International Journal of Pathology and Clinical Research
Japanese Crisis of HPV Vaccination
Masayuki Sekine1*, Risa Kudo1, Sosuke Adachi1, Manako Yamaguchi1, Yutaka Ueda2, Tomomi Takata2, Akiko Morimoto2, Yusuke Tanaka2, Asami Yagi2, Etsuko Miyagi3 and Takayuki Enomoto1
1Department of Obstetrics and Gynecology, Niigata University Graduate School of Medical and Dental Science, Niigata, Japan
2Department of Obstetrics and Gynecology, Osaka University Graduate School of Medicine, Osaka, Japan
3Department of Obstetrics, Gynecology and Molecular Reproductive Science, Yokohama City University Graduate School of Medicine, Yokohama, Japan
*Corresponding author:
Masayuki Sekine, MD, PhD, Department of Obstetrics and Gynecology, Niigata University Graduate School of Medical and Dental Science, 1-757 Asahimachi-dori, Niigata 951-8510, Japan, Tel: +81-25-227-2320, Fax: +81-25-227-0789, E-mail: masa@med.niigata-u.ac.jp
Int J Pathol Clin Res, IJPCR-2-039, (Volume 2, Issue 2), Review Article; ISSN: 2469-5807
Received: April 26, 2016 | Accepted: May 25, 2016 | Published: May 30, 2016
Citation: Sekine M, Kudo R, Adachi S, Yamaguchi M, Ueda Y, et al. (2016) Japanese Crisis of HPV Vaccination Int J Pathol Clin Res 2:039. 10.23937/2469-5807/1510039
Copyright: © 2016 Sekine M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Cervical cancer has been increasing in younger generation, especially in 20's and 30's, so it has been a serious problem that the disease leads to loss of fertility as an urgent issue in Japan. HPV vaccine was added to the national list of routine vaccinations for Japanese girls aged 12 to 16 years by law since April, 2013. Three months later, however, the Japanese Ministry of Health, Labour, and Welfare suspended recommendations for HPV vaccination after several reports of possible adverse events. We investigated the HPV vaccination rates in 7-11th grade girls (ages 12 to 16 years) in Niigata from April, 2011 to March, 2015 (from 2011 to 2014 fiscal year) to clarify a recent trend of the HPV vaccination rates in Japan in detail. As a result, HPV vaccination rate was 63.3% in 7-11th grades (ages 12 to 16 years), especially among them, that in 7th grade was 82.7% at the end of March, 2012. After the suspension of the government's recommendation, the vaccination rates of the 7-11th graders were 3.5% and 0.19% in March, 2014 and 2015, respectively. In conclusion, in case of continuation of the suspension of recommendations for HPV vaccination, it will leave anxiety about a possibility that only Japan will become a country where the incidence of cervical cancer is still high in the future.
Keywords
Cervical cancer, HPV vaccination, Japanese crisis
Background
Approximately 10,000 women are diagnosed with cervical cancer every year, with nearly 3,000 deaths each year in Japan [1]. Cervical cancer has been increasing in younger generation, especially in 20's and 30's [2]. It has been a serious problem that cervical cancer leads to loss of fertility as an urgent issue in Japan. Human papillomavirus (HPV) vaccination has proven to be effective for prevention of HPV infection and HPV-associated precancerous cervical lesions [3,4]. Free HPV vaccinations became available to Japanese girls aged 13 to 16 years with public aid from 2010 and the vaccine was added to the national list of routine vaccinations for girls aged 12 to 16 years by lawsince April, 2013. HPV vaccination has been available for women aged 15-45 years old other than the target age by law at their private expense without government support. Three months later, however, the Japanese Ministry of Health, Labour, and Welfare suspended recommendations for HPV vaccination after several reports of possible adverse events such as chronic pain and motor impairment, although a causal relationship between HPV vaccination and these symptoms could not be discovered. Recently, it was reported that the suspension of recommendations for vaccination has decreased the vaccination rate to a low 3% in 2013 [5]. Here, we aimed to clarify a more recent trend of the HPV vaccination rates in Japan in detail.
