International Journal of Pathology and Clinical Research
Transforming the 24/7 Clinical Laboratories - Design Considerations, Workflow and Relocation to New Diagnostics Building
Wai Yoong Ng* and Chin Pin Yeo
Clinical Biochemistry Laboratories, Department of Pathology, Singapore General Hospital, Singapore
*Corresponding author:
Ng Wai Yoong, Department of Pathology, Clinical Biochemistry Laboratories, Academia, Diagnostics Tower, 20 College Road, Singapore 169856, Singapore, Tel: +6563213489; Fax: +65 62208678; E-mail: ng.wai.yoong@sgh.com.sg
Int J Pathol Clin Res, IJPCR-2-024, (Volume 2, Issue 1), Perspective; ISSN: 2469-5807
Received: December 11, 2015 | Accepted: February 22, 2016 | Published: February 26, 2016
Citation: Ng WY, Yeo CP (2016) Transforming the 24/7 Clinical Laboratories - Design Considerations, Workflow and Relocation to New Diagnostics Building. Int J Pathol Clin Res 2:024. 10.23937/2469-5807/1510024
Copyright: © 2016 Ng WY, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Laboratory services in Singapore General Hospital (SGH), a national referral centre, acute tertiary and teaching hospital has changed through the years with its beginning since 1821 and as a modern hospital in 1926. An opportunity to design new facilities to transform the delivery of clinical tests and embrace future challenges came up with the confirmation of a new purpose-built Pathology building in 2009.
The Clinical Biochemistry Laboratories and the central specimen reception (CSR) were to be on L8 floor of the new 13-storey building. Design of laboratory layout and workflow started with provisional space allocations and this process continued even as construction works approached L8. During the design journey, updates and decisions also incorporated directives from senior management.
An early decision was that the 24/7 Clinical Biochemistry laboratories be in close proximity to CSR. Two laboratory automation systems with high-speed on-line analysers were installed to support workload (5-7% annual increase) from SGH campus and outreach sites. The pneumatic tube system was enhanced to connect the rest of the hospital to L8 and a vertical transport system was built to connect L8 to the rest of the pathology laboratories at L9-L13.Centralized laboratory-grade water supplies were established for uninterrupted support to the chemistry analysers.
Post go-live on 22nd April 2013, turnaround times (TAT) target for stat tests remains fully met. TAT for routine outpatient and inpatient requests were significantly improved. These performance results fully affirm the design and layout’s intended promise towards a highly efficient journey from specimen collection to result reporting.
Keyword
Clinical pathology, Design layout, Lab relocation, Turnaround
Introduction
As medical services expand through the years, clinical laboratories are also seeing increasing test workload and service performance expectations. Turnaround times are now part of the laboratory’s key performance indicators. Round-the-clock operations are usually implemented and tests reaching to the millions. Annual increases in test workload meant that test equipment will get more complex to support these. Our pathology laboratory serves the parent hospital, Singapore General Hospital with its modern beginnings in 1926 (acute tertiary, national referral and teaching) and national specialty centres in an academic medicine campus.
Our Clinical Biochemistry and its sister laboratories of the Pathology department have been operating at 2 locations - a Pathology building originally built in the 1950s and at the main hospital complex. In 2010, our clinical biochemistry laboratories were getting 7.41 million tests that continued to 7.69 million in the following year. The latest 2014 figures showed 8.37 million tests. Today, we have integral features expected of the modern service laboratory - computer-driven processes, highly accurate analyte testing, wide test menu, quick result reporting - that the laboratory needs to maintain and expected to improve on. Planning for future needs can be considered an art more than science and the opportunity came to Clinical Biochemistry with the approval in 2009 for the construction and erection of a new purpose built building housing clinical diagnostics and research.
While there are publications on the considerations of implementing, choosing laboratory automation, pre-analytics, information technology and management of connectivity, little is available for the laboratory upgrade as in its physical movement across grounds and its planning stages to a new or refurbished laboratory building [1-7].
