International Journal of Pathology and Clinical Research
Anaplastic Large Cell Lymphoma as a Potential Diagnostic Challenge and Pitfall in Aspiration Biopsies of Thoracic Tumours
Justin Bateman, Celeste Morley, Bruce F. Burns and Shahid Islam*
Department of Pathology and Laboratory Medicine, the Ottawa Hospital and University of Ottawa, Ottawa, ON, Canada
*Corresponding author:
Shahid Islam, MD, PhD, FRCPC, FCAP, The Ottawa Hospital, General Campus Site, Critical Care Wing, Rm # 4115 , 501 Smyth Road, K1H 8L6, Ottawa, ON, Canada, Tel: 613-737-8899, Ext 78297, 613-739-6881, E-mail: sislam@toh.on.ca
Int J Pathol Clin Res, IJPCR-2-033, (Volume 2, Issue 2), Case Report and Literature Review; ISSN: 2469-5807
Received: February 18, 2016 | Accepted: March 28, 2016 | Published: April 01, 2016
Citation: Bateman J, Morley C, Burns BF, Islam S (2016) Anaplastic Large Cell Lymphoma as a Potential Diagnostic Challenge and Pitfall in Aspiration Biopsies of Thoracic Tumours. Int J Pathol Clin Res 2:033. 10.23937/2469-5807/1510033
Copyright: © 2016 Bateman J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Anaplastic large cell lymphoma (ALCL) is a peripheral T-cell Lymphoma, which can be broadly divided into ALK positive or negative disease. Accounting for only a small proportion of Non-Hodgkin Lymphomas, ALCL can present a significant diagnostic challenge for pathologists. The disease shows a slight male predominance and bimodal distribution, affecting both young and older adults. ALCL often presents with lymphadenopathy, systemic B-symptoms, and extra-nodal disease. With current treatment, prognosis is relatively favourable for ALK-positive disease.
Case report: The patient is a 54 year-old male who presented in January 2015 with increasing dyspnea and weight loss over several months, eventually requiring intubation and admission to ICU for respiratory failure. He had a remote history of testicular seminoma and 'melanoma'. A trans-bronchial Wang needle aspirate biopsy of the right sub-carinal lymph node was preformed and showed sheets of large malignant cells with pleomorphic nuclei, multiple prominent nucleoli, and abundant cytoplasm. The tumour cells are positive for CD45, CD3, CD30 and ALK-1.
Discussion: ALCL can be seen in needle aspiration biopsies, and one must consider this disease when poorly differentiated malignant cells are present. Cytomorphology on a FNAB can aid in the diagnosis of ALCL, however, can be very challenging. There are often overlapping features that contribute to diagnostic pitfalls, particularly when the patient presents with multiple malignancies. Immunohistochemistry is essential to ensure an accurate diagnosis. Patients with ALCL of the thorax typically present with rapidly progressive respiratory failure, and clinicians must maintain a high index of suspicion for this entity.
Keywords
Anaplastic, T-Cell, Lymphoma, Cytology, Aspiration, Thoracic
Background
Anaplastic large cell lymphoma (ALCL) is a peripheral T-cell Lymphoma, which can be broadly divided into two categories: Anaplastic Lymphoma Kinase (ALK) positive or negative disease [1]. ALCL is more common in the pediatric age group, where it accounts for up to 20% of lymphomas in developed nations [2]. In adults ALCL accounts for only 2% of non-hodgkin lymphomas, and can present a significant diagnostic challenge for pathologists given its rarity and poorly differentiated morphology [1].
In adults this disease shows a male predominance and bimodal age distribution, affecting both younger (2-3rd decades of life) and older adults (6-7th decades of life) [1,3]. ALK status also seems to be affected by this bimodal distribution, with older patients more likely harbouring ALK-negative disease [1,3]. ALCL often presents with lymphadenopathy and systemic B-symptoms, and unfortunately typically presents at advanced stages [1,2]. Extra-nodal site involvement is common including skin, bone, airway, solid organs, and soft tissue [1-3]. With current treatment the prognosis is relatively favourable for ALK-positive disease, and 5-year survival can be as high as 80% [1]. In contrast, treated ALK-negative ALCL has a 5-year survival rate quoted at 48% [1]. ALCL has also been associated with immunosuppression and silicone breast implants in women [3,4].
