International Journal of Pathology and Clinical Research
Flow Cytometric Analysis of Cell Size in B-Cell Non-Hodgkin Lymphoma: Reliability and Potential Diagnostic Significance
Aaron Cotrell, Farzaneh Sayedian, Michelle Huang, Xia Chen, Ming Xie, Marc Smith and James Huang*
Department of Clinical Pathology, Oakland University William Beaumont School of Medicine, USA
*Corresponding author:
James Huang, MD, Department of Clinical Pathology, Beaumont Hospital, 3601 W. 13 Mile Road, Royal Oak, MI 48073-6769, USA, Tel: 248-551-0878, Fax: 248-551-0557-8148, E-mail: james.huang@beaumont.org
Int J Pathol Clin Res, IJPCR-1-005, (Volume 1, Issue 1), Research Article; ISSN: 2469-5807
Received: June 25, 2015 | Accepted: July 10, 2015 | Published: July 13, 2015
Citation: Cotrell A, Sayedian F, Huang M, Chen X, Xie M, et al. (2015) Flow Cytometric Analysis of Cell Size in B-Cell Non-Hodgkin Lymphoma: Reliability and Potential Diagnostic Significance. Int J Pathol Clin Res 1:005. 10.23937/2469-5807/1510005
Copyright: © 2015 Cotrell A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
To explore if lymphoma cell size can be reliably evaluated by flow cytometry, we examined average cell size of 144 cases of B-NHL using the mean forward scatter (FSC). We found that the mean cell size of internal reactive T-cells (RTCs) was essentially the same among diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), and small non-germinal center B-cell lymphoma (SNGCBL). In contrast, mean cell size of neoplastic B-cells (NBCs) of DLBCL (n=26, FSC=596) was significantly greater than FL grade3 (n=26, FSC=492), FL grade 1-2 (n=59, FSC=407) or SNGCBL (n= 33, FSC=412). Furthermore, the mean cell size of NBCs of FL grade 3 was significantly greater than that of FL grade 1-2, while the mean cell size of SLL and FL grade 1-2 were about the same. Therefore, evaluation of average size of NBCs by flow cytometry is a potentially useful diagnostic parameter in classification and grading of B-NHL.
Keywords
CP hematopathology, CP flow cytometry, Forward scatter, Diffuse large B-cell lymphoma, Follicular lymphoma, Marginal zone lymphoma, Small lymphocytic lymphoma, Mantle cell lymphoma
Introduction
Evaluation of lymphoma cell size is a key step in the subclassification of B-cell lymphomas. Lymphoma cells can be classified as large, small or (less commonly) medium; but recognition of large B-cells is of particular importance because of their diagnostic and prognostic significance. Lymphomas with increased large cells are often associated with more aggressive clinical course [1-3]. For instance, neoplastic B-cells of diffuse large B-cell lymphoma (DLBCL) are predominately large cells. Follicular lymphoma, grade 3 (FL3) typically have more large neoplastic cells than follicular lymphoma; grade 1-2 (FL1-2). When indolent small B-cell lymphomas transform into more aggressive lymphomas, large neoplastic cells are often markedly increased.
Traditionally, assessment of lymphoma cell size is done by microscopically comparing the lymphoma cells with histiocytes or benign small lymphocytes within the same tissue. Neoplastic cells are considered large if their nuclear size equal or exceeding normal macrophage nuclei or more than twice the size of normal lymphocytes. Unfortunately, this assessment is not always so simple. Lymphoma cells may show a mixture of sizes within the same tissue. In the case of follicular lymphoma, the grading is dependent on counting or estimating the absolute number of centroblasts (typically large) in 10 representative neoplastic follicles, expressed per 40x high-power microscopic field; but this counting method is quite time consuming and has been demonstrated to be poorly reproducible [4-7].