Methods
We investigated the HPV vaccination rates in 7-11th grade girls (ages 12 to 16 years) in Niigata, a city in northwest Japan with a population of 8 hundred thousand people, from April, 2011 to March, 2015 (from 2011 to 2014 fiscal year). The fiscal year in Japan starts from April and ends in March. The Niigata city used to be one of the cities with highest rates of HPV vaccination in Japan. At present, we have conducted a large population-based cohort study to assess the effect of HPV vaccination in Japanese girls in the city. We obtained the data concerning the number of HPV vaccination and target girls from the Niigata city public health center. The HPV vaccination by public expenditure support in Niigata city started for all girls in 7th-11th grade students from January, 2011. The National HPV Vaccination established in April, 2013. After that, suspension of the government's recommendation for the vaccination happened on June 14, 2013.
Results
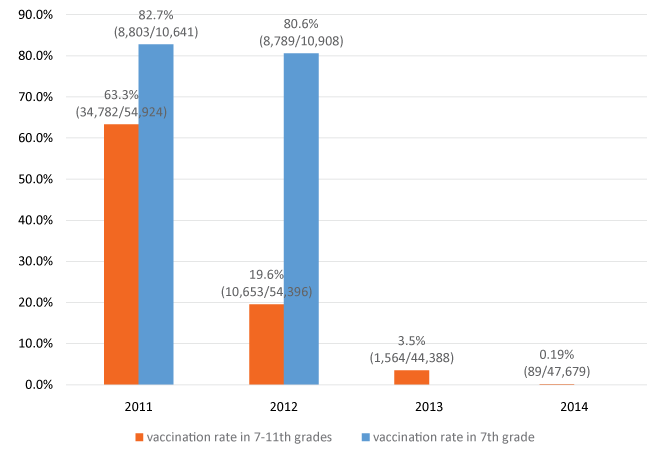
The HPV vaccination rate in Niigata is shown in figure 1. We analyzed the rate by the number of injections in 7-11th girls (ages 12 to 16 years) from 2011 to 2014 fiscal year. The target girls for vaccination was 18,308 girls in 2011 fiscal year, 18,132 in 2012, 14,796 in 2013, and 15,893 in 2014, respectively. Three injections are available to prevent the HPV infection, so the number of injection for target girls was 54,924 in 2011 fiscal year, 54,396 in 2012, 44,388 in 2013, and 47,679 in 2014, respectively. As a result, the HPV vaccination rate was 63.3% (34,782/54,924) in 7-11th grades, especially among them, that in 7th grade was 82.7% (8,803/10,641) at the end of March, 2012. Vaccination targets were 7-11th graders, however, vaccinations at the 7th-grade was considered the most desirable. Hence, the target girls received HPV vaccination mostly in the 7th grade. Although the vaccination rate was 19.6% (10,653/54,396) in 7-11th grades in March, 2013, the rate in 7th grade was maintained at 80.6% (8,789/10,908). However, the suspension of the government's recommendation for the vaccination on June, 2013 has led to public distrust and a dramatic decrease in HPV vaccination rates. After that event, the vaccination rates of the 7-11th graders were 3.5% (1,564/44,388) and 0.19% (89/47,679) in March, 2014 and 2015, respectively. There were 15,893 candidate girls for public support vaccination at March, 2015, however, the girls who received HPV vaccination among them was less than 30.
.
Figure 1: HPV vaccination rate in Niigata, Japan-HPV vaccination rate was 63.3% in 7-11th grades (ages 12 to 16 years), especially among them, that in 7th grade was 82.7% at the end of March, 2012. Although the vaccination rate was 19.6% in 7-11th grades in March, 2013, the rate in 7th grade was maintained at 80.6%. After the suspension of the government's recommendation for the vaccination in June, 2013, the vaccination rates of the 7-11th graders were 3.5% and 0.19% in March, 2014 and 2015, respectively. There is no data about the vaccination rate in 7th graders in 2014-2015.