Here we present a case of thoracic ALK-positive ALCL diagnosed on Wang needle aspiration biopsy in a patient presenting with rapidly progressive respiratory failure. With this case report and literature review, we hope to: 1) Review the key cytomorphological features seen in ALCL. 2) Emphasize the wide differential diagnosis that must be considered, and discuss the role of morphology and immunohistochemistry (IHC) in making this diagnosis on aspiration biopsy. 3) Highlight this particular clinical presentation, and reinforce the need for considering ALCL when patients present in this manner.
Case Report
The patient was a 54 year-old male who presented to the emergency department in January 2015 with shortness of breath that had been worsening over the last three months. A CT scan of his thorax showed numerous small pulmonary nodules in the right lung, a right upper lobe consolidation, and multiple enlarged thoracic lymph nodes causing obstruction of the right middle and lower bronchi. Over the next four days his oxygen requirements continued to increase (up to 100% FiO2), and he subsequently required intubation, bronchial stenting, and ICU admission. His past medical history was significant for a classic seminoma, treated with radical orchiectomy and radiation, and a self-reported history of 'melanoma', though a pathology report could not be found at time of biopsy interpretation. He was otherwise fairly healthy and had never smoked. He was not known to have HIV or to be otherwise immunosuppressed. The primary differential diagnosis being considered was metastatic melanoma or seminoma versus a primary lung carcinoma.
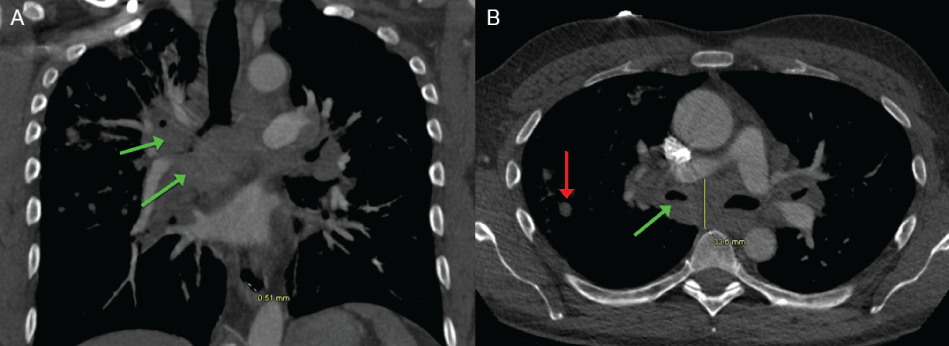
A repeat CT scan of the chest one week later showed significant interval growth of the pulmonary nodules and obstructive lymphadenopathy (Figure 1). While in ICU, a trans-bronchial Wang needle aspirate biopsy of an enlarged sub-carinal lymph node was performed.
.
Figure 1: A) CT Thorax with contrast, coronal view, showing obstruction of the right bronchi by the peri-hilar soft tissue mass (green arrows); B) CT Thorax with contrast, axial view, showing significant right peri-hilar obstructive adenopathy (green arrow) along with small right lung nodules (red arrow).