An alternative method for cell size evaluation is flow cytometry, which measure the size of live cells in single cell suspension based on their forward scatter (FSC). The advantage of using flow cytometry as opposed to microscopy lies in its high throughput, precision, and accuracy. To explore if lymphoma cell size can be better evaluated by flow cytometry, we analyzed the average size of neoplastic B-cells (NBCs) in several types of B-cell lymphomas by flow cytometry, comparing the variation in average cell size within diagnostic type, and between different diagnostic types. We also assess the potential diagnostic significance of these results in lymphoma classification and grading.
Materials and Methods
Case selection
This study was approved by the Human Investigation Committee at William Beaumont Hospital. We retrospectively identified 144 lymph node biopsies with B-cell non-Hodgkin lymphoma (B-NHL) diagnosed at William Beaumont Health System from 2006 to 2012. They included 26 cases of DLBCL, 26 cases of FL3, 59 cases of FL1-2, and 33 cases of small non-germinal center B-cell lymphoma, including small lymphocytic lymphoma (13 cases), marginal zone B-cell lymphoma (15 cases) and mantle cell lymphoma (5 cases). The flow cytometric list mode data of these cases were retrieved and reanalyzed. Pathology slides were reviewed for morphologic correlation and confirmation of diagnosis.
Flow cytometric immunophenotyping
Five-color flow cytometric analysis was performed in the clinical flow cytometry laboratory at William Beaumont Hospital. Specimens were stained and analyzed within 24 hours of collection. Fresh tissues of lymph node biopsy specimens were gently teased apart and minced using a forceps and scalpel to prepare single cell suspension. After washing twice and filtering through a 74μm nylon mesh, the single cell suspension was adjusted to a concentration to 5-7x106 cell/ml in phosphate-buffered saline containing 0.1% sodium azide and 0.1% bovine serum albumin (wash buffer). Antibodies were added to a cellular suspension containing 5×105 cells in 100μl. After incubation for 15 minutes in the dark at room temperature, cells were washed twice with the wash buffer and fixed in 1% formaldehyde before acquisition on a flow cytometer (FC500, Beckman Coulter, Miami, FL).
Cell size measurement
All list mode data were re-reanalyzed using CXP software (Beckman Coulter). The gating strategy was designed to measure and compare average mean channel of FSC of NBCs and RTCs. As displayed in Figure 1, debris was excluded based on a density plot of FSC vs. a T-cell marker, typically CD5 or CD3. Lymphocytes and their subsets were identified based on their surface markers (CD3, CD5, CD10, CD19 and CD20). NBCs were identified based on the presence of surface light chain restriction, or (occasionally) absence of surface light chain expression. The mean FSC on a linear scale was collected as a surrogate marker of average cell size of cell populations. Analysis of variance (ANOVA) was applied to compare the difference in average lymphoma cell size among the groups.
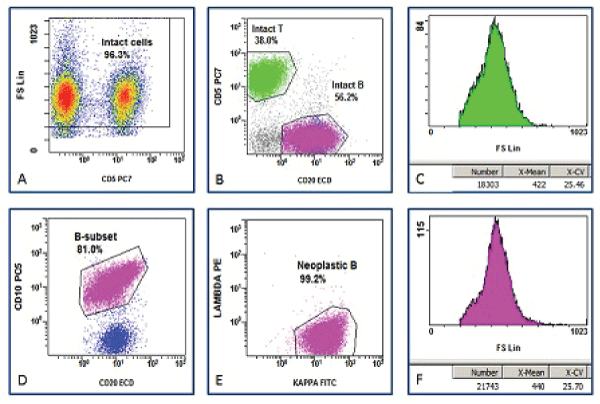
.
Figure 1: Gating protocol for measuring average neoplastic B-cell size. A. Intact cells are gated based a density plot of forward scatter (FSC) vs. CD5. B. Intact T-cells and B-cells are identified based on the expression of CD5 and CD20. C. The average FSC of intact reactive T-cells (RTCs) is displayed. D. Intact B-cell subsets are identified based on expression of CD10 and CD20. E. Intact neoplastic B-cells (NBCs) are identified on light chain expression pattern. E. The average FSC of NBCs is displayed.
