International Journal of Pathology and Clinical Research
BRAF V600E Immunohistochemistry in Cutaneous Langerhans Cell Histiocytosis: An Analysis of 20 Cases
Ryan C Romano1*, Wonwoo Shon2, Sarah M Jenkins3 and Karen J Fritchie1
1Department of Laboratory Medicine and Pathology, Mayo Clinic, Rochester, Minnesota, USA
2University of Florida College of Medicine, Gainesville, Florida, USA
3Division of Biomedical Statistics and Informatics, Mayo Clinic, Rochester, Minnesota, USA
*Corresponding author:
Ryan C. Romano, D.O., Department of Laboratory Medicine and Pathology, Mayo Clinic, 200 First Street SW, Rochester, MN 55905, USA, Tel: 507-284-2511, Fax: 507-284-1875, E-mail: romano.ryan@mayo.edu
Int J Pathol Clin Res, IJPCR-2-036, (Volume 2, Issue 2), Review Article; ISSN: 2469-5807
Received: March 20, 2016 | Accepted: May 20, 2016 | Published: May 25, 2016
Citation: Romano RC, Shon W, Jenkins SM, Fritchie KJ (2016) BRAF V600E Immunohistochemistry in Cutaneous Langerhans Cell Histiocytosis: An Analysis of 20 Cases. Int J Pathol Clin Res 2:036. 10.23937/2469-5807/1510036
Copyright: © 2016 Romano RC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: The role of BRAF V600E immunohistochemistry (IHC) has not been fully elucidated in cutaneous Langerhans cell histiocytosis (LCH).
Methods: Twenty dermatologic biopsy specimens from 14 patients (9 adults and 5 children) were retrieved. All cases were immunostained with BRAF mutation-specific antibody. Selected BRAF V600E IHC-positive cases were analyzed for BRAF V600E mutation by pyrosequencing. Clinical parameters were correlated with IHC status.
Results: BRAF V600E protein overexpression was identified in 15 of 20 (75%) cases (9/14 patients; 5 adults and 4 children). All pediatric patients with bone involvement were BRAF V600E IHC positive. 4/5 (80%) patients without BRAF V600E expression had CNS involvement (p = 0.02). The presence of BRAF V600E mutation was identified in 1/5 (20%) BRAF V600E IHC-positive cases.
Conclusions: Higher rates of BRAF V600E protein over expression were noted in children. BRAF V600E IHC appeared to correlate with bone involvement and showed a negative correlation with CNS involvement. The discordance between IHC and molecular analysis seen in our study may be indicative of the cellular heterogeneity inherent to LCH, sampling error, or non-specific staining, but also raises the possibility that BRAF IHC may not be a reliable surrogate for mutation status in cutaneous LCH.
Keywords
BRAF, Histiocytosis, Langerhans cell, Immunohistochemistry, VE1
Introduction
Langerhans cell histiocytosis (LCH) is a rare proliferative disorder with a wide spectrum of clinical manifestations. Disease distribution is generally classified as unifocal, multifocal, or disseminated [1]. The skin is the second most common organ system affected by LCH after the skeletal system, with 30-40% of patients developing cutaneous involvement and skin sites predominating in younger patients, specifically those less than 1 year of age [1-3]. LCH has been known by many different eponyms since its initial descriptions over a century ago, primarily because there was a delay in realizing that many entities considered to be clinically distinct diseases were actually minor variants of what we now call LCH [4]. It was not until 1953 that these conditions were unified under a single designation by Lichtenstein, and recent studies have shown that oncogenic BRAF V600E mutations likely play a role in the pathogenesis of LCH, supporting the theory that LCH is a neoplastic process [5-7]. To date there is limited data on the rate of BRAF V600E immunoreactivity and mutation status in cutaneous LCH (Table 1) [6-9]. Furthermore, except for a correlation with younger age, there has been no significant association with BRAF status and clinical parameters [6]. In addition, though the specificity of the commercially available BRAF mutation specific antibody used for immunohistochemistry (IHC) has been shown to be superb in malignant melanoma, studies examining this correlation in cutaneous LCH are limited [10]. Thus, in this study, we sought to explore the presence of BRAF V600E protein overexpression in a series of well-characterized cutaneous LCH with emphasis on clinicopathologic correlation, followed by genotyping in a selected subset of cases to compare BRAF status between immunohistochemical and molecular methods.