View Figure 1
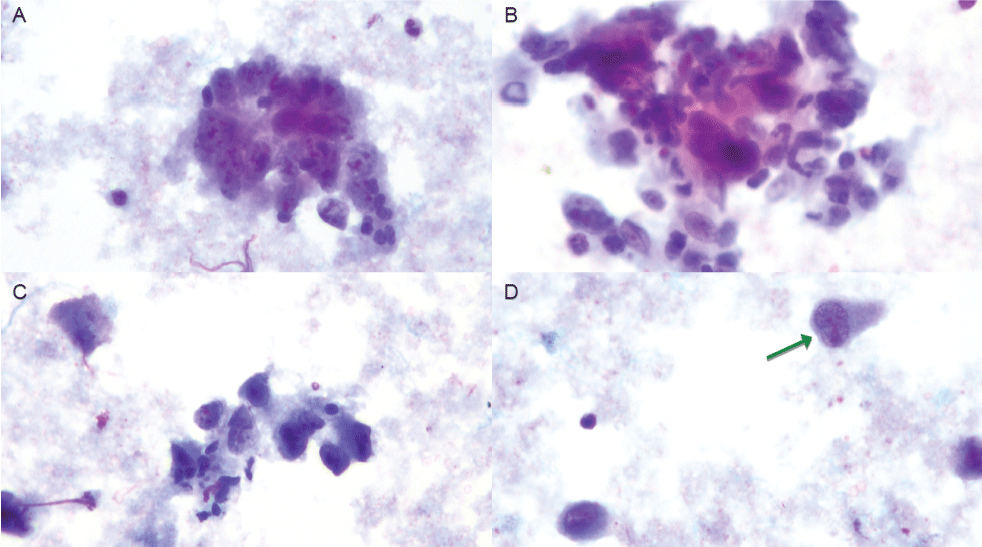
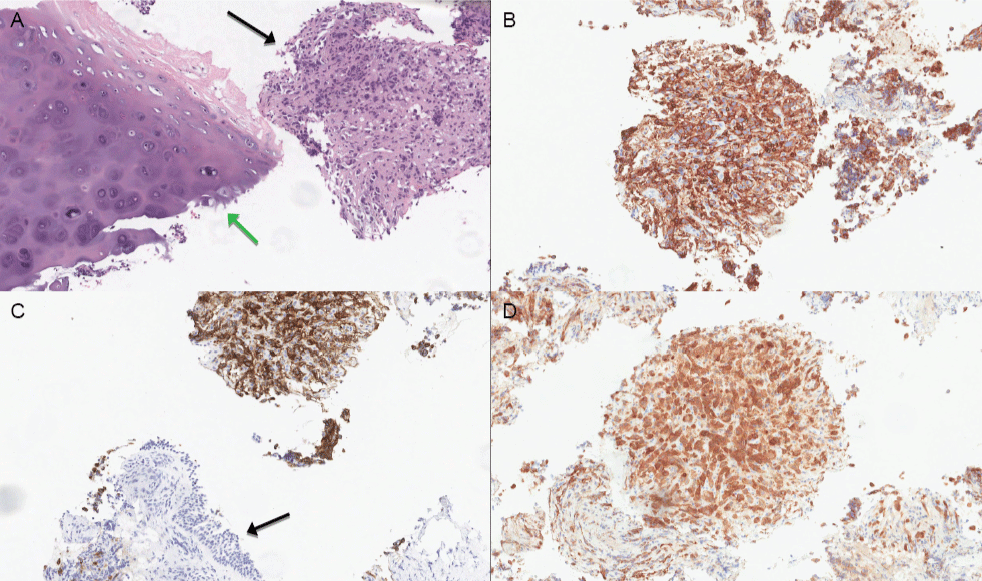
The aspirate showed malignant cells seen singly and in groups (Table 1 and Figure 2). The cells had abundant amphophilic cytoplasm, large hyper-chromatic and pleomorphic nuclei, and multiple prominent/bizarre nucleoli. Cell-block IHC was positive for CD45, CD3, CD30, and ALK protein (Figure 3). Cells were negative for CK7, CK20, AE1/3, TTF-1, S-100, HMB 45, Pax5, Pax8, CD15, and CD20.
![]()
Table 1: Comparing the cytological features seen in our case report versus those reported in the literature.
View Table 1
.
Figure 2: A-D) Aspiration biopsy showing malignant cells singly and in groups, with basophilic cytoplasm, poorly defined cell borders, large hyper-chromatic and pleomorphic nuclei, multi-nucleation, and multiple bizarre shaped nucleoli. (Papanicolaou stain, 40x magnification). Green arrow demonstrates a hand-mirror cell morphology.
View Figure 2
The patient was subsequently treated with cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP). His admission was further complicated by pulmonary embolism and pneumothorax requiring chest tube placement. He continued to deteriorate and died in early March of 2015.
.
Figure 3: A) Cell-block, H&E stain showing poorly differentiated malignant cells (black arrow) next to bronchial cartilage (green arrow); B) Cell-block, CD45 IHC demonstrating strong cytoplasmic staining; C) Cell-block, CD30 IHC showing strong cytoplasmic reactivity. Note the adjacent non-staining respiratory epithelium (black arrow); D) Cell-block, ALK IHC is strongly reactive in this case (A-D, 10x magnification).