View Figure 1
Results
Gating strategy
The cell size spectrum was often continuous on the linear scale of FSC. A numerical cutoff to dividing small, medium and large cells would be arbitrary and difficult to apply reproducibly because a slight change in the position of gate cursor may significantly change their relative percentages. Therefore, we decided to use the mean FSC as a parameter to assess the average cell size of the populations of interest. Cellular fragments were excluded from the analysis. Specifically, events with FSC significantly lower than RTCs on a density plot were considered cellular fragments in all cases. Cases with poorly preserved cell integrity or excessive cellular debris were excluded in this study. This protocol was tested by three of the authors involved in the analysis. No statistically significant differences were seen on the mean FSC of RTCs or NBCs within the same lymphoma types of all cases (data not shown).
The Average cell size of reactive T-cells
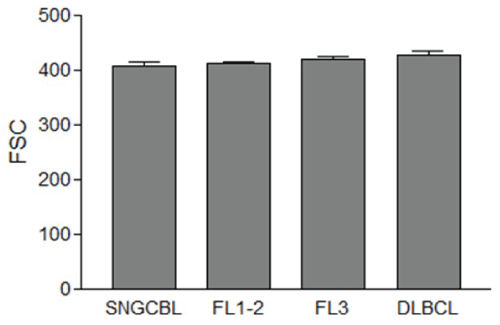
RTCs were detected in all samples of lymphoma tissues in this study. The mean FSC channel values of RTCs were variable from case to case. The mean FSC of RTCs ranged from 351 to 514 among 26 cases of DLBCL, 362 to 474 among 26 samples of FL3, 371 to 487 among 59 samples of FL1-2, and 332 to 470 among 33 cases of SNGCBCL. The mean FSC of RTCs was not affected by lymphoma type or grade (428, 420, 413, and 408 for DLBCL, FL3, FL1-2, SNGCBL, respectively, p>0.05, Figure 2).
.
Figure 2: The average forward scatter (FSC) of reactive T-cells (RTCs) within lymphoma tissue. The FSC means and the standard error of the mean are displayed. The mean FSC of RTCs is not affected by lymphoma type or grade (p>0.05).
SNGCBL: Small Non Germinal Center B-cell Lymphoma, FL1-2: Follicular Lymphoma grade 1-2, FL3: Follicular Lymphoma grade 3, DLBCL: Diffuse Large B-cell Lymphoma.
View Figure 2
Average cell size of neoplastic B-cells
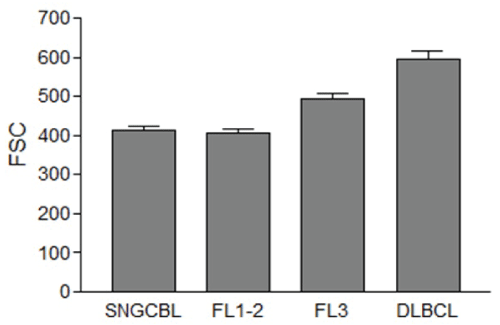
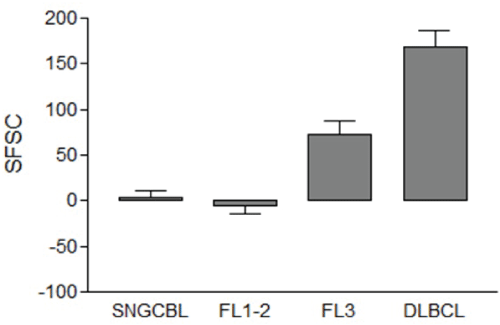
The mean FSC of NBCs of DLBCL, FL3, FL1-2, and SNGBCL were 596, 492, 407 and 412, respectively (Figure 3). The mean FSC of NBCs in DLBCL was significantly greater than FL3, FL1-2 or SNGCBL (p<0.001). The mean FSC of FL3 was also significantly greater than FL1-2 (p<0.001). The mean FSC of FL1-2 was about the same as SNGCBL (p>0.05). The range of mean FSC was between 404 and 799 among 26 DLBCL, 339 to 710 among 26 cases of FL3, 299 to 625 among 59 cases of FL1-2, and 301 and 575 among 33 cases of SNGCBL. To control for variation in