![]()
Table 1: Summary of studies evaluating BRAFV600E mutation status in LCH. NT: not tested; WT: wild-type BRAF allele.
View Table 1
Methods
Patients and samples
Twenty dermatologic biopsy specimens of LCH were retrieved from the Mayo Clinic archives. Four cases of non-LC histiocytic proliferation were utilized as controls including: 2 cases of spongiotic dermatitis with histiocytic (Langerhans cells) aggregates, 1 case of juvenile xanthogranuloma, and 1 case of indeterminate cell histiocytosis. Hematoxylin and eosin sections, along with pertinent existing immunohistochemical studies (CD1a, langerin, CD68, S-100 protein), were re-reviewed to confirm the diagnosis of LCH. Following a comprehensive medical record review, staining profiles were correlated with a variety of clinical parameters including age, gender, ethnicity and sites of disease.
Immunohistochemistry
4-μm-thick sections were cut from formalin-fixed, paraffin-embedded tissue and placed on charged slides. Slides were dried and melted at 62 degree C for twenty minutes. Sections were then immunostained with mouse monoclonal BRAF V600E antibody (clone VE1, 1:100, Spring Bioscience, Pleasanton, CA) raised against a synthetic peptide corresponding to amino acids 596-606 (GLATEKSRWSG) of mutated BRAF. The Ventana BenchMarkXT (Ventana Medical Systems, Tucson, AZ) platform was used for slide staining. The staining protocol utilized on-line deparaffinization, heat induced epitope retrieval with Ventana Cell Conditioning 1 for 32 minutes, and primary antibody incubation for 32 minutes at 37 degrees C. Antigen-antibody reactions are visualized using the Ventana OptiViewTM Amplification kit and Ventana OptiviewTM Universal DAB Detection Kit (Optiview HQ Linker 8 minutes, Optiview HRP Multimer 8 minutes, Optiview Amplifier H2O2/Amplifier 4 minutes, Optiview Amplifier Multimer 4 minutes, Optiview H2O2/DAB 8 minutes, Optiview Copper 4 minutes). Counterstaining was performed online using Ventana Hematoxylin II for 8 minutes, followed by bluing reagent for 4 minutes. Slides were then dehydrated and coverslipped for examination. This antibody had been previously validated at our institution using confirmed BRAF-mutated cases of malignant melanoma. Any non-ambiguous cytoplasmic staining of viable tumor cells was considered positive in accordance with previously published scoring strategies, whereby application of such strategies rendered a score of negative for the following situtations: staining of macrophages or monocytes, faint or weak diffuse cytoplasmic staining, rare nuclear staining, or rare staining of isolated interposed cells [11,12].
Mutational analysis
BRAF V600E IHC-positive cases with sufficient residual tissue (n = 6) were then selected for mutational analysis. Following pathologist identification on the hematoxylin and eosin slides of the reference area to be sampled, corresponding tissue from the unstained sections was microdissected using the QIAamp DNA Mini Kit (Qiagen, Valencia, CA). Exon 15 of the BRAF gene was amplified by polymerase chain reaction using the ABI GeneAmp 9700 (Life Technologies, Grand Island, NY). The following PCR primers were used for analysis of the antisense strand of BRAF exon 15: Biotin labeled forward PCR primers 5'-biotin-TGACTGAATAGAACTTGTGGTAGTG-3' and reverse primer 5'-TCGTCCACATATGATTCTAA-3' at final concentration of 0.1μM. The presence or absence of a BRAF V600E mutation was analyzed using the PSQ HS 96 SNP Reagent Kit (Biotage, Charlotte, NC).
