PA from intracapsular to frank invasion, and essential for motility and matrix invasion [5]. Katori, et al. noted that EGFR is strongly positive in high grade CA-ex-PA [5,10].
The potential role of vascular endothelial growth factor (VEGF), and glucose transporter 1 (Glut1) in carcinogenesis have been analysis. According to Wonkim's study, these markers were more immunoreactive in CA-ex-PA than in PAs [18]. Cell adhesion molecules such as E-cadherin, neural cell adhesion molecule (N-CAM) and beta-catenin have an important role in cell to cell adhesion and maintaining epithelial morphology [5]. In some studies, described cell adhesion molecular expression in high grade CA-ex-PA has been noted [47-49].
Diagnosis
Histopathological examination is the gold standard for the diagnosis of CA-ex-PA. Other non-aggressive methods such as fine needle aspiration cytology (FNAC), sonography, computed tomography (CT) scan and magnetic resonance imaging (MRI) scan can be help to detect the differential diagnosis. But, the studies in about the effect of these methods in diagnosis of CA-ex-PA are few [5]. According to the studies, in CT diagnosis of head and neck lesions, ring or rim enhancement on contrast-enhanced CT imaging has been observed. In Sano's study, an enlarged mass with a central low-density area and an irregular rim enhancement were showed [49]. In contrast to this study, Som's study described the aggressive area in the CA-ex-PA as having "a necrotic center, thick, irregular walls, and infiltrating margins on CT" [50]. Thus, the irregular ring enhancement on the image was considered to reflect this histopathologic condition [49]. In the Kato's study also showed the use of diffusion-weighted MRI to enhance the differentiation of cancer area in CA-ex-PA [51].
Furthermore, FNAC is the most pre-operatively method for diagnosis CA-ex-PA. The main disadvantage of FNAC is insufficient sensitivity related to sampling error rather than misinterpretation of cytologic smears [10]. Most false-negative finding was misdiagnosed as PA [52]. The some studies showed that high grade malignancy was more likely to be diagnosed than low grade malignancy [53]. However, there is not any pre-operative methods cannot be used alone for the diagnosis of CA-ex-PA [5].
Treatment
Primary treatment for CA-ex-PA involves ablative or radical resective procedure, which the surgery dependent to the location and the amount of progression of the tumor and involvement of adjacent structures [3,5].
Because the most location of CA-ex-PA is the parotid gland, the ablative surgery often involves parotidectomy. Superficial parotidectomy should be done for intracapsular or non-invasive and minimally invasive cases of CA-ex-PA with involvement of superficial lobe of the parotid gland. The total or radical parotidectomy is also indicated for the more invasive cases of CA-ex-PA cases [5].
Total parotidectomy involves the resection of both the deep and superficial lobes of the parotid gland. In the total resection, surgeon should be attempting to preserve the facial nerve. However, in the cases of CA-ex-PA with the facial nerve involvement, a radical parotidectomy including resection en bloc of the facial nerve along with the deep and superficial lobes of the parotid is performed [1,8].
Furthermore, neck dissection is necessary for the patients with evidence of cervical lymph node involvement. Neck dissection can be functional, modified, or radical [1,5,8]. According to the most literature, the risk of occult nodal metastasis in the clinically negative neck is not established [10]. The need for elective neck dissection in the patients with CA-ex-PA is still controversy in the otolaryngology literature. However, the study of Lim suggested that all patients with N+ neck disease underwent therapeutic neck dissection, although the extent of neck dissection was not consistently carried out [54]. Furthermore, Zhao's study recommended that neck dissection should be performed for the most of CA-ex-PA patients, except for some intracapsular or minimally invasive tumors [3]. 25% to 33% temporary facial nerve and 2.3% to 6% permanent facial palsy have been observed after surgery [55,56]. The risk of facial palsy depends on the histopatholgy, location and the size of the CA-ex-PA [7].
Immediate reconstructive surgery may be performed after surgical procedure to remove the tumor. Immediate nerve grafting, soft tissue reconstruction like radial for example free flap, a sternomastoid flap or a cervical rotation flap can be performed in some patients [5].