patients, laboratory procedures and instrument setting, we standardized the measurement of mean FSC values of lymphoma cells against that of RTCs. The difference in mean FSC values between NBCs and RTCs was calculated by subtracting mean FSC of RTCs from that of NBCs and the net value was defined as a standardized FSC (sFSC). When NBCs were larger than RTCs, the sFSC value became positive. When NBCs were smaller than RTCs, the sFSC value became negative. As displayed in Figure 4, the average sFSC values were 169 for DLBCL, 72 for FL3, -6 for FL1-2 and 3 for SNGCBL. Statistical significance (p<0.01) was observed in sFSC between DLBCL and FL3, and between FL1-2 and FL3. No statistical significance (p>0.05) was detected in sFSC between FL1-2 and SNGCBL. The sFSC was near zero in both FL1-2 and SNGCB indicating that the NBCs in those lymphomas and RTCs were about equal size. The typical cytological features of representative cases are displayed in Figure 5. The sFSC values were predictive of cell size appreciated microscopically. All 26 cases of DLBCL had mean sFSC values greater than 50. Only three of 33 cases of SNGCBL had sFSC >50. These three cases included one case of marginal zone lymphoma with increased scattered large cells. The other two cases were SLL with relatively abundant paraimmunoblasts (as seen Figure 6A) due to increased proliferation centers. There was no overt evidence of large cell transformation in any of the three cases.
.
Figure 3: The average forward scatter (FSC) of neoplastic B-cells (NBCs). The FSC means and the standard error of the mean are displayed. The mean FSC of NBCs in DLBCL is significantly greater than FL3, FL1-2 or SNGCBL (p<0.001). The mean FSC of FL3 is also significantly greater than FL1-2 (p<0.001). The mean FSC of FL1-2 was about the same as SNGCBL (p>0.05).
SNGCBL: Small Non Germinal Center B-cell Lymphoma, FL1-2: Follicular Lymphoma grade 1-2, FL3: Follicular Lymphoma grade 3, DLBCL: Diffuse Large B-cell Lymphoma
View Figure 3
.
Figure 4: The average standardized FSC (sFSC) of neoplastic B-cells (NBCs). The sFSC means and the standard error of the mean are displayed. Statistical significance (p<0.01) is observed in sFSC between DLBCL and FL3, and between FL1-2 and FL3. No statistical significance (p>0.05) is detected in sFSC between FL1-2 and SNGCBL.
sFSC: FSC of neoplastic B-cells- FSC of RTCs, SNGCBL: Small Non Germinal Center B-cell Lymphoma, FL1-2: Follicular Lymphoma grade 1-2, FL3: Follicular Lymphoma grade 3, DLBCL: Diffuse Large B-cell Lymphoma.
View Figure 4
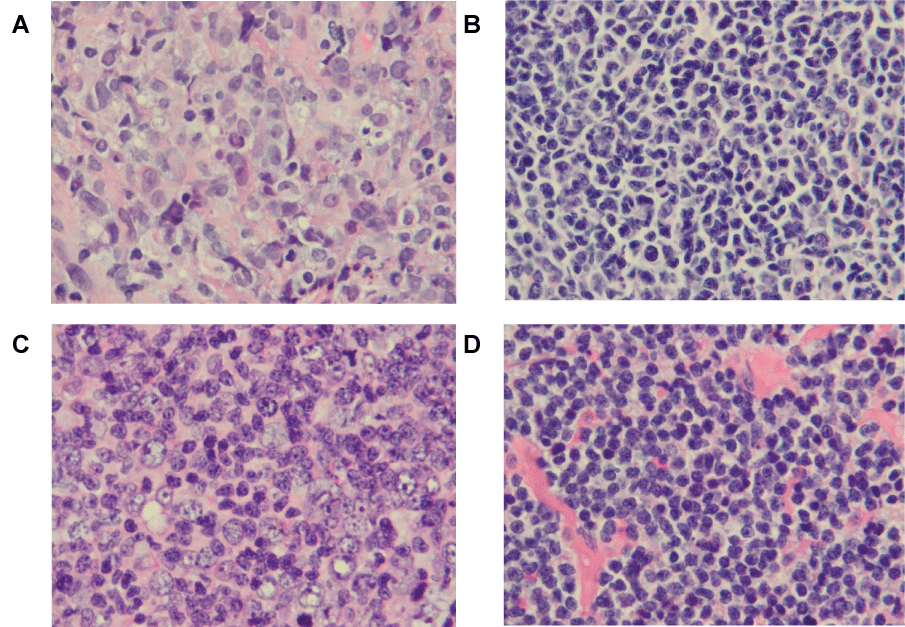
.