This report is on the design journey, workflow changes and turnaround time (TAT) performance following the full relocation to the new building. We share our experiences in this aspect of relocation of the entire clinical biochemistry laboratories within a weekend, with no downtime in laboratory services.
Journeys
Design planning
Experience in the adoption and running a laboratory automation system implemented in 2007, gave the planning team valuable insights for the new construction. The team needed infrastructure support to be given top priority, industry support for new automation technical improvements, and a re-look at the workflow and staffing needs. Expansion space to support emerging tests and equipment evaluation was considered. Services priorities for specialty tests (on mass spectrometry, high performance liquid chromatography platforms) were also not forgotten. However, as general chemistries and immunoassays for metabolic monitoring are the top tests in any clinical biochemistry workload, it was imperative that high consideration be given to expand and develop the laboratory automation systems that support them.
The key challenges faced by the modern laboratory are the need to cope with high workload while aiming to improve TAT and quality control procedures, and also maintain manpower status. Segregation of stat and routine specimens would allow some priority and also increasingly having to perform add-on tests on archived specimens. In addition, there should also be some capacity for future growth. Pathology laboratories were to be housed in L8 to L13 floors of the new building. The Clinical Biochemistry Laboratories and the central specimen reception (CSR) were to be on L8 floor. The design process journey looked into the evolution of workflow processes and new technology to be adopted with that in mind.
The consultancies looked into right siting of the pneumatic tube system (PTS) and lab automation lines, supporting ancillary process items and also with the placement of the central specimen reception and its support of specimen routing to the other laboratories. The CSR and client services merged into the full-fledged Client & Specimen Management section within the department as a result. As the anchor tenant of the floor, Clinical Biochemistry placed emphasis on the adjacencies of services. Regular conversations with the local vendors support teams were scheduled as the planning process gain momentum. The pneumatic tube system was enhanced to connect the rest of the hospital to L8 and a vertical transport system was built to connect L8 to L9-L13 laboratories for specimen routing. Centralized laboratory-grade water supplies were established for uninterrupted support to the chemistry analysers.
The final layout has in its center, a vertical transport system for specimens to the above floors, and a conveyor belt and requisition areas to serve this role. The center has also the improved PTS that embraces point-to-point (PTP) and hub services connecting the entire campus network. The Emergency department (A&E) and Specialist Outpatient Clinics(SOC) have direct PTP connection while colour-stripped specimen bags give a visual cue to prioritize handling of specimens from the Emergency department, intensive care units (ICUs) and Specialist Outpatient Clinic’s urgent specimens. Closest to the PTS is the specimen inlet load point of the new chemistry/immunoassay automation (Power Express, PE) line by Beckman Coulter. Direct computerized physician-order entry (CPOE) labels reading by the analysers augment the target of short turnaround times.
Technical clarifications were sorted out with Beckman Coulter headquarters through its local office. The local team was also very accommodating to our demands for various versions of architectural drawings to be made readily available for the fast-paced decision making process. Once the deal was inked with senior management approval, the entire footprint and layout of the PE line were mapped out together with the architects. As the final layout (Figure 1) took shape, concurrent plans were being made to plan for the relocation of the laboratory facilities from both facilities’ locations.