View Figure 1
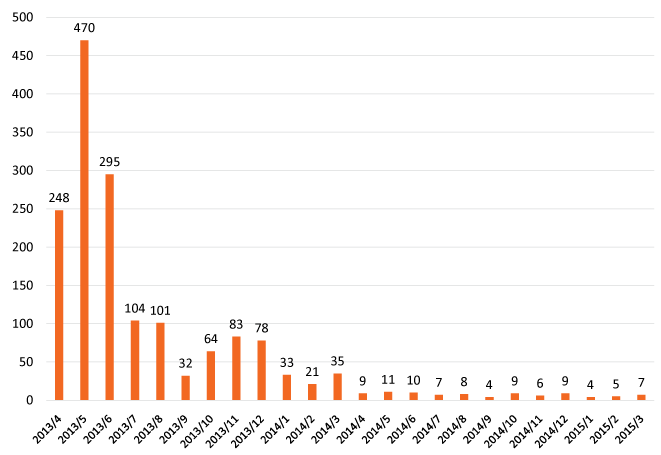
Figure 2 demonstrates the transition of the number of HPV vaccination from April, 2013 to March, 2015. The number of HPV vaccination was the times of injection per month. The number of candidates for vaccination in April, 2013 was approximately 15,000 girls, so it is estimated that the times of injection for the candidates will be 45,000. Hence, the average of injection will be calculated at approximately 3,700 times each month (45,000 times/12 months). Although there were still 470 injections in May, 2013, the number of vaccination dramatically decreased after the suspension of the government's recommendation, finally, there were only 7 injections in March, 2015.
.
Figure 2: Number of HPV vaccination in Niigata, Japan-The number of HPV vaccination was the times of injection per month. Although there were still 470 injections in May, 2013, the number of vaccination dramatically decreased after the suspension of the government's recommendation. Finally, there was only 7 injections in March, 2015.
View Figure 2
Discussion
Although, so far, there has been no study which provides scientific or epidemiologic evidence to demonstrate the causal relationship between HPV vaccination and adverse reactions, the HPV vaccination rate has decreased more and more after the suspension of the government's recommendation. At last the rates have been almost 0% in Japan. Two reports have been previously published in this issue. Ueda et al. reported that the vaccination rates of the 7-10th graders who had an opportunity for vaccination prior to the media reports were between 57.8-74.8% (2012), however, following the adverse reports, the rate plunged from 65.4% (2012) to 3.9% (2013) as titled "Japan's failure to vaccinate girls against human papillomavirus" [5]. Hanley et al. consisted "HPV vaccination crisis in Japan", and they reported that HPV vaccination after suspension completion rates plummeted to just 0.6% in Sapporo, Japan in March, 2014 [6]. According to the comprehensive survey of National Cancer Center, the percentage of women who have received cervical cancer screening is approximately 30% in Japan, which is obviously lower than those in Western countries, so it is difficult that the screening test reduces the number of deaths from cervical cancer alone [7]. Both HPV vaccination and the screening test are required for the prevention of cervical cancer.
It was reported that the total number of adverse reactions, such as chronic pain and motor impairment, was 176 among 8,898,000 girls vaccinated in Japan, so the incidence of adverse reactions is equivalent to 2.0 per 100,000 vaccinations at the meeting of the Investigative Committee on Adverse Reactions in July 2014 [8,9]. Starting from this year, "Result presentation of scientific research for reactions after HPV vaccination by Health, Labour and Welfare" was held on March 16, 2016. Principal investigator reported in the meeting that there are many obscure points in the cause and etiology of adverse reactions in HPV vaccination at present, subsequently, the Ministry of Health, Labor, and Welfare released the statement on April 18, 2016 that the cause-and-effect relationship between a vaccination and an adverse reaction, such as memory disorder, was not shown [10,11]. However, the news media emphatically reported the severe events after vaccination repeatedly as if the events were caused by HPV vaccination. The most important aspects are that the media should give Japanese parents and girls the precise knowledge and information of adverse reactions, as well as HPV vaccine effectiveness against cervical cancer.
