International Journal of Oral and Dental Health
Medication-Related Osteonecrosis of the Jaw with the mTOR Inhibitor Everolimus in a Patient with Estrogen-Receptor Positive Breast Cancer: A Case Report
Cameron YS Lee1*, Kristin L Lee2, Kirk Y Hirata3 and Jon B Suzuki4
1Department of Periodontology and Oral Implantology, Kornberg School of Dentistry, Temple University, Philadelphia, USA
2Creighton University School of Dentistry, Omaha, USA
3John A. Burns School of Medicine, University of Hawaii, Honolulu, USA
4School of Medicine, Kornberg School of Dentistry, Temple University, Philadelphia, USA
*Corresponding author:
Cameron YS Lee, DMD, MD, PHD, MPH, Private Practice in Oral, Maxillofacial and Reconstructive Surgery, Aiea, Hawaii; Clinical Professor, Department of Periodontology and Oral Implantology, Kornberg School of Dentistry, Temple University, Philadelphia, PA 19140, USA, Tel: 808-484-2288, Fax: 808-484-1181, E-mail: clee555294@aol.com
Int J Oral Dent Health, IJODH-2-033, (Volume 2, Issue 3), Case Report; ISSN: 2469-5734
Received: August 08, 2016 | Accepted: September 15, 2016 | Published: September 17, 2016
Citation: Lee CYS, Lee KL, Hirata KY, Suzuki JB (2016) Medication-Related Osteonecrosis of the Jaw with the mTOR Inhibitor Everolimus in a Patient with Estrogen-Receptor Positive Breast Cancer: A Case Report. Int J Oral Dent Health 2:033. 10.23937/2469-5734/1510033
Copyright: © 2016 Lee CYS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: In the treatment of bone metastases from breast cancer, bisphosphonates are considered the cornerstone of treatment to prevent adverse skeletal related events. Intravenous bisphosphonates reduce bone resorption by inhibiting osteoclast function. However, osteonecrosis of the jaws is a commonly reported side effect with intravenous bisphosphonates to treat malignancies, such as breast cancer and restricts their clinic use. New targeted therapies have recently been introduced to improve progression-free survival in postmenopausal women with estrogen receptor-positive advanced breast cancer. Everolimus is a mammalian target of rapamycin (mTOR) inhibitor used in combination with an aromatase inhibitor to treat advanced breast cancer, but may lead to development of osteonecrosis of the jaw that has a similar clinical presentation from bisphosphonate therapy.
Case description: This case report describes the clinical presentation and management of a postmenopausal woman who developed osteonecrosis of the jaw while on targeted therapy with everolimus after extraction of a mandibular molar tooth.
Practical implications: As everolimus has recently been approved by the Food and Drug Administration in the treatment of advanced breast cancer, the dental and medical community must be aware that such new targeted therapies in breast cancer treatment are potential risk factors for development of osteonecrosis of the jaw.
Keywords
Everolimus, Exemestane, Estrogen-receptor positive advanced breast cancer, Mammalian target of rapamycin inhibitor, Medication-related osteonecrosis of the jaw (MRONJ)
Introduction
Breast cancer is the most common cancer diagnosed in women globally and the most frequent cause of death among all cancers in women [1,2]. In the United States, 21% of women are diagnosed with breast cancer under the age of 50-years (approximately 49,000 women) and is a leading cause of death in this age group [1]. It is estimated that over 40,000 women will die of this disease in 2015 [2]. More than 50% have estrogen-receptor positive (ER+) disease, although estrogen is a steroid sex hormone required for normal breast development [3]. There is a strong correlation between exposure to estrogen and development of breast cancer [3,4]. Breast cancer due to elevated blood levels of estrogen is the most frequent type of invasive malignancy that affects over 1 million women worldwide [4]. Breast carcinogenesis is due to metabolism of estrogen to genotoxic metabolites and cellular growth and proliferation that leads to initiation, promotion and development of breast cancer [5].
Targeted estrogen therapies are the current treatment regimen for postmenopausal women diagnosed with advanced ER+ breast cancer [6]. Aromatase inhibitors (Exemestane, Pfizer) combined with the mammalian target of rapamycin (mTOR) inhibitor everolimus (Afinitor, Novartis Pharmaceuticals) are chemotherapeutic drugs that decrease estrogen levels and have been found to significantly reduce invasive breast cancers in postmenopausal women [6,7]. They are also more effective in preventing recurrence of malignant disease than the selective estrogen-receptor modulator (SERM) medications tamoxifen and raloxifene [8]. As a result, there is improved survival rates due to the increased use of such targeted estrogen therapies [6-8].