Postoperative radiation in salivary gland tumors is controversial [57]. Postoperative radiotherapy usually performed for high grade malignancy especially in the patients with questionable resection adequacy, lymph node and peri-neural invasion [3,27]. Furthermore, the patients treated with postoperative radiation had better local control rates despite the significantly more incidence of positive margins, peri-neural invasion, facial nerve and lymph node involvement [8,58]. Thus, the more studies showed the effective role of radiation in the central of cellular proliferation [58]. In contrast, the Chen's study showed the postoperative radiotherapy have not effect on survival rates [58,59].
Combined radiotherapy and chemotherapy may be suggested to the patients There is a few studies on the effectiveness of chemotherapy in the management of CA-ex-PA [1,7,27]. However chemoradiotherapy is usually used for the cases with distant metastasis [7].
Prognosis
The prognosis of CA-ex-PA depends on the histopathological staging including the degree of invasion, lymph node involvement, and regional or distant metastasis [15,34]. According to the studies, CA-ex-PA has a 5 year survival of as low as 37%, locoreginal recurrence in 55%, and distant metastasis in up to 42% of patients [16]. Also, overall 5 years survival for invasive and non-invasive carcinoma was 80% to 100% respectively. However, there is a wide range in survival rates of this cancer that is associated with different clinicopathological parameters. First of all parameters, the extent of invasion on histopathological feature of CA-ex-PA has been shown to the correlate with recurrence and prognosis. Invasive CA-ex-PA compared with the non-invasive type has a better prognosis [54,59]. Furthermore, histopathological grade identified as the predictors of CA-ex-PA outcome. The patients with high-grade CA-ex-PA showed a poor prognosis than those with low-grade CA-ex-PA [17]. Thus, the survival rate of CA-ex-PA was worse than the most malignant salivary gland tumors [5].
There are few studies on the effect of peri-neural invasion on the prognosis of CA-ex-PA [3]. Luers, et al. showed that 23% of patients have facial palsy [27]. In Zhao's study, 37% of patients with CA-ex-PA have facial paralysis. According to these studies, the peri-neural invasion had associated with locoregional recurrence, distant metastasis, and survival rate. Also, peri-neural invasion was identified as an independent prognostic factor for this tumor [3].
In addition, the prognosis of CA-ex-PA depends on the complete of the tumor removal. Accurate diagnosis and radical surgery and radiotherapy can be increasing the survival rates of patients with CA-ex-PA [34]. One of the major prognostic factors for patients with CA-ex-PA is loco-regional recurrence [5]. In Olsen's study, the local recurrence in 23% and regional recurrence in 18% of the patients have been observed. After detection of progression or recurrence, the prognosis of CA-ex-PA is poor with a median survival of less than 1 year [8].
The management of neck disease in malignant tumors can be effect on survival rate. The most studies suggested that all patients with N+ neck disease should be neck dissection [54]. In addition, adjuant radiation was associated with a better prognosis in the patients with > 1 positive lymph node. However, the presence of multiple nodal metastases has relation with poor prognosis [59].
However, the most studies showed despite having similar stage of cancer and surgical resection with CA-ex-PA had a lower disease-specific survival rate compared to non-CA-ex-PA high grade primary parotid cancer [54]. In addition, histological grade, T stage, lymph node involvement, peri-neural invasion and extent of invasion are important prognostic factors of CA-ex-PA in the major salivary glands [3].
Conclusion
CA-ex-PA is an uncommon malignant tumor with the particular biological behavior in contrast to the other salivary gland neoplasm. It is important to be aware of the tumor and early detection of pleomorphic adenoma is important for the prevention of Ca-ex-PA.
References
-
Nouraei SA, Hope KL, Kelly CG, McLean NR, Soames JV (2005) Carcinoma ex benign pleomorphic adenoma of the parotid gland. Plast Reconstr Surg 116: 1206-1213.
-
Thompson L (2006) World Health Organization classification of tumours: pathology and genetics of head and neck tumours. Ear Nose Throat J 85: 74.
-
Zhao J, Wang J, Yu C, Guo L, Wang K, et al. (2013) Prognostic factors affecting the clinical outcome of carcinoma ex pleomorphic adenoma in the major salivary gland. World J Surg Oncol 11: 180.
-
Zbaren P, Zbaren S, Caversaccio MD, Stauffer E (2008) Carcinoma ex pleomorphic adenoma: diagnostic difficulty and outcome. Otolaryngol Head Neck Surg 138: 601-605.