Statistical Analysis
The specimen and patient characteristics were summarized with frequencies and percentages for categorical data, and with means and ranges for age. When summarizing the unique patients and for statistical testing, we selected the first observation for patients who had multiple specimens. The presence of involvement from different organ systems was compared with BRAF V600E staining result with Fisher’s exact tests. P-values less than 0.05 were considered statistically significant. All analyses were conducted using SAS version 9 (SAS Institute, Cary, NC).
Results
The results are summarized in table 2. The twenty cases of cutaneous LCH were from 14 patients (9 adults, 5 children; mean age 30.8 years; median age 38 years; range 1 to 78 years). 8 patients were male, and 6 patients were female. BRAF V600E protein overexpression was identified in 15 of the 20 (75%) biopsies. In patients with multiple biopsies, BRAF status by (IHC) was concordant among all biopsies resulting in 9 of 14 (64%) patients harboring BRAF V600E IHC-positive lesions. In the adult group, 5 of 9 (56%, 2 male, 3 female) patients showed positive staining. In the pediatric group, BRAF V600E expression was identified in 4 of 5 patients (80%, 3 males, 1 female). All children with positive staining were 2 years or younger. When immunoreactivity was evaluated only relative to organ system involved, 4 of the 5 (80%) patients with absent staining had involvement of the central nervous system at the time of their only biopsy (Patients 5, 7, 9, 12), and this was statistically significantly greater than what was observed in those with positive staining (1/9, 11%, p = 0.02; Patient 6). Two of the patients with positive staining (Patients 4, 10), both of whom had multiple biopsies which were concordantly IHC-positive, went on to develop central nervous system involvement after the index biopsy. One of the 4 pediatric patients with positive staining had concurrent bone involvement at the time of his only cutaneous biopsy (Patient 2), while the lone pediatric patient with absent staining did not have bone involvement (Patient 5). Two additional pediatric patients (Patients 3, 4), both of whom had multiple biopsies which were concordantly IHC-positive, later developed bone involvement. Bone involvement was seen in four adults: three at the time of their only biopsy (Patients 8, 11, 14) and in one adult at the time of the index biopsy (Patient 10), three of which had positive immunostaining (Patients 8, 10, 14). Among all ages, BRAF V600E protein expression was seen in 3 of the 4 (75%) patients (Patients 2, 8, 14) with concurrent bone involvement at the time of the index (Patient 14) or only (Patients 2, 8) biopsy, though this did not reach statistical significance. No correlations were observed between staining profiles and gender or ethnicity. Clinical and histomorphologic features were not predictive of IHC status (Figure 1). No control cases showed BRAF V600E expression (0/4).
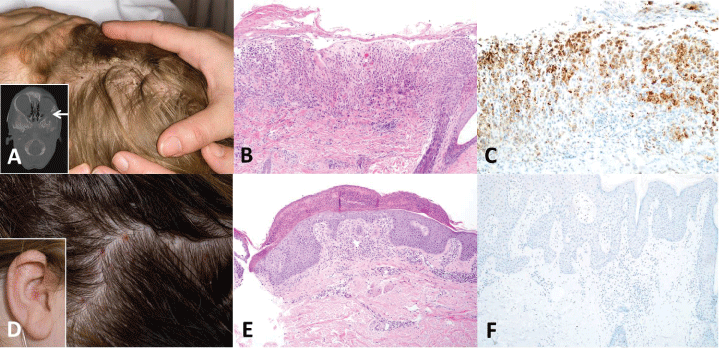
.