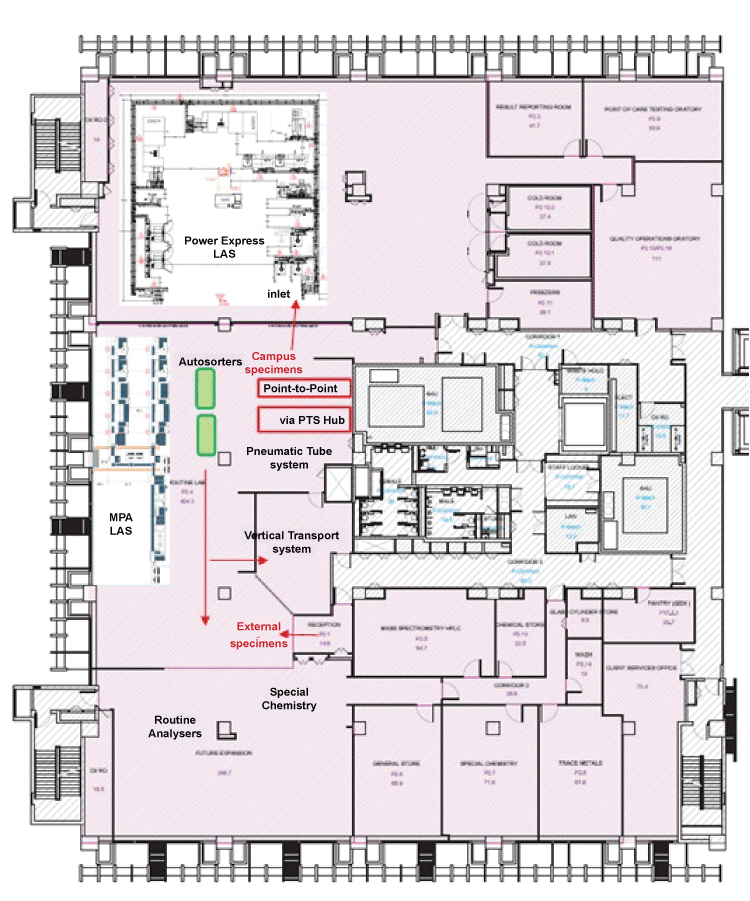
.
Figure 1: Specimen routing (arrows) from campus and external clients separated to allow priority handling for Stat/routine requests. High-throughput analysers and laboratory automation placed near to specimen reception.
View Figure 1
The relocation objectives remained the same i.e. for the 24/7 clinical laboratory services to have no downtime and to allow business/services continuity during relocation to the new building which by now was named Academia with its twin Diagnostics and Discovery tower blocks. This directive meant that we were to provide full testing capabilities upon achieving the Ministry of Health’s licence to practise and result reporting and at the point of go-live. In readiness to this requirement and that new analysers are going to be deployed, we started to establish and document the tests evaluations as early as in April 2012 with the delivery of the first autosorter. This autosorter was to be placed close to the PTS stations for handling workload during peak specimen delivery periods.
New technology and preparedness
To execute the relocation timeline, we also consulted with both the Pathology Relocation committee and building consultants on the delivery and installation timeline for our laboratory automation systems which by then comprised of the main Beckman Coulter PE LAS and the cobas 8000 system (two lines of c502 and e602 modules) from Roche Diagnostics (Modular Pre-Analytics Laboratory Automation System (MPA LAS)). Firm up on the timeline meant that the vendors put up project schedules for the delivery, installation, testing and setup of the equipment and to train staff on its operations. Staffs were also actively engaged in planning the move logistics and performing evaluations of new automated analyser platforms. We held lunch time talks, hands-on training on the new analysers and meetings in preparation for the move to the new premises. In tandem with new hardware and software improvements there was also a need to refresh our workflows to streamline work processes and improve on service quality standards like turnaround times.
The Beckman Coulter PE was delivered a week after the building’s Temporary Occupation Permit (TOP) was given on 30 January 2013. The additional new analysers came in first: an AU5822, an AU680, two UnicelDxI 800s. In close succession, the other items of the PE line arrived in a span of two weeks. Setting up equipment and operating in a building still classified as ‘Under Construction’ (as rectification works were being done) was a demanding process as it meant that all workers (including laboratory and vendor staff involved) were required to have hard hats and access to L8 floor was monitored and restricted due to the prevailing policies on security and insurance. Day-to-day activities were also monitored on-site by the presence of one of the Biochemistry team. This helped immensely to manage, engage and rectify critical issues arising on-the-spot during the installation period. After many hours, days and nights spent on setting up and testing the entire system by the installation crew, the installation was completed on 18 March as promised in the project schedule. It was then left to the applications crew to test the software applications that the system needs to function properly - the analysis, IT middleware, result reporting interface protocols. The crew also engaged the SGH IT team on the network infrastructure, performance and stability to have a proper communication. Testing include integrity of test messages transmissions, result reporting and analyser test performance specifications.