-
Antony J, Gopalan V, Smith RA, Lam AK (2012) Carcinoma ex pleomorphic adenoma: a comprehensive review of clinical, pathological and molecular data. Head Neck Pathol 6: 1-9.
-
Torabinia N, Khalesi S (2014) Clinicopathological study of 229 cases of salivary gland tumors in Isfahan population. Dent Res J (Isfahan) 11: 559-563.
-
Yoshida N, Hara M, Kanazawa H, Iino Y (2013) Large carcinoma ex pleomorphic adenoma of the parotid gland: a case report and review of literature. J Oral Maxillofac Surg 71: 2196.
-
Olsen KD, Lewis JE (2001) Carcinoma ex pleomorphic adenoma: a clinicopathologic review. Head Neck 23: 705-712.
-
Matsubayashi S, Yoshihara T (2007) Carcinoma ex pleomorphic adenoma of the salivary gland: an immunohistochemical study. Eur Arch Otorhinolaryngol 264: 789-795.
-
Nadershah M, Alshadwi A, Salama A (2013) An unusual case of carcinoma ex pleomorphic adenoma of the parotid metastasizing to the mandible: case report and review of the literature. J Oral Maxillofac Surg 71: 437-442.
-
Katabi N, Ghossein R, Ho A, Dogan S, Zhang L, et al. (2015) Consistent PLAG1 and HMGA2 abnormalities distinguish carcinoma ex-pleomorphic adenoma from its de novo counterparts. Hum Pathol 46: 26-33.
-
Hayes MM, Lesack D, Girardet C, Del Vecchio M, Eusebi V (2005) Carcinoma ex-pleomorphic adenoma of the breast. Report of three cases suggesting a relationship to metaplastic carcinoma of matrix-producing type. Virchows Arch 446: 142-149.
-
Dewan K, Owens J, Silvester K (2007) Maintaining a high level of suspicion for recurrent malignant disease: report of a case with periapical involvement. Int Endod J 40: 900-907.
-
Baredes S, Ludwin DB, Troublefield YL, Langer PD, Mirani N (2003) Adenocarcinoma ex-pleomorphic adenoma of the lacrimal sac and nasolacrimal duct: a case report. Laryngoscope 113: 940-942.
-
Gnepp DR (1993) Malignant mixed tumors of the salivary glands: a review. Pathol Annu 28 Pt 1: 279-328.
-
Seethala RR (2011) Histologic grading and prognostic biomarkers in salivary gland carcinomas. Adv Anat Pathol 18: 29-45.
-
Lewis JE, Olsen KD, Sebo TJ (2001) Carcinoma ex pleomorphic adenoma: pathologic analysis of 73 cases. Hum Pathol 32: 596-604.
-
Kim JW, Kwon GY, Roh JL, Choi SH, Nam SY, et al. (2011) Carcinoma ex pleomorphic adenoma of the salivary glands: distinct clinicopathologic features and immunoprofiles between subgroups according to cellular differentiation. J Korean Med Sci 26: 1277-1285.
-
el-Naggar AK, Lovell M, Callender DL, Ordonez NG, Killary AM (1998) Concurrent cytogenetic, interphase fluorescence in situ hybridization and DNA flow cytometric analyses of a carcinoma ex-pleomorphic adenoma of parotid gland. Cancer Genet Cytogenet 107: 132-136.
-
Altemani A, Martins MT, Freitas L, Soares F, Araujo NS, et al. (2005) Carcinoma ex pleomorphic adenoma (CXPA): immunoprofile of the cells involved in carcinomatous progression. Histopathology 46: 635-641.
-
Martins C, Fonseca I, Roque L, Pereira T, Ribeiro C, et al. (2005) PLAG1 gene alterations in salivary gland pleomorphic adenoma and carcinoma ex-pleomorphic adenoma: a combined study using chromosome banding, in situ hybridization and immunocytochemistry. Mod Pathol 18: 1048-1055.
-
Katabi N, Gomez D, Klimstra DS, Carlson DL, Lee N, et al. (2010) Prognostic factors of recurrence in salivary carcinoma ex pleomorphic adenoma, with emphasis on the carcinoma histologic subtype: a clinicopathologic study of 43 cases. HUM PATHOL 41: 927-934.
-
Nishijima T, Yamamoto H, Nakano T, Nakashima T, Taguchi K, et al. (2015) Dual gain of HER2 and EGFR gene copy numbers impacts the prognosis of carcinoma ex pleomorphic adenoma. Hum Pathol 46: 1730-1743.