Figure 1: The presence and quantitation of lesional cells with the BRAF V600E mutation is difficult to determine based on clinical or histomorphologic features. Patient 4 is a 2 year old boy with scattered yellow brown scaling papules on the scalp (A) and a lytic mass in the lateral wall of the left orbit (inset, arrow). Hematoxylin and eosin sections showed a band-like proliferation of Langerhans cells, scattered eosinophils and multinucleated giant cells (B) and BRAF immunohistochemistry showed strong, diffuse cytoplasmic staining of the lesional cells (C). In contrast, patient 7 is a 37 year old woman with perifollicular erythematous papules on the scalp (D) and coalescing papules within the right ear (inset). Hematoxylin and eosin sections show a predominantly dermal infiltrate of Langerhans cells with admixed eosinophils (E), but BRAF immunohistochemistry is negative (F). (Original magnifications: B-C, F, x200; E, x100).
View Figure 1
![]()
Table 2: Clinical features, immunohistochemistry, and molecular analysis of 14 patients with cutaneous LCH; CNS, central nervous system; GI, gastrointestinal; V600E mutation in BRAF with resultant amino acid change; NT, not tested due to insufficient residual tissue. Statistical analysis included only the first observation/specimen for each patient, and concurrent systemic (other organ system) involvement at the time the earliest biopsy was performed. In some cases, involvement of other organ systems occurred after the index biopsy.
View Table 2
Sufficient residual tissue was available for mutational analysis in 6 cases that exhibited positive staining by IHC. Mutational analysis was successful in 5 of these 6 (83%), but the presence of BRAF V600E mutation was detected in only 1 of the 5 (20%) cases tested.
Discussion
Although Langerhans cell histiocytosis has a complicated history including the use of numerous eponyms, significant advances in the understanding of this entity over the last century have led to the commonly accepted theory that LCH represents a clonal rather than a reactive process, with this concept incorporated into the current World Health Organization definition for LCH [4-5,13]. This is supported by recent work identifying oncogenic BRAF mutations in a majority LCH cases across multiple series [6-8]. However, data regarding BRAF status in cutaneous lesions and its clinical implication is limited.
Our present study demonstrated the presence of BRAF V600E protein overexpression in 75% of LCH cases involving the skin. Similar to previous studies, we did see a correlation between positive immunoreactivity and younger age. The average age of patients with BRAF V600E IHC-positive lesions was 27.8 years compared to an average age of 36.2 years for patients with specimens showing a lack of staining, though this correlation was not statistically significant (p-value = 0.37). Of the 5 pediatric patients included in our study, 4 (80%) showed positive staining, all 4 of which were 2 years of age or below. The single pediatric patient with negative staining was also the oldest (12 years of age) in our pediatric population. Compared to the 80% of pediatric patients with positive staining, immunoreactivity was observed in 56% of adults.
Central nervous system involvement was observed in only 1 of the 9 patients with positive immunoreactivity (11%). In contrast, central nervous system involvement (pituitary stalk lesions, diabetes insipidus) was seen in 4 of the 5 (80%) patients with absent staining, and this correlation was statistically significant (p-value = 0.02). The reasons for this negative correlation are unclear, as to our knowledge, no such observation has been reported previously.
3 of the 4 BRAF V600E IHC-positive pediatric patients had bone involvement at some time during the course of disease (though not necessarily at the time of the index biopsy), whereas the only BRAF V600E IHC-negative pediatric patient was without bone involvement. Thus, oncogenic BRAF mutations may be more specifically implicated in bone involvement in children with LCH.
Six BRAF V600E IHC-positive cases had sufficient residual tissue to be tested for mutational analysis. Unexpectedly, the BRAF V600E mutant allele was identified in only 1 of the 5 specimens in which sequencing was successful (Patient 4). This discordance between the immunohistochemical and genotyping results is intriguing and prompted us to re-review the hematoxylin and eosin sections along with the immunohistochemical studies. Upon re-review of the immunostained slides, the one case confirmed to have the mutant allele by genotyping (Patient #4 in our series) had a significantly higher number of BRAF V600E IHC-positive cells staining positive when compared to the other cases which were wild-type (Figure 2). However, all tissue selected for DNA extraction and pyrosequencing contained at a minimum 15% IHC-positive cells in the most concentrated areas (the areas selectively chosen for analysis), with the majority of cases containing at least 40% IHC-positive cells, well above the limit of detection for BRAF V600E IHC. The pyrosequencing assay utilized in our current study has a validated analytical sensitivity of 5% tumor. Despite adherence to strict interpretation criteria, the number of lesional IHC-positive cells in the 4 negative cases may still have been below the threshold needed for detection by molecular analysis, possibly causing false negative genotyping results in our series, similar to what has been reported by other authors [6,8,14,15]. LCH lesions are highly heterogeneous with a prominent inflammatory component making it difficult to isolate mutated cells, even in areas with a dense proliferation of pathogenic Langerhans cells.