As the days drew closer to inspection by the Ministry of Health for consideration of licence approval, advanced teams of Biochemistry key users started their training and familiarization of the automation systems. Full documentation of test and systems checks and evaluation were readied to be inspected. Inspection day came on 8 April with start of the inspection process at 9.30 am. At the end of pouring over documents and records, questioned on infrastructure readiness and presentation of evidence of testing regimes with their evaluations, we received the delightful news that approval was granted in-principle to operate live on 22 April 2013 (Table 1)
![]()
Table 1: Timeline of events.
View Table 1
Our laboratory is the first site in the world to go-live with the Power Express Total Laboratory Automation System. Its unique features are;
a) 4 specimen transport lanes that allow separate high-priority management of stat specimens
b) RFID (radio frequency identification) of specimens that enhances specimen routing speed thereby allowing 3-4 times faster throughput than traditional barcode routing
c) 3 (up to 4) centrifuges connection to overcome bottle-neck seen in other current LAS
d) Auto-centrifugation and auto-decapping improving safety for laboratory staff by eliminating potential exposure to blood-borne pathogens
e) Connected high speed AU chemistry and DXI immunoassay analysers
f) 2 5000 tubes (5K)-stockyards allowing easy automatic retrieval of specimens for reruns or additional tests
g) Automatic aliquoting to split specimens for analysis on offline analysers.
Turnaround performance
Monitoring of stat TAT performance showed that following a dip in performance for 3 weeks post go-live, the target performances were fully met - this followed weekly meetings to appraise the situation with fine tuning of work processes. It is noteworthy that good performances are not static but dynamic with daily operational issues that can impact. Hence, stat test requests from the Emergency department and ICUs were promptly resolved (Figure 2). With the new central specimen reception area, work processes to give priority to test requests from the outpatient clinic setting also got a boost, making same-day laboratory tests cum outpatient consultations a positive option for our clinicians (Figure 3). Routine test requests are also now returning results faster, at under 45 min for 80%. These improvements were achieved through the priority workflow and positioning of PTP outlets serving both the Emergency department and SOC. An added prioritizing process for stat SOC specimens is that their blue-stripped specimen bags (different from the red-stripped bags for A&E and ICUs) offered quick visual cue (on arrival at the PTP outlet) for priority handling. Immediate accessioning of test order with a single scan of the CPOE label followed by placing to the close-by PE LAS inlet affords the quickest specimen routing as design-intended.
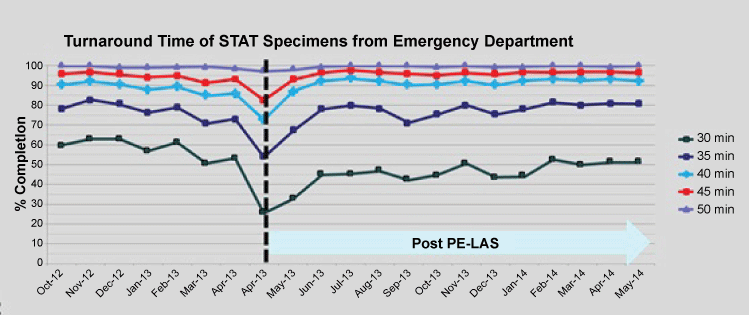
.
Figure 2: Turnaround performance for Emergency department’s STAT specimens. Vertical break-line depicts go-live week. Post go-live 1-year period illustrated.
