International Journal of Pathology and Clinical Research
Opana-Induced Renal and Hepatic Injury: A Case Report and Review
Yahya M Kurdi1*, Sergey Brodsky2 and Martha Yearsley2
1Department of Internal Medicine, Akron General Medical Center, Affiliate of Cleveland Clinic Northeast Ohio Medical University, Rootstown, Ohio, USA
2Department of Pathology, The Ohio State University Wexner Medical Center, Columbus, OH, USA
*Corresponding author:
Yahya M Kurdi, MD, MS, Akron General Avenue, Department of Internal Medicine, Akron General Medical Center, Akron, OH, USA, Fax: 330-535-9270, Tel: 508-395-9754, E-mail: yahya.kurdi@akrongeneral.org
Int J Pathol Clin Res, IJPCR-2-034, (Volume 2, Issue 2), Case Report and Literature Review; ISSN: 2469-5807
Received: February 14, 2016 | Accepted: April 28, 2016 | Published: April 30, 2016
Citation: Kurdi YM, Brodsky S, Yearsley M (2016) Opana-Induced Renal and Hepatic Injury: A Case Report and Review. Int J Pathol Clin Res 2:034. 10.23937/2469-5807/1510034
Copyright: © 2016 Kurdi YM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A 34-year-old Caucasian female presented with headache, fatigue, mild abdominal pain, and dehydration. She admitted to intravenous abuse of "melted" Opana ER tablets (extended-release oral formulation of oxymorphone hydrochloride). Noted clinical signs included tachycardia, tachypnea, and trace peripheral edema. Abnormal laboratory tests included low creatinine clearance, hemoglobin, hematocrit, and platelet. Histological studies of hepatorenal biopsies showed evidence of severe active thrombotic microangiopathy (TMA) involving the arterioles and small arteries of the kidney and scattered portal and lobular granulomata and hepatic vascular changes involving the hepatic artery branches in some portal tract. To our knowledge of the published English literature, this is probably the first case report of Opana induced thrombotic microangiopathy (TMA) with evidence of histological involvement to both the kidney and the liver.
Keywords
Thrombotic microangiopathy, Oxymorphone, Opana, Liver, Kidney
Introduction
Long-acting opioids formulations have been used for treatment of moderate to severe chronic pain. Opana ER, a selective μ-opioid agonist made by Endo Pharmaceuticals, is an oral extended-release formulation of oxymorphone hydrochloride originally approved by US Food and Drug Administration (FDA) in 2006 for the treatment of chronic pain [1]. The first cases of thrombotic thrombocytopenic purpura (TTP) - mimicking illness associated with intravenous abuse of Opana ER were reported in 2012 in Tennessee, U.S.A. [2]. Despite warnings by FDA and CDC and release of a crush-resistant formulation of Opana by the drug maker, patients were still able to melt the tablets for intravenous abuse [3-8]. TTP is a well-established cause of thrombotic microangiopathy (TMA) [9]. We report the first case of Opana induced thrombotic microangiopathy (TMA) involving both the kidney and the liver.
Case Report
A 34-year-old Caucasian female presented to the hospital emergency with headache, fatigue, mild abdominal pain, and dehydration over the past 4 days. Past medical history was notable for intravenous (IV) drug abuse and hepatitis. She admitted to active intravenous abuse of Opana ER, an extended release oral formulation of oxymorphone hydrochloride, after first melting the tablet. She endorsed a family history of HTN. Patient is a social alcohol drinker and a one pack per day tobacco smoker. Review of systems was pertinent for malaise and thirst. Patient denies chest pain, palpitations, SOB, coughing, nausea, vomiting, diarrhea, burning sensation with urination, weakness, numbness, or incontinence. She weighs 140 lb and is 67 inches tall. She was afebrile with a pulse of 110 bpm, BP of 193/133 mmHg, and respiratory rate of 22 breaths per minute. The BP later improved to 155/108 mmHg after medical interventions. Physical exam pertinent findings included dry lips and mucous membranes with trace peripheral edema. No heart murmurs, hepatosplenomegaly, or jaundice were noted. Laboratory tests showed serum creatinine of 3.77 (0.56-1.0 mg/dl), blood urea nitrogen of 44 (7-22 mg/dl), and creatinine clearance of 14 (75-125 ml/min). Serum sodium, potassium, phosphorus, and calcium were within normal limits. In addition to normal white blood cell count, complete blood count test revealed hemoglobin, hematocrit, and a platelet count of 9.2 (12.0-15.5 g/dl), 27.8 (34.9 to 44.5%), and 112,000 (150,000-400,000 platelets per microliter) respectively. Erythrocyte sedimentation rate was 15. Hepatic panel was normal except for mild increase in Alanine and Aspartate transaminases. Serological tests were positive for Hep B core Ag IgM and HCV IgG. Other serological studies including ANA, Anti-DNA, anti-GBM, ANCA, RF, Coombs Test, and C Profile were negative. Urinalysis showed mild hematuria (0-2 RBCs/HPF), pH of 6, specific gravity of 1.010, Albumin of 30 mg/dl, and 24-hour urine protein of 715 mg in a 24-hour urine volume of 1750 ml. Blood and urine cultures showed no growth. Echocardiogram revealed possible valvular vegetations. Liver and kidney biopsies were performed. Kidney pathological findings were consistent with severe active thrombotic microangiopathy (TMA) with moderate chronicity involving the arterioles and small arteries with fragmented red blood cells in the vascular wall and focal fibrinoid changes (Figure 1, panel A). Direct immunofluorescence and electron microscopy revealed no evidence of immune complex deposition. Liver biopsy showed hepatic vascular changes involving the hepatic artery branches in some portal tracts, similar to those described in the kidney biopsy, and scattered portal and lobular granulomata (Figure 1, panel B).