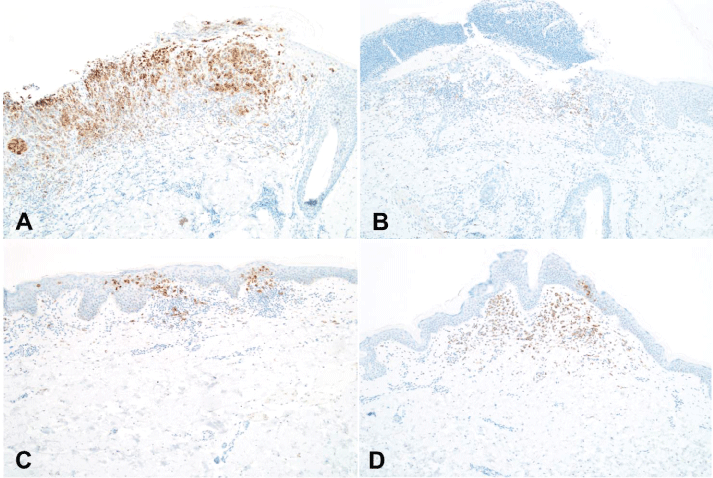
.
Figure 2: BRAF V600E mutational status (presence of the mutant allele) was concordant between IHC and pyrosequencing in patient 4 (A) in which there are numerous BRAF V600E IHC-positive lesional Langerhans cells. In contrast, cases from patients 1 (B), 13 (C), and 3 (D) were wild-type by pyrosequencing and clearly show fewer lesional cells with positive immunoreactivity (original magnification: A-D, x100).
View Figure 2
This heterogeneous inflammatory background may play a role in the discordant immunohistochemical and molecular findings. Further, Fisher and colleagues acknowledge that non-specific staining may sometimes be attributed to macrophages and histiocytes, thus underscoring the need for strict IHC interpretation criteria and a better understanding of the specificity of this monoclonal antibody [16]. While our criteria were similar to those used by previous authors, in that any non-ambiguous cytoplasmic staining of viable tumor cells was considered positive, consensus on a more rigid set of criteria for BRAF V600E IHC scoring is certain to be of benefit [11,12]. However, even stringent guidelines may not entirely prevent false positive IHC results with this antibody, just as Fisher et al and Ehsani and colleagues reported in cases of melanoma and thyroid malignancies [17,18]. Additionally, Hoang and colleagues found that the commercially available VE1 clone, as used in our study, stained normal anterior pituitary and adrenal cortical tissues even in the absence of detectable BRAF V600E mutations [19]. Similar studies citing a lack of specificity continue to emerge [20-22]. Despite the apparent antibody limitations, it remains to be seen if BRAF IHC may be diagnostically useful in small biopsy specimens, or for aiding targeted selection of lesional tissue for molecular analysis. In fact, in malignant melanoma, sequential use of immunohistochemistry to isolate mutated cells followed by pyrosequencing for definitive analysis has been advocated, and similar approaches may prove to be advantageous in proper diagnosis and management in LCH.