Figure 5: Representative cases of B-cell lymphomas with typical morphology. The images were taken from H-E sections of formalin fixed and paraffin-embedded at a magnification of 1000 x. A. Diffuse Large B-cell Lymphoma (DLBCL) with sFSC of 242. B. Follicular Lymphoma grade 1 (FL1-2) with sFSC of -72. C. Follicular Lymphoma grade 3 (FL3) with sFSC of 126. D. Small Lymphocytic Lymphoma (SLL) with sFSC of -1.
sFSC: Standardized Forward Scatter
View Figure 5
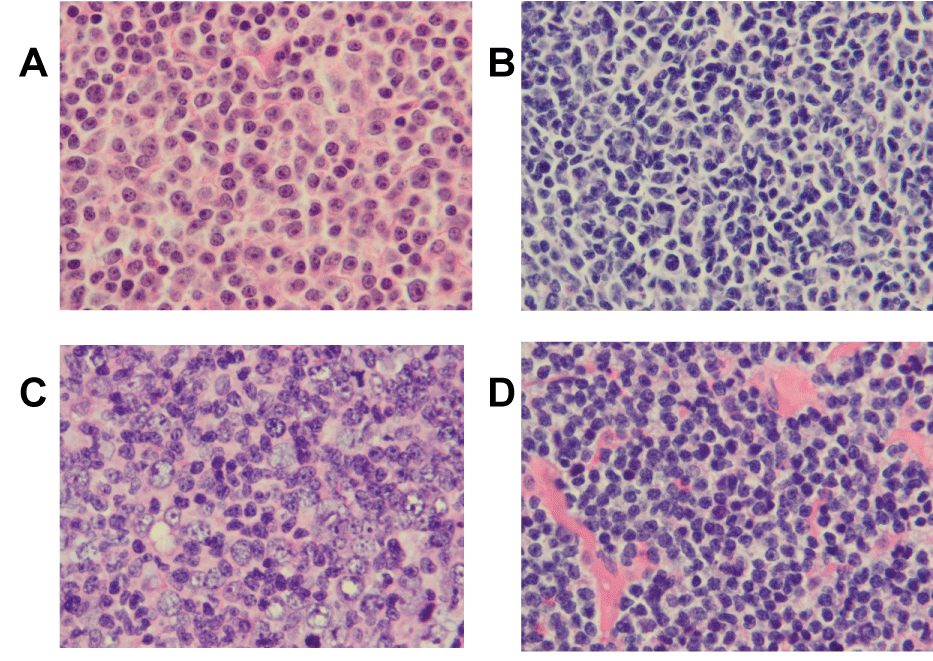
.
Figure 6: Representative cases of B-cell lymphomas with atypical morphology. The images were taken from H-E sections of formalin fixed and paraffin-embedded at a magnification of 1000 x. A. Small Lymphocytic Lymphoma (SLL) with increased paraimmunoblasts with sFSC of 70. B. Follicular Lymphoma grade 3 (FL3) with small centroblasts with sFSC of -5. C. Follicular Lymphoma grade 1-2 (FL1-2) with large centrocytes with sFSC of 121. D. Follicular Lymphoma grade 1-2 (FL1-2) with clear cell changes with sFSC of 128.