In the global health community, the World Health Organization (WHO), International Federation of Gynecology and Obstetrics (FIGO), and American Society of Clinical Oncology (ASCO) published the statements supporting the safety and effectiveness of the HPV vaccination, and the necessity for the continuation of HPV vaccination in adolescent boys and girls as a national strategy [12-14]. Furthermore, the WHO published the declaration which is apprehensive about a current situation in Japan [12]. The Japan Society of Obstetrics and Gynecology (JSOG) strongly demand the immediate resumption of recommendations for HPV vaccination, with the aim of eradicating cervical cancer [9].
In conclusion, in case of continuation of the suspension of recommendations for HPV vaccination, it will leave anxiety about a possibility that only Japan will become a country where the incidence of cervical cancer is still high in the future. If the situation goes on like this, Japanese girls will lose the benefits of HPV vaccination for cervical cancer prevention before they know.
Funding
Dr. Sekine, Ueda, Miyagi, and Enomoto received research funding from Japan Agency for Medical Research and Development (Grant No. 15ck0106103h0102) and Merck Sharp & Dohme, a lecture fee from Merck Sharp & Dohme and Glaxo Smith Kline/ Japan Vaccine, and advisory fee from Merck Sharp & Dohme.
Dr. Miyagi received lecture fees from Roche Diagnostic K. K., Japan Vaccine Co., Ltd., QIAGEN K. K.-Japan, and received honoraria for supervision of educational materials from Roche Diagnostic K. K., Japan Vaccine Co. Ltd.
Financial support
A Health and Labor Research Grant, and a Japan Agency for Medical Research and Development Grant, supported this research. The grants were H26-innovative-cancer research-general-102 and 15ck0106103h0102.
References
-
Cancer statistics, Center for Cancer Control and Information Services, National Cancer Center, Japan.
-
Motoki Y, Mizushima S, Taguri M, Takahashi K, Asano R, et al. (2015) Increasing trends in cervical cancer mortality among young Japanese women below the age of 50 years: An analysis using the kanagawa population-based cancer registry, 1975-2012. Cancer Epidemiol 39: 700-706.
-
Ault KA, Future II Study Group (2007) Effect of prophylactic human papillomavirus L1 virus-like-particle vaccine on risk of cervical intraepithelial neoplasia grade 2, grade 3, and adenocarcinoma in situ: a combined analysis of four randomised clinical trials. Lancet 369: 1861-1868.
-
Paavonen J, Jenkins D, Bosch FX, Naud P, Salmeron J, et al. (2007) Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: an interim analysis of a phase III double-blind,randomised controlled trial. Lancet 369: 2161-2170.
-
Ueda Y, Enomoto T, Sekine M, Egawa-Takata T, Morimoto A, et al. (2015) Japan's failure to vaccinate girls against human papillomavirus. Am J ObstetGynecol 212: 405-406.
-
Hanley SJ, Yoshioka E, Ito Y, Kishi R (2015) HPV vaccination crisis in Japan. Lancet 385: 2571.
-
Comprehensive Survey of Living Conditions: Center for Cancer Control and Information Services, National Cancer Center, Japan.
-
The 10th Vaccination Sub-committee Meeting: Investigative Committee on Adverse Reactions, Health Sciences Council, the Japanese Ministry of Health, Labour, and Welfare (July 4, 2014).
-
Fujii T (2015) Declaration to demand the resumption of recommendations for human papillomavirus (HPV) vaccination for cervical cancer prevention. J Obstet Gynaecol Res 41: 1859-1860.
-
Result presentation of scientific research for reactions after HPV vaccination by the Japanese Ministry of Health, Labour and Welfare (March 16, 2016).
-
http://www.mhlw.go.jp/bunya/kenkou/kekkaku-kansenshou28/tp160316.html
-
GACVS (Global Advisory Committee on Vaccine Safety) safety update on HPV Vaccines Geneva,13 June 2013.
-
Human papillomavirus vaccines: WHO position paper, October 2014. No. 43, 2014, 89, 465-492.
-
Howard H Bailey, Linus T Chuang, Nefertiti C duPont, Cathy Eng, Lewis E Foxhall, et al. (2016) American Society of Clinical Oncology Statement: Human Papillomavirus Vaccination for Cancer Prevention. J Clin Oncol.