Finally, our mutational analysis only included interrogation for the BRAF V600E mutation, as much emphasis has been placed on the role of the BRAF V600E mutation in driving LCH. However, Kansal and colleagues reported the presence of BRAF V600D mutation in exon 15 of the BRAF oncogene in a patient with LCH [23]. It is possible that a subset of the BRAF wild-type samples in our series harbored mutations such as V600D, V600K or V600R. Whether or not these other mutations play a role in cutaneous LCH has yet to be fully explored, but there is also recent evidence arguing against the putative role of V600E in LCH. Tong et al reported a series of 18 adult patients in which no mutations were detected, even in patients with multisystem disease, regardless of the anatomic site of origin for tested specimens [24].
Conclusions
We found a slightly higher rate of BRAF V600E protein expression in pediatric cases of LCH compared to adult cases (80% vs. 56%). Additionally, central nervous system involvement correlated with a lack of staining, though the explanation for this negative correlation is not readily apparent. We also observed an association between bone involvement and positive immunoreactivity in the pediatric population, but this did not reach statistical significance. Although a statistically significant difference (p < 0.05) was seen between the control cases, all of which were negative by IHC, and the study cases, 75% of which were positive by IHC, suggesting excellent specificity and reasonable sensitivity of BRAF V600E IHC, our series casts doubt on both the clinical utility and specificity of this immunostain in cutaneous LCH in light of the considerable discordance between IHC and mutational analysis. Thus, further study of this antibody in cases of cutaneous LCH is imperative, not only to better elucidate its diagnostic performance and role as a surrogate marker, but also certainly before targeted therapy and treatment algorithms can be based on immunohistochemical profiles.
Conflict of Interest
The corresponding author is the guarantor of submission. All authors consent to the publication of this manuscript and have contributed substantially to writing and revising this manuscript. All authors attest to the accuracy of the information presented. The authors have no other conflicts of interest.
References
-
Guyot-Goubin A, Donadieu J, Barkaoui M, Bellec S, Thomas C, et al. (2008) Descriptive epidemiology of childhood Langerhans cell histiocytosis in France, 2000-2004. Pediatr Blood Cancer 51: 71-75.
-
Ness MJ, Lowe GC, Davis DM, Hand JL (2014) Narrowband ultraviolet B light in Langerhans cell histiocytosis: a case report. Pediatr Dermatol 31: e10-12.
-
Windebank K, Nanduri V (2009) Langerhans cell histiocytosis. Arch Dis Child 94: 904-908.
-
Badalian-Very G, Vergilio JA, Fleming M, Rollins BJ (2013) Pathogenesis of Langerhans cell histiocytosis. Annu Rev Pathol 8: 1-20.
-
Lichtenstein L (1953) Histiocytosis X: integration of eosinophilic granuloma of bone, Letterer-Siwe disease, and Schuller-Christian disease as related manifestations of a single nosologic entity. Am Med Assoc Arch Pathol 56: 84-102.
-
Badalian-Very G, Vergilio JA, Degar BA, MacConaill LE, Brandner B, et al. (2010) Recurrent BRAF mutations in Langerhans cell histiocytosis. Blood 116: 1919-1923.
-
Haroche J, Charlotte F, Arnaud L, von Deiming A, Helias-Rodzewicz Z, et al. (2012) High prevalence of BRAF V600E mutations in Erdheim-Chester disease but not in other non-Langerhans cell histiocytoses. Blood 120: 2700-2703.
-
Sahm F, Capper D, Preusser M, Meyer J, Stenzinger A, et al. (2012) BRAFV600E mutant protein is expressed in cells of variable maturation in Langerhans cell histiocytosis. Blood 120: e28-34.
-
Satoh T, Smith A, Sarde A, Lu H, Mian S, et al. (2012) B-RAF mutant alleles associated with Langerhans cell histiocytosis, a granulomatous pediatric disease. PLoS ONE 7: e33891.
-
Long GV, Wilmott JS, Capper D, Preusser M, Zhang YE, et al. (2013) Immunohistochemistry is highly sensitive and specific for the detection of V600E BRAF mutation in melanoma. Am J Surg Pathol 37: 61-65.