sFSC: Standardized Forward Scatter
View Figure 6
To define the potential reference range for sFSC in small cell B-NHL, cases of FL1-2 and SBGCBL were combined into one data set. Of these 92 cases, 14 had sFSC >50, likely due to increased large cells. These 14 cases were considered outliers and therefore excluded from reference range calculation. Of the remaining 78 cases, the average sFSC was -20 with range of -94 to 48 and standard deviation of 37. The 95% and 99% reference range upper limits for sFSC were 54 and 91, respectively. When this reference range was applied to 85 cases of follicular lymphomas, 21 cases showed some discordance between sFSC and reported pathology grade. 13 of 26 (50%) cases of FL3 had unexpected sFSC <54 and 8 of 59 (14%) cases of FL1-2 had unexpected sFSC >54. The H&E stained sections of these cases were re-examined microscopically. All 13 cases of FL3 with relatively low sFSC were excisional biopsies. Ten of these 13 cases showed significant admixture of grade 1-2 and grade 3. The reported pathology grades were based on grade 3. The remaining three cases of FL3 had centroblasts that were predominately small to medium in size (Figure 6B).
Of 8 cases of FL1-2 with unexpectedly high sFSC (>54), 5 cases had increased large centrocytes (Figure 6C). One case consisted predominately of large lymphoma cells with small nuclei and clear cytoplasm (Figure 6D), and two cases were very small needle core biopsies without morphological evidence of multiple grades or significant large centrocytes. Increased sFSC in these two cases suggested the needle core submitted flow cytometry may represent an area different from area the core taken for morphology evaluation. One of these two patients was diagnosed with DLBCL 11 months later in an excisional biopsy.
Discussion
Our flow cytometric approach to evaluate the lymphoma cell size is in agreement with the well-established histologic descriptions of cell size in B-NHLs. Our methods, however seek to improve the objectivity and reproducibility of this critical diagnostic and prognostic assessment. For lymphomas with predominately small or large B-cells, such as typical SLL or DLBCL, histologic reproducibility is likely adequate. But for lymphomas with a mixture of small and large B-cells, such as the centrocytes and centroblasts commonly seen in FL. Quantitative evaluation of relative or absolute number of large cell component based on morphological evaluation is quite challenging and there is room for improvement. It was recommended that in the current WHO classification system that follicular lymphoma should be graded by counting or estimating the absolute number of centroblasts in at least 10 representative and unselected neoplastic follicles. This rule is difficult to follow in practice, especially to avoid the selection bias. Neoplastic follicles with grade 1-2 and grade 3 are commonly mixed together, making histologic grading even more difficult.
Multiple studies have investigated alternative methods for cell size evaluation of histological sections, including computerized image processing [8-11]. To our knowledge, none of these methods have been adopted for routine clinical laboratory practice.
Flow cytometry is often performed in lymphoma workup, mainly for the purpose of immunophenotypic characterization [12]. Only a few studies focused on flow cytometric analysis of lymphoma cell size have been published, and standard operating procedures or interpretation guidelines have yet to be established. Gong et al. [13] studied a series of non-Hodgkin lymphomas by flow cytometry. In this study, neoplastic B-cells were compared with T-cells. A small neoplastic B-cell population was defined as one that had complete overlap of the neoplastic B-cell population with the T-cell population on the forward scatter histogram. A large B-cell population was defined as a complete shift of the neoplastic B-cell population from the T-cell population without overlap. If the neoplastic B-cell population exhibited a significant shift but substantial overlap with the T-cell population, the cells were defined as intermediate. They found that large B-cell lymphomas usually had > 40% large B-cells while low-grade lymphomas had <20% large B-cells. However, the sample size of this study was small with < 10 cases per lymphoma group and statistical analysis was not provided. In practice, this gating strategy is difficult to apply reproducibly, especially in lymphoma tissue with lymphoma cells of variable sizes because of the arbitrary nature of gating cursor placement.