-
Voz ML, Agten NS, Van de Ven WJ, Kas K (2000) PLAG1, the main translocation target in pleomorphic adenoma of the salivary glands, is a positive regulator of IGF-II. Cancer Res 60: 106-113.
-
Hashimoto K, Yamamoto H, Shiratsuchi H, Nakashima T, Tamiya S, et al. (2011) S100P expression in ductal type of carcinoma ex pleomorphic adenoma. Am J Surg Pathol 35: 346-355.
-
Demasi AP, Furuse C, Soares AB, Altemani A, Araujo VC (2009) Peroxiredoxin I, platelet-derived growth factor A, and platelet-derived growth factor receptor alpha are overexpressed in carcinoma ex pleomorphic adenoma: association with malignant transformation. Hum Pathol 40: 390-397.
-
Luers JC, Wittekindt C, Streppel M, Guntinas-Lichius O (2009) Carcinoma ex pleomorphic adenoma of the parotid gland. Study and implications for diagnostics and therapy. Acta Oncol 48: 132-136.
-
Negahban S, Daneshbod Y, Shishegar M (2006) Clear cell carcinoma arising from pleomorphic adenoma of a minor salivary gland: Report of a case with fine needle aspiration, histologic and immunohistochemical findings. Acta Cytol 50: 687-690.
-
Sherif S. Pathology of Carcinoma Ex Pleomorphic Adenoma.
-
Nakamori K, Ohuchi T, Hasegawa T, Hiratsuka H (2009) Carcinoma ex pleomorphic adenoma of the buccal region is composed of salivary duct carcinoma and squamous cell carcinoma components. Int J Oral Maxillofac Surg 38: 1116-1118.
-
Cheuk W, Chan JK (2007) Salivary gland tumors. In: Fletcher CD, editor. Diagnostic histopathology of tumors. London: Elsevier 239-326.
-
Di Palma S, Skalova A, Vanieek T, Simpson RH, Starek I, et al. (2005) Non-invasive (intracapsular) carcinoma ex pleomorphic adenoma: recognition of focal carcinoma by HER-2/neu and MIB1 immunohistochemistry. Histopathology 46: 144-152.
-
Brandwein M, Huvos AG, Dardick I, Thomas MJ, Theise ND (1996) Noninvasive and minimally invasive carcinoma ex mixed tumor: a clinicopathologic and ploidy study of 12 patients with major salivary tumors of low (or no?) malignant potential. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 81: 655-664.
-
Barnes L, Eveson JW, Reichart P, Sidransky D, editors (2005) World health organization classification of tumors. Pathology and genetics of head and neck tumors. Lyon: IARC Press.
-
El-Naggar AK, Callender D, Coombes MM, Hurr K, Luna MA, et al. (2000) Molecular genetic alterations in carcinoma ex-pleomorphic adenoma: a putative progression model? Genes Chromosomes Cancer 27: 162-168.
-
Roijer E, Nordkvist A, Strom AK, Ryd W, Behrendt M, et al. (2002) Translocation, deletion/amplification, and expression of HMGIC and MDM2 in a carcinoma ex pleomorphic adenoma. Am J Pathol 160: 433-440.
-
Namboodiripad PC (2014) A review: Immunological markers for malignant salivary gland tumors. J Oral Biol Craniofac Res 4: 127-134.
-
Fowler MH, Fowler J, Ducatman B, Barnes L, Hunt JL (2006) Malignant mixed tumors of the salivary gland: a study of loss of heterozygosity in tumor suppressor genes. Mod Pathol 19: 350-355.
-
Ihrler S, Weiler C, Hirschmann A, Sendelhofert A, Lang S, et al. (2007) Intraductal carcinoma is the precursor of carcinoma ex pleomorphic adenoma and is often associated with dysfunctional p53. Histopathology 51: 362-371.
-
Patel RS, Rose B, Bawdon H, Hong A, Lee CS, et al. (2007) Cyclin D1 and p16 expression in pleomorphic adenoma and carcinoma ex pleomorphic adenoma of the parotid gland. Histopathology 51: 691-696.
-
Tarakji B, Nassani MZ, Sloan P (2010) Immunohistochemical expression of estrogens and progesterone receptors in carcinoma ex pleomorphic adenoma-undifferentiated and adenocarcinoma types. Med Oral Patol Oral Cir Bucal 15: e432-6.