-
Colomba E, Helias-Rodzewicz Z, Von Deimling A, Marin C, Terrones N, et al. (2013) Detection of BRAF p.V600E mutations in melanomas: comparison of four methods argues for sequential use of immunohistochemistry and pyrosequencing. J Mol Diagn 15: 94-100.
-
Capper D, Berghoff AS, Magerle M, Ilhan A, Wohrer A, et al. (2012) Immunohistochemical testing of BRAF V600E status in, 120 tumor tissue samples of patients with brain metastases. Acta Neuropathol 123: 223-233.
-
Mackelvie AA, Park WW (1950) Letterer-Siwe Disease. Arch Dis Child 25: 93-98.
-
Roden AC, Hu X, Kip S, Parrilla Castellar ER, et al. (2014) BRAF V600E expression in Langerhans cell histiocytosis: clinical and immunohistochemical study on 25 pulmonary and 54 extrapulmonary cases. Am J Surg Pathol 38: 548-551.
-
Méhes G, Irsai G, Bedekovics J, Beke L, Fazakas F, et al. (2014) Activating BRAF V600E mutation in aggressive pediatric Langerhans cell histiocytosis: demonstration by allele-specific PCR/direct sequencing and immunohistochemistry. Am J Surg Pathol 38: 1644-1648.
-
Fisher KE, Cohen C, Siddiqui MT, Palma JF, Lipford EH 3rd, et al. (2014) Accurate detection of BRAF p.V600E mutations in challenging melanoma specimens requires stringent immunohistochemistry scoring criteria or sensitive molecular assays. Hum Pathol 45: 2281-2293.
-
Ehsani L, Cohen C, Fisher KE, Siddiqui MT (2014) BRAF mutations in metastatic malignant melanoma: comparison of molecular analysis and immunohistochemical expression. Appl Immunohistochem Mol Morphol 22: 648-651.
-
Fisher KE, Neill SG, Ehsani L, Caltharp SA, Siddiqui MT, et al. (2014) Immunohistochemical investigation of BRAF p.V600E mutations in thyroid carcinoma using 2 separate BRAF antibodies. Appl Immunohistochem Mol Morphol 22: 562-567.
-
Mordes DA, Lynch K, Campbell S, Diaz-Santagata D, Nose V, et al. (2014) VE1 antibody immunoreactivity in normal anterior pituitary and adrenal cortex without detectable BRAF V600E mutations. Am J Clin Pathol 141: 811-815.
-
Farzin M, Toon CW, Clarkson A, Siosan L, Gill AJ (2014) BRAF V600E mutation specific immunohistochemistry with clone VE1 is not reliable in pituitary adenomas. Pathology 46: 79-80.
-
Sperveslage J, Gierke M, Capper D, Honegger J, Sipos B, et al. (2013) VE1 immunohistochemistry in pituitary adenomas is not associated with BRAF V600E mutation. Acta Neuropathol 125: 911-912.
-
Adackapara CA, Sholl LM, Barletta JA, Hornick JL (2013) Immunohistochemistry using the BRAF V600E mutation-specific monoclonal antibody VE1 is not a useful surrogate for genotyping in colorectal adenocarcinoma. Histopathology 63: 187-193.
-
Kansal R, Quintanilla-Martinez L, Datta V, Lopategui J, Garshfield G, et al. (2013) Identification of the V600D mutation in Exon 15 of the BRAF oncogene in congenital, benign langerhans cell histiocytosis. Genes Chromosomes Cancer 52: 99-106.
-
Tong C, Jia X, Jia Y, He Y (2014) Langerhans cell histiocytosis in Chinese adults: absence of BRAF mutations and increased FOXP3(+) regulatory T cells. Int J Clin Exp Pathol 7: 3166-3173.
-
Varga E, Korom I, Polyánka H, Szabó K, Széll M, et al. (2015) BRAFV600E mutation in cutaneous lesions of patients with adult Langerhans cell histiocytosis. J Eur Acad Dermatol Venereol 29: 1205-1211.