View Figure 3
Discussion
A number of morphologic patterns of ALCL have been described on histology including: common-type (70% of cases), small-cell, sarcomatoid, lymphohistiocytic, giant-cell rich, and Hodgkin-like [1,5]. The cytomorphological features of ALCL have been beautifully described in prior series' performed by Das et al., Ng et al., and Rapkiewicz et al. [6-8]. Features common to all ALCL variants on cytology include discohesiveness, significant nuclear pleomorphism, mono- and multinucleated giant cells, prominent nucleoli, amphophilic-basophilic cytoplasm, and a lack of lymphoglandular bodies [6-8]. The most frequently discussed cell morphology seen in ALCL is the so-called 'hallmark cell', with it's eccentrically placed horseshoe-shaped nucleus and prominent or rod-like nucleoli [6-8]. These cells are sometimes sub-categorized as type A, B, or C, depending on the nuclear appearance and degree of indentation [6]. Hallmark cells however, may only be present in ~50% of cases, and over-reliance on their detection in cytology specimens could lead to under-diagnosis [6,8]. Furthermore, distinguishing hallmark cells from Reed-Sternberg cells is crucial in avoiding misdiagnosis [7]. Other ALCL cell morphologies include multinucleated cells with wreath-like arrangement of nuclei, doughnut, dumbbell, and hand-mirror cells [6,7]. Small round cells and plasmacytoid cells are more commonly seen in the small-cell variant [7]. While the diagnosis of ALCL should not be diagnosed on cytomorphology alone, careful attention to these cytological features (especially the presence of hallmark and multinucleated giant cells with wreath-like arrangement of nuclei) has been shown to accurately diagnose ALCL on aspiration biopsy [8,9]. Interestingly, our case did not show any of these classic cell morphologies other than a hand-mirror cell.
The differential diagnosis is vast when dealing with aspirates of ALCL. One must consider other myeloid and lymphoid neoplasms including Hodgkin lymphoma (especially the lymphocyte-depleted variant), Non-Hodgkin lymphoma (i.e. diffuse large B-cell lymphoma and T-cell rich B-cell lymphoma), and myeloid sarcoma [6-11]. Because of its poorly differentiated morphology, other categories of neoplasm must also be considered including sarcoma, melanoma, germ-cell tumours (i.e. seminoma and embryonal carcinoma), sarcomatoid carcinomas, and other poorly differentiated carcinomas [6-11]. Nasopharyngeal carcinoma can mimic ALCL, especially in younger patients with the appropriate clinical context [2,6]. Small-cell variant ALCL can be confused with reactive lymphocytes, and 'plasmacytoid' cells have been mistaken for true plasma cell neoplasms [6,7]. Furthermore, the lymphohistiocytic variant of ALCL could potentially be mistaken for a true histiocytic neoplasm [7]. With the use of immunocytochemistry or IHC performed on cell-block, it is possible to definitively diagnose ALCL purely on the basis of aspiration biopsy [7]. ALCL typically expresses CD45 (a pan-lymphoid antigen), CD3 and CD5 (T-cell markers), and CD30 (with membranous and para-nuclear dot-like reactivity) [7]. While CD30 positivity is required for the diagnosis of ALCL, caution must be used with this marker, as it can be positive in other lymphomas and in embryonal carcinoma [7]. Negative staining for Pax5, CD15, and CD20 can be useful in ruling out Hodgkin and other B-cell lymphomas [1]. In the appropriate context, CD68 and CD138 could be used to rule-out a histiocytic or plasma cell neoplasm, respectively [7]. It is crucial in this setting to exclude melanoma by showing negative staining with markers such as S100 and HMB-45 [11]. Applying broad cytokeratin markers can help in excluding a poorly differentiated carcinoma (eg. AE1/3). It is worth noting that EMA can be positive in ALCL [8]. In our case, we also applied markers for Pax8 and TTF-1 to specifically rule-out carcinomas arising from the lung and thyroid. When the diagnosis of ALCL is made, ALK status can be tested by IHC, and equivocal cases can be subjected to FISH assays [1]. In our case, the history we received indicated that he had previously been diagnosed with melanoma, which could easily bias a reviewer towards that diagnosis [11]. Of note, when the patient's prior skin biopsy report was later recovered, it turned out to be squamous cell carcinoma. In our case, the cytological and cell-block IHC analysis was sufficient for diagnosis, and thus this patient never required a solid tissue biopsy.