In the treatment of bone metastases from breast cancer, bisphosphonates are considered the cornerstone of treatment [9]. Intravenous bisphosphonates reduce bone resorption by inhibiting osteoclast function, but an adverse side effect that restricts their clinic use is osteonecrosis of the jaw [10]. Medication-related osteonecrosis of the jaw (MRONJ) is a rare disease characterized by exposed bone with or without pain of patients prescribed bisphosphonates in the treatment of malignancies such as multiple myeloma, and cancer of the prostate gland and breast [10-12].
Since the first report of osteonecrosis of the jaw was reported by Marx [13] in 2003, there have been reports of other pharmacotherapeutic classes of drugs that may also cause osteonecrosis of the jaw. Therefore, the American Association of Oral and Maxillofacial Surgeons changed the nomenclature from bisphosphonate-related osteonecrosis of the jaw (BRONJ) to medication-related osteonecrosis of the jaw (MRONJ) [11,12].
Similar to bisphosphonate medications, everolimus has been implicated as a risk factor in the development of osteonecrosis of the jaw [14]. To our knowledge, this is the first case report in the English dental and medical world literature describing the adverse effect of osteonecrosis of the jaw where an mTOR inhibitor may be an independent risk factor without past or concurrent use of intravenous bisphosphonates. In this case report, the goal was to determine the role of everolimus, an mTOR inhibitor as a possible independent risk factor for the development of this debilitating condition of the jaw. The clinical presentation appears very similar to osteonecrosis of the jaw with bisphosphonate therapy.
Estrogen and Bone Physiology
In the adult human skeleton, bone remodeling and metabolism is a result of a homeostatic balance between resorption of bone by osteoclasts and bone remodeling by osteoblasts [15,16]. During bone remodeling, estrogen has antiresorptive effects on osteoclastic activity and stimulates osteoclast apoptosis [17,18]. This steroid sex hormone also modulates osteoblast cytokines which decreases differentiation and maturation of osteoclasts. However, estrogen may also induce direct effects on osteoblasts, where bone formation occurs more than resorption.
In the postmenopausal state, estrogen production is increased from the ovaries to adipose tissue, adrenal glands, smooth muscle and bone [17,18]. Androgens produced by the adrenal glands circulates in peripheral tissues and is converted to estrogen by the action of aromatase (also known as estrogen synthetase) that is critical for overall bone health. Estrogen deficiency that occurs naturally or in the postmenopausal state can increase osteoclastic activity. The result is increased bone resorption and poor bone quality and increased rates of skeletal fractures [18].
Aromatase Inhibitors
Estrogen is the primary hormone involved in the development and proliferation of breast tumor cells [3,19]. Suppression of estrogen synthesis is a key targeted strategy against breast cancer [3-5,8]. There are two classes of third generation orally administered aromatase inhibitors: exemestane, which is a steroid inhibitor, and the nonsteroidal inhibitors, anastrozole and letrozole [20,21]. Aromatase inhibitors suppress plasma estrogen levels by inactivating aromatase in postmenopausal women, which is the enzyme responsible for estrogen synthesis from the androgenic substrates androstenedione and testosterone in peripheral tissues [20-22]. The overall result is a reduction of circulating endogenous estrogen levels in peripheral blood and tumors that are important in preventing recurrence in estrogen receptor positive breast cancer. However, estrogen targeted therapy with aromatase inhibitors in the treatment of breast cancer can also affect the health of the human skeleton [23]. As estrogen production is suppressed with targeted therapy, this may lead to additional bone resorption and increased rates of skeletal fracture.
Mammalian Target of Rapamycin Inhibitor
Everolimus is a mammalian target of rapamycin (mTOR) inhibitor which has been approved for the treatment of ER+ advanced breast cancer in postmenopausal women with exemestane [6]. Activation of the mTOR signaling pathway by cancer cells is a strategic survival mechanism with endocrine (estrogen) resistance [24,25]. The mammalian target of rapamycin (mTOR) is a serine-threonine kinase that stimulates cell growth, proliferation, angiogenesis, protein and ribosomal biosynthesis [26]. Activation of this kinase stimulates cancer growth.