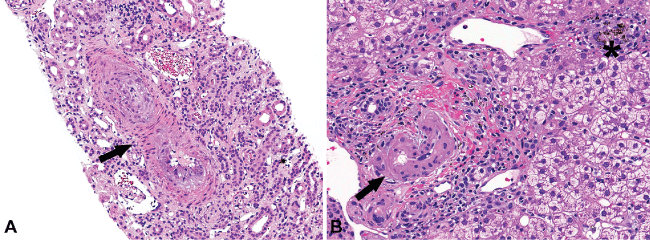
.
Figure 1: Thrombotic microangiopathy vascular (arrow) changes in the kidney (A) and in the liver (B). In addition, a portal based granuloma containing pigment (asterisk) is present in a portal tract (Hematoxylin and Eosin stain, 200X). Of note, renal biopsy specimen contained 69 glomeruli in the tissue submitted for light microscopy, 23 glomeruli in the frozen tissue submitted for immunofluorescence, and 4 glomeruli in tissue submitted for electron microscopy. Of those, up to 4 glomeruli were globally sclerotic; the glomeruli with open capillary loops were slightly enlarged and appeared ischemic with wrinkled glomerular basement membrane. There were no glomerular proliferative lesions or cellular crescents in the biopsy specimen.
View Figure 1
Discussion
This is the first case report of TMA secondary to Opana ER IV abuse demonstrating involvement of the kidney and liver by TMA that was confirmed histologically. The term TMA was first described by Symmers [10]. TMA is characterized by thrombocytopenia, microangiopathic hemolytic anemia, occlusive microvascular thrombosis, and variable severity of end-organ damage [9,11]. All features of TMA were present in our patient.
TTP and hemolytic uremic syndrome (HUS) are well-known primary causes of TMA. To a lesser extent, secondary TMA has been associated with infection, chemotherapy, disseminated malignancy, radiation, and toxins [12]. Drug-induced TMA is rare but associated with higher morbidity and mortality. Examples of such drugs include Mitomycin, Tacrolimus, Interferon, and Clopidogrel [9]. The first cases of TTP-like illnesses caused by intravenous abuse of Opana ER were reported by CDC in Tennessee in 2012 after state-wide investigation triggered by a nephrologist report to The Tennessee Department of Health of three cases of unexplained TTP [2].
While more TTP-like cases secondary to IV administration of melted Opana ER tablets have been reported, histological studies on kidney biopsies from such cases have been very limited with no histological studies done on liver. Histological changes described in renal biopsies included interlobular arterial intimal edema and partial occlusion, loss of mesangial architecture, and ischemic changes in associated glomeruli [7,13-16]. This case of Opana IV abuse uniquely show evidence of histological findings of TMA in both the kidney and liver as shown in figure 1.
The patient in this Case Report had an acute infection secondary to chronic IV administration of melted Opana tablets and a history of hepatitis. Sepsis is usually associated with acute glomerulonephritis, which was ruled out. The histology of kidney biopsy showed evidence of TMA with moderate chronicity; finding more likely associated with chronic IV abuse of melted Opana ER tablets. Hepatitis is associated with general inflammatory changes. Although the scattered granulomata are non-specific and likely associated to the patient’s history of IV drug abuse, there were vascular changes similar to those seen in the kidney biopsy, thus supporting TMA involvement of the liver.
After the report of two case fatalities due to Opana ER abuse in 2009 [13], more focus was placed on how to improve the screening test for Opana ER as the widely available opiate immunoassays were poorly sensitive to this compound compared with the gas chromatography-mass spectrometry GC-MS. Lefebvre et al. found out that combination of DRI oxy assay and CEDIA Opi test was a good screening test to detect opiate, including oxymorphone, in urine [17].