Brandao et al. [14] reported a study of 53 cases of lymph node fine-needle aspiration from patients with histologically confirmed FL. The percentage of CD10 positive small B-cells and large B-cells was calculated based on dual-color flow cytometry data. The cutoff and gating protocols used for separating large from small B-cells was not described. Interestingly, they showed that the percentages of large B-cells or small B-cells based on flow cytometry were able to distinguish follicular lymphoma grade 1 from grade 2 but unable to distinguish grade 2 from grade 3. More recently, Mourad et al. [15] studied thirty-six cases of FL by flow cytometry. They attempted to identify centroblasts using CD19/forward scatter dot plots. The percentage of cells identified beyond the 500-channel mark on CD19/forward scatter dot plot ranged from 0.12% to 12.55% in low grade FL (grades 1-2, median 4.9%) and 6.22% to 51.95% in grade 3FL (median 21%; P=0.00001). In practice, we find it very difficult to use a specific cutoff, such as the 500-channel mark, on the CD19/FSC dot plot to separate small from large cells for several reasons. First, FSC measurement is a relative value and could be significantly affected by instrument settings. Secondly, lymphoma cells in majority of tissue samples show a broad spectrum of FSC without clear separation between small and large cells. A slight change in gating position may significantly change the relative percentage of small and large cells.
Recognizing the arbitrary nature in separating lymphoma cells into small, medium and large, and the likelihood that the percentages generated are not reproducible, we developed a novel approach in the evaluation of cell size. Taking advantage of the ability of flow cytometry to identify neoplastic B-cells, we measured the average size of neoplastic populations. Our protocol does not require separating lymphoma cells into small and large categories, and it can be easily adopted for routine clinical use to generate average cell size (mean FSC) of NBCs and RTCs (as internal control). Laboratories using five color instruments, which are relatively common now, can develop an antibody combination that stains neoplastic B-cells and T-cells simultaneously. These cell populations can then be measured and compared with data obtained from the same tube.
Using this method, we were able to show that average cell size measured in mean FSC of lymphoma cells were statistically different among DLBCL, FL3 and FL1-2. We also confirmed that average cell size measured in mean FSC of neoplastic B-cells were not statistically different among RTCs, FL1-2, and SNGCBL. To control for artificial variations in cell size introduced by instrument settings, tissue preservation, single cell suspension preparation or immunostaining, FSC of NBCs (lymphoma cells) were standardized against that of RTCs (normal small lymphocytes) in this study. RTCs appear to be a valid internal control for three reasons: 1) RTCs are present in all cases of B-cell lymphoma we have studied to date; 2) Although the average size of RTCs measured by flow cytometry varied from sample to sample, in our study, it was not affected by lymphoma type; and 3) RTCs were a convenient population for guiding placement of the gate used for excluding cellular fragments, as RTCs are mostly intact small cells with a homogeneous appearance on FSC vs. CD3 or CD5.
In our study, we refer to the difference in FSC between the NBCs and RTCs as the standardized FSC or sFSC. We believe this parameter can become a useful diagnostic and prognostic tool in many circumstances. Specifically, further study of sFSC in lymphomas diagnosed by fine needle aspiration should be undertaken to determine if grading can be performed on a cytology sample only, obviating the need for excisional biopsy, especially for surgically difficult cases. In addition, sFSC may prove to be a useful tool in comparing cells size in histologic and cytologic specimens from the same patient, acquired simultaneously or separately. More importantly, sFSC in FL may offer a more reproducible and quantitative measurement of lymphoma cell size in assisting lymphoma classification. Still, validation of potential sFSC cutoffs for prognostic grading with clinical outcome correlation would be necessary in follicular lymphoma.