Research has discovered a close relationship between estrogen receptor signaling and the mTOR intracellular pathway [27-29]. Everolimus combined with exemestane selectively targets mTOR resulting in antitumor activity [7]. Inhibition of mTOR down regulates the cellular process of growth and proliferation of cancer cells. By inhibition of mTOR, expression of vascular endothelial growth factor (VEGF) and angiogenesis are also depressed [14]. Based on the BOLERO-2 trial, combining everolimus with exemestane statistically improved progression-free survival in postmenopausal women with estrogen-receptor positive advanced breast cancer, as both drugs synergistically decrease growth and proliferation of cancer cells [14].
Reported adverse side effects with the use of everolimus include the following: stomatitis, gingivitis, dermatitis, pneumonitis, metabolic and blood disorders, infections, bleeding, impairment of wound healing and osteonecrosis of the jaw [14].
Case Report
A 66-year-old female was referred to our oral and maxillofacial surgery practice (cysl) from her family dentist for consultation and management of a painful, non-healing extraction site in the left posterior mandible. The patient's dental history was significant for extraction of tooth #18 four months ago by her dentist. Past medical history was significant for hypothyroidism and estrogen receptor positive advanced cancer of the left breast diagnosed in 2007. The patient denied a history of osteoporosis or osteopenia. Treatment consisted of surgical lumpectomy and the nonsteroidal aromatase inhibitor, tamoxifen. In 2014, there was a recurrence of the disease and mastectomy of the left breast in April 2015. The patient's medical history was negative for radiation treatment, bisphosphonate or monoclonal antibody therapy. Her medications at the time of presentation to our office included the following: levothyroxine 100 mcg, exemestane, 25 mg and everolimus, 10 mg daily.
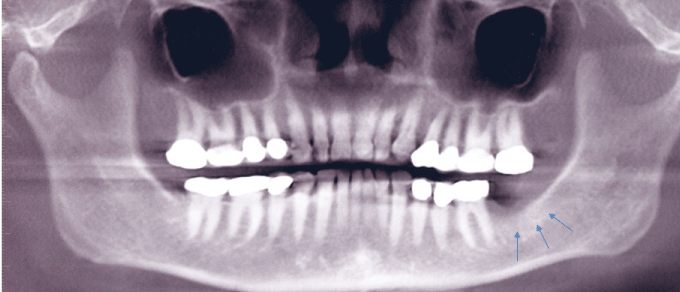
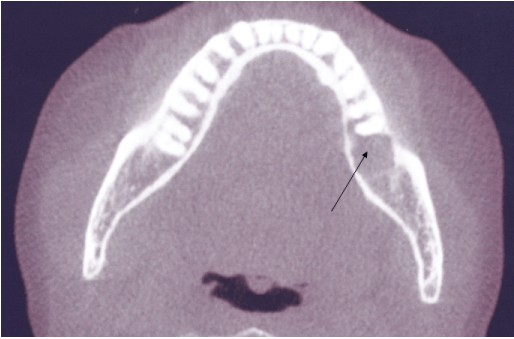
At the time of presentation, the maxillofacial examination was unremarkable. No facial swelling, or orocutaneous fistula was observed. Oral examination of the left posterior mandible revealed edema and erythema of the gingiva and mucosa in the area of the extraction site with exposed bone. The non-healing area was extremely tender to palpation. No purulent discharged was observed. Imaging studies consisting of a panoramic radiograph revealed increased osteosclerosis and trabecular density (Figure 1). Cone beam CT scan in the axial view showed osteolysis in the area of the extraction site (Figure 2).
.
Figure 1: Panoramic radiograph revealed increased osteosclerosis and trabecular density.
View Figure 1
.
Figure 2: Axial view cone beam CT scan of left posterior mandible shows osteolysis in area of extraction site.
View Figure 2
Based on the clinical examination with a greater than 8 week history of exposed bone in the mandible and no history of radiation treatment, the diagnosis was consistent with Stage II osteonecrosis of the mandible that was indistinguishable from Stage II osteonecrosis of the mandible with bisphosphonate therapy based on the American Association of Oral and Maxillofacial Surgeons MRONJ Classification [11,12]. Stage II is characterized by erythema, exposed necrotic bone for greater than 8 weeks, and pain without purulent discharge. However, in this specific case, MRONJ was not associated with bisphosphonate therapy.