While therapeutic plasma exchange has been the main stay of therapy for Opana induced TMA, Miller et al. demonstrated in a case series that such treatment is unnecessary and supportive care and treatment of the underlying condition is key [16]. This may suggest that the effect of IV abuse of melted Opana ER on kidney and other organs is likely chemical or formulatory and less likely antibody-mediated. Opana ER contains a hydrophilic matrix made of xanthan and locust bean gum. Oxymorphone is more lipid soluble than morphine due to a ketone-group substitution and more structurally related to hydromorphone. Its half-life via oral administration was prolonged in renal failure, while no information was established on its clearance and extraction from the liver [18]. The exact mechanism of how the IV abuse of melted Opana ER tablets, a sustained-release oral formulation, causes TMA is yet to be deciphered. Our patient did not receive the plasma exchange but did not finish the recommended course of treatment as she elected to leave the hospital prematurely.
Clinicians need to have high suspicion for Opana and similar IV drug abuse in a patient presenting with TTP-like illness. Diagnosing Opana-induced TMA is crucial since it is associated with increased mortality and hepatorenal pathology.
References
-
Craig DS (2010) Oxymorphone Extended-Release Tablets (Opana ER) For the Management of Chronic Pain: A Practical Review for Pharmacists. P T 35: 324-357.
-
Centers for Disease Control and Prevention (CDC) (2013) Thrombotic thrombocytopenic purpura (TTP)-like illness associated with intravenous Opana ER abuse--Tennessee, 2012. MMWR Morb Mortal Wkly Rep 62: 1-4.
-
Endo Pharmaceuticals announces reformulated version of Opana ER with INTAC technology designed to be crush-resistant accounts for more than 90 percent of Opana ER total prescription volume. PR Newswire. September 6, 2012.
-
US Food and Drug Administration (FDA) (2012) FDA warns about serious blood disorder resulting from misuse of Opana ER.
-
Centers for Disease Control and Prevention (CDC) (2013) Cluster of cases of thrombotic thrombocytopenic purpura (TTP) associated with intravenous nonmedical use of Opana ER. Health Alert Network (HAN). October 26, 2012.
-
Bartholomäus J, Ashworth J, Strothmann K (2012) Innovative formulation technology protecting intended drug action. Drug Development & Delivery 12: 8.
-
Ambruzs JM, Serrell PB, Rahim N, Larsen CP (2014) Thrombotic microangiopathy and acute kidney injury associated with intravenous abuse of an oral extended-release formulation of oxymorphone hydrochloride: kidney biopsy findings and report of 3 cases. Am J Kidney Dis 63: 1022-1026.
-
US Food and Drug Administration (FDA) (2013) FDA statement: original Opana ER relisting determination. May 10, 2013.
-
Pisoni R, Ruggenenti P, Remuzzi G (2001) Drug-induced thrombotic microangiopathy: incidence, prevention and management. Drug Saf 24: 491-501.
-
Izzedine H, Perazella MA (2015) Thrombotic microangiopathy, cancer, and cancer drugs. Am J Kidney Dis 66: 857-868.
-
Symmers WS (1952) Thrombotic microangiopathic haemolytic anaemia (thrombotic microangiopathy). Br Med J 2: 897-903.
-
Kwaan HC (1987) Miscellaneous secondary thrombotic microangiopathy. Semin Hematol 24: 141-147.
-
McIntyre IM, Sherrard JL, Nelson CL (2009) Case report: Oxymorphone-involved fatalities: a report of two cases. J Anal Toxicol 33: 615-619.
-
Amjad AI, Parikh RA (2013) Opana-ER used the wrong way: intravenous abuse leading to microangiopathic hemolysis and a TTP-like syndrome. Blood 122: 3403.
-
Kapila A, Chhabra L, Chaubey VK, Summers J (2014) Opana ER abuse and thrombotic thrombocytopenic purpura (TTP)-like illness: a rising risk factor in illicit drug users. BMJ Case Rep 2014.
-
Miller PJ, Farland AM, Knovich MA, Batt KM, Owen J (2014) Successful treatment of intravenously abused oral Opana ER-induced thrombotic microangiopathy without plasma exchange. Am J Hematol 89: 695-697.
-
Gingras M, Laberge MH, Lefebvre M (2010) Evaluation of the usefulness of an oxycodone immunoassay in combination with a traditional opiate immunoassay for the screening of opiates in urine. J Anal Toxicol 34: 78-83.
-
Prommer E (2006) Oxymorphone: a review. Support Care Cancer 14: 109-115.