If clinical validation is pursued, it is conceivable that flow cytometry could afford some advantages in grading lymphomas versus histology. For instance, it is quite common to see variable numbers of centroblasts from follicle to follicle. If a few individual follicles with grade 3 are not clinically significant, it may be better to use the average cell size based on flow cytometry. On the other hand, rare cases of follicular lymphomas with small centroblasts, large centrocytes or clear cell change, might pose a challenge. It would be interesting to see which correlates better with clinical outcome: cell type by histology or cell size by flow cytometry. Of great interest, in this regard, is a recent study by Kjeldsen et al. [16] that reported that centroblasts and centrocytes from normal human tonsil tissues were distinctively different in expression of CXCR4. If this finding proves to be reproducible and applicable to neoplastic follicular lymphoma cells, it could help mitigate discrepancies between histologic type and cell size evaluation by flow cytomety. More importantly, it will allow flow cytometry to further stratify follicular lymphomas based on more objective and reproducible evaluation of the relative percentage of large centroblasts, large centrocytes, small centroblasts and small centrocytes.
References
-
Chisté M, Vrotsos E, Zamora C, Martinez A (2013) Chronic lymphocytic leukemia/small lymphocytic lymphoma involving the aortic valve. Ann Diagn Pathol 17: 295-297.
-
Jaffe ES, Harris NL, Stein H, Isaacson PG (2008) Classification of lymphoid neoplasms: the microscope as a tool for disease discovery. Blood 112: 4384-4399.
-
Marcos-Gragera R, Allemani C, Tereanu C, De Angelis R, Capocaccia R, et al. (2011) Survival of European patients diagnosed with lymphoid neoplasms in 2000-2002: results of the HAEMACARE project. Haematologica. 96:720-728
-
Mann RB, Berard CW (1983) Criteria for the cytologic subclassification of follicular lymphomas: a proposed alternative method. Hematol Oncol 1: 187-192.
-
Nathwani BN, Metter GE, Miller TP, Burke JS, Mann RB, et al. (1986) What should be the morphologic criteria for the subdivision of follicular lymphomas? Blood. 68: 837-845.
-
A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin's lymphoma (1997) The Non-Hodgkin's Lymphoma Classification Project. Blood 89: 3909-3918.
-
Piccaluga PP, Pileri SA, Agostinelli C, Klapper W, Lennert K (2011) Follicular lymphoma: still Six characters in search of an author? Leuk Lymphoma 52: 1655-1667.
-
Preston K Jr, Firestone LM, Nathwani BN (1990) Use of three-dimensional mathematical morphology in analyzing histologic images, with application to the subtyping of follicular lymphomas. Anal Quant Cytol Histol.12: 399-416.
-
Link NG, Nathwani BN, Preston K Jr (1989) Subclassification of follicular lymphomas by computerized image processing. Anal Quant Cytol Histol 11: 119-130.
-
Martinez AE, Lin L, Dunphy CH (2007) Grading of follicular lymphoma: comparison of routine histology with immunohistochemistry. Arch Pathol Lab Med 131: 1084-1088.
-
Sertel O, Lozanski G, Shana'ah A, Gurcan MN (2010) Computer-aided detection of centroblasts for follicular lymphoma grading using adaptive likelihood-based cell segmentation. IEEE Trans Biomed Eng 57: 2613-2616.
-
Craig FE, Foon KA (2008) Flow cytometric immunophenotyping for hematologic neoplasms. Blood 111: 3941-3967.
-
Gong JZ, Williams DC Jr, Liu K, Jones C (2002) Fine-needle aspiration in non-Hodgkin lymphoma: evaluation of cell size by cytomorphology and flow cytometry. Am J Clin Pathol 117: 880-888.
-
Brandao GD, Rose R, McKenzie S, Maslak P, Lin O (2006) Grading follicular lymphomas in fine-needle aspiration biopsies: the role of ThinPrep slides and flow cytometry. Cancer 108: 319-323.
-
Mourad WA, Rawas F, Shoukri M, Tbakhi A, Al Omari M, et al. (2006) Grading of follicular lymphoma using flow cytometry. Ann Saudi Med 26: 205-210.
-
Kjeldsen MK, Perez-Andres M, Schmitz A, Johansen P, Boegsted M, et al. (2011) Multiparametric flow cytometry for identification and fluorescence activated cell sorting of five distinct B-cell subpopulations in normal tonsil tissue. Am J Clin Pathol 136: 960-969.