-
Kawata R, Hyo S, Araki M, Takenaka H (2010) Expression of cyclooxygenase-2 and microsomal prostagalandin E synthase-1 in head and neck squamous cell carcinoma. Auris Nasus Larynx 37: 482-487.
-
Katori H, Nozawa A, Tsukuda M (2007) Increased expression of cyclooxygenase-2 and Ki-67 are associated with malignant transformation of pleomorphic adenoma. Auris Nasus Larynx 34: 79-84.
-
Powers CJ, McLeskey SW, Wellstein A (2000) Fibroblast growth factors, their receptors and signaling. Endocr Relat Cancer 7: 165-197.
-
Martinez EF, Demasi AP, Miguita L, Altemani A, Araujo NS, et al. (2010) FGF-2 is overexpressed in myoepithelial cells of carcinoma ex-pleomorphic adenoma in situ structures. Oncol Rep 24: 155-160.
-
Furuse C, Miguita L, Rosa AC, Soares AB, Martinez EF, et al. (2010) Study of growth factors and receptors in carcinoma ex pleomorphic adenoma. J Oral Pathol Med 39: 540-547.
-
Prabhu S, Kaveri H, Rekha K (2009) Benign, malignant salivary gland tumors: comparison of immunohistochemical expression of e-cadherin. Oral Oncol 45: 594-599.
-
Genelhu MC, Gobbi H, Arantes DC, Cardoso SV, Cassali GD (2007) Immunolocalization of beta-catenin in pleomorphic adenomas and carcinomas ex-pleomorphic adenomas of salivary glands. Appl Immunohistochem Mol Morphol 15: 273-278.
-
Sano K, Fujita S, Sekine J, Uehara M, Sakihama N, et al. (2012) Metachronous manifestation of carcinoma ex pleomorphic adenoma in a buccal minor salivary gland and the contralateral parotid gland: a case report and review of the literature. J Oral Maxillofac Surg 70: 2701-2712.
-
Som PM, Brandwein-Gensler MS (2011) Anatomy and pathology of the salivary gland, in Som PM, Curtin HD, Head and Neck Imaging (5 edn), St Louis, MO, Elsevier 2525-2535.
-
Kato H, Kanematsu M, Mizuta K, Ito Y, Hirose Y (2008) Carcinoma ex pleomorphic adenoma of the parotid gland: radiologic-pathologic correlation with MR imaging including diffusion-weighted imaging. AJNR Am J Neuroradiol 29: 865-867.
-
Stodulski D, Rzepko R, Kowalska B, Stankiewicz C (2007) Carcinoma ex pleomorphic adenoma of major salivary glands--a clinicopathologic review. Otolaryngol Pol 61: 687-693.
-
Klijanienko J, El-Naggar AK, Vielh P (1999) Fine-needle sampling findings in 26 carcinoma ex pleomorphic adenomas: diagnostic pitfalls and clinical considerations. Diagn Cytopathol 21: 163-166.
-
Lim CM, Hobson C, Kim S, Johnson JT (2015) Clinical outcome of patients with carcinoma ex pleomorphic adenoma of the parotid gland: a comparative study from a single tertiary center. Head Neck 37: 543-547.
-
Guntinas-Lichius O, Klussmann JP, Wittekindt C, Stennert E (2006) Parotidectomy for benign parotid disease at a university teaching hospital: outcome of 963 operations. Laryngoscope 116: 534-540.
-
Koch M, Zenk J, Iro H (2010) Long-term results of morbidity after parotid gland surgery in benign disease. Laryngoscope 120: 724-730.
-
Terhaard CH, Lubsen H, Rasch CR, Levendag PC, Kaanders HH, et al. (2005) The role of radiotherapy in the treatment of malignant salivary gland tumors. Int J Radiat Oncol Biol Phys 61: 103-111.
-
Chen AM, Bucci MK, Singer MI, Garcia J, Kaplan MJ, et al. (2007) Intraoperative radiation therapy for recurrent head-and-neck cancer: the UCSF experience. Int J Radiat Oncol Biol Phys 67: 122-129.
-
Chen MM, Roman SA, Sosa JA, Judson BL (2014) Predictors of survival in carcinoma ex pleomorphic adenoma. Head Neck 36: 1324-1328.