View Figure 2
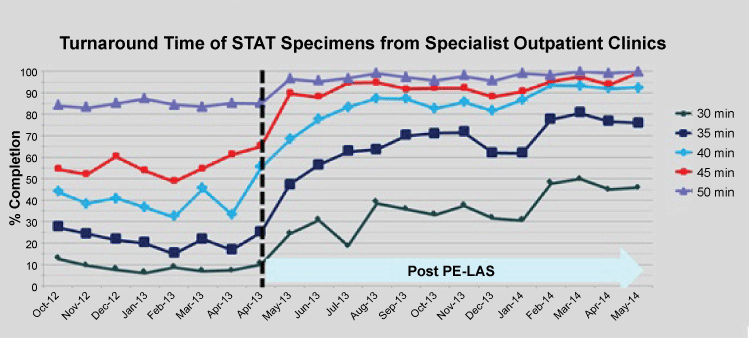
.
Figure 3: Turnaround performance for SOC STAT specimens. Vertical break-line depicts go-live week. Post go-live 1-year period illustrated.
View Figure 3
Lessons
Lab automation systems are now an integral feature of the modern clinical laboratory. Post go-live on 22nd April 2013, TAT target for stat tests remained fully met. TAT for routine outpatient and inpatient requests were significantly improved. These performance results fully affirm the design and layout’s intended promise towards a highly efficient journey from specimen collection to result reporting. Key learning points, tips and pitfalls are given in Table 2.
![]()
Table 2: Key lessons learnt - tips and pitfalls.
View Table 2
While large scale lab automation is perhaps key to any clinical biochemistry laboratory with the test volume, it has been debated whether lab automation can apply to other areas of clinical laboratory disciplines eg microbiology, molecular and histopathology. Clearly for microbiology there is still a debate on whether it is wise to implement although it is recognised that key benefits would be archiving of images for re-appraisal, tighter consistency of plating and growth conditions, and the system-takeover of multiple manual tasks currently [8-10].
In the entire journey of the laboratory relocation, there were identified 'champions' to key areas eg. equipment and test evaluation, building progress, logistics arrangements and certifications. Regular meetings on progress and co-operations between user, building consultants, site security and equipment suppliers were necessary for a timely scheduling. It is important that full support be given to the core laboratory team in the design, planning, executing and managing of the entire project.
Acknowledgement
We are grateful to our colleagues and the Division of Ambulatory and Clinical Support Services in making the relocation a no-stress event.
References
-
Burke MD (2000) Laboratory medicine in the 21st Century. Am J Clin Pathol 114: 841-846.
-
Middleton SR (2000) Developing an automation concept that is right for your laboratory. Clin Chem 46: 757-763.
-
Seaberg RS, Stallone RO, Statland BE (2000) The role of total laboratory automation in a consolidated laboratory network. Clin Chem 46: 751-756.
-
Melanson SE, Lindeman NI, Jarolim P (2007) Selecting automation for the clinical chemistry laboratory. Arch Pathol Lab Med 131: 1063-1069.
-
Da Rin G (2009) Pre-analytical workstations: a tool for reducing laboratory errors. Clin Chim Acta 404: 68-74.
-
Streiberg GS, Bwititi PT, Angel L, Sikaris KA (2009) Automation and expert systems in a core chemistry laboratory. JALA 14: 94-105.
-
Armbruster DA, Overcash DR, Reyes J (2014) Clinical chemistry laboratory automation in the 21st century - Amat Victoria curam (Victory loves careful preparation). Clin Biochem Rev 35: 143-153.
-
Greub G, Prod'hom G (2011) Automation in clinical bacteriology: what system to choose? Clin Microbiol Infect 17: 655-660.
-
Bourbeau PP and Ledeboer NA (2013) Automation in clinical microbiology. J Clin Microbiol 51: 1658-1665.
-
Mutters NT (2014) Laboratory automation in clinical microbiology: a quiet revolution. Austin J Infect Dis 1: 2-3.