Another interesting feature of this case was its presentation. Our patient presented with worsening shortness of breath, lobar consolidation, and bronchial obstruction leading to respiratory failure. In our report, the patient required intubation within four days of presentation to hospital. The wonderful case series by Xu, was able to find nine cases of ALCL presenting with bronchial (one case involved the trachea) obstruction and respiratory failure [3]. The median age was 16 years, with a range of 7-44 years of age. HIV status was positive in two cases [3]. Interestingly, the M:F ratio in this series was 1:2, which is in contrast to the overall male predominance of ALCL [3]. Of the nine patients, 75% harbored ALK-positive disease [3]. Disease-free survival of at least five months was reported in six cases [3]. We were able to find in the literature two other cases of ALCL with airway obstruction. In one case, a 54 year-old male presented with tracheal obstruction and respiratory failure, requiring stenting and radiation [12]. The other case could not be accessed through our institutional subscription [13].
With this case report and discussion, we hope to review the key cytomorphological features of ALCL on aspiration biopsy, and to discuss its vast differential diagnosis. We discussed how the use of morphology, combined with immunophenotyping, can definitively diagnosis ALCL on aspiration biopsy. Lastly we described how this case report and literature review emphasize the need for clinicians and pathologists alike to maintain a high index of suspicion for ALCL when patients present in this manner.
Ethical Statement
This case report and literature review complies with the ethical standards for research at The Ottawa Hospital and University of Ottawa.
References
-
Boi M, Zucca E, Inghirami G, Bertoni F (2015) Advances in understanding the pathogenesis of systemic anaplastic large cell lymphomas. Br J Haematol 168: 771-783.
-
Iyer VK (2013) Pediatric lymphoma diagnosis: Role of FNAC, biopsy, immunohistochemistry and molecular diagnostics. Indian J Pediatr 80: 756-763.
-
Xu X (2013) ALK-negative anaplastic large cell lymphoma primarily involving the bronchus: a case report and literature review. Int J Clin Exp Pathol 7: 460-463.
-
Brody GS, Deapen D, Taylor CR, Pinter-Brown L, House-Lightner SR, Andersen JS, Epstein AL (2015) Anaplastic large cell lymphoma occurring in women with breast implants: analysis of 173 cases. Plast Reconstr Surg 135: 695-705.
-
Pletneva MA, Smith LB (2014) Anaplastic large cell lymphoma: features presenting diagnostic challenges. Arch Pathol Lab Med 138: 1290-1294.
-
Das P, Iyer VK, Mathur SR, Ray R (2010) Anaplastic large cell lymphoma: a critical evaluation of cytomorphological features in seven cases. Cytopathology 21: 251-258.
-
Ng WK, Ip P, Choy C, Collins RJ (2003) Cytologic and immunocytochemical findings of anaplastic large cell lymphoma: analysis of ten fine-needle aspiration specimens over a 9-year period. Cancer 99: 33-43.
-
Rapkiewicz A, Wen H, Sen F, Das K (2007) Cytomorphologic examination of anaplastic large cell lymphoma by fine-needle aspiration cytology. Cancer 111: 499-507.
-
Kolonic SO, Prasek-Kudrna K, Roso V, Radic-Kristo D, Planinc-Peraica A, et al. (2010) Value of fine-needle aspiration cytology in diagnosis of Hodgkinâ lymphoma and anaplastic large cell lymphoma: One centre experience. Collegium Antropologicum 34: 75-79.
-
Vij M, Dhir B, Verma R, Agrawal V, Agarwal V, et al. (2011) Cytomorphology of ALK+ anaplastic large cell lymphoma displaying spindle cells mimicking a sarcomatous tumor: Report of a case. Diagnostic Cytopathology 39:775-779.
-
Pulitzer M, Brady MS, Blochin E, Amin B, Teruya-Feldstein J (2013) Anaplastic large cell lymphoma: a potential pitfall in the differential diagnosis of melanoma. Arch Pathol Lab Med 137: 280-283.
-
Damas C, Fernandes G, Magalhães A, Hespanhol VP (2006) Unusual diagnosis of tracheal obstruction. Rev Port Pneumol 12: 303-308.
-
Zuluaga P, Marti S, Navarro JT, Mejia E (2015) Endobronchial mass as the first manifestation of an anaplastic T-cell lymphoma. Med Clin (Barc) 144: 333-334.