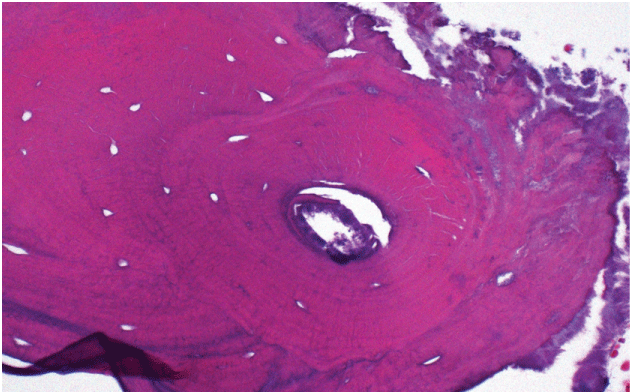
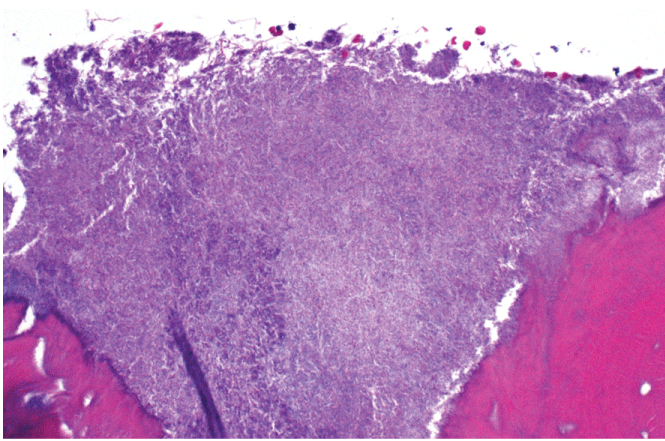
The patient was informed of the clinical findings and was treated with surgical debridement with a round bur in a high speed hand piece under cool water irrigation under local anesthesia until bleeding viable bone was observed. Exploration of the buccal and lingual cortex and alveolus of the extraction site revealed areas of bone that appeared non-vital. Adjunctive therapy with platelet rich plasma obtained from venous blood of the right upper extremity was sprayed on to the mandible and soft tissues to enhance wound healing [30-33]. To rule-out metastasis to the mandible, a hard tissue biopsy was performed. Histologic examination was negative for metastases from the breast, but consistent with osteonecrosis. Cultures revealed mixed microbial flora with actinomyces (Figure 3 and Figure 4). To manage the actinomycosis, the patient was referred to an infectious disease specialist for antibiotic therapy which consisted of a 12-week regimen of oral clindamycin 300 mg four times per day as the patient was allergic to penicillin. Follow-up observation over the following six months revealed complete soft tissue healing with no signs of refractory osteonecrosis and exposed bone.
.
Figure 4: High power view demonstrates colony of actinomyces (purple staining) along necrotic bone.
View Figure 4
Discussion
Breast cancer affects over 1 million women world-wide with metastases occurring in 65% to 75% of patients [29]. Intravenous bisphosphonates are highly effective in preventing complications due to bone metastases and skeletal related events [10-12]. They inhibit bone resorption and facilitate osteoclast apoptosis [34-36]. However, a major adverse side effect is osteonecrosis of the jaw (ONJ) [11,12,37,38]. Recently, there have been reports of MRONJ from non-bisphosphonate therapy, such as denosumab (human monoclonal immunoglobulin antibody) [39-41], sunitinib and bevacizumab (VEGF inhibitors) [42,43] and trastuzumab (antiangiogenic) [44] and the mTOR inhibitor, everolimus [14,45-47]. All of these medications disrupt the bone remodeling cycle due to their different mechanisms of action.
Concurrent administration with bisphosphonates may also increase the risk for the development of MRONJ [44-47]. There have only been three reported cases in the world literature describing osteonecrosis of the jaws from targeted cancer therapy with everolimus. In each case report the patient was previously treated with the intravenous bisphosphonate, zoledronic acid [45-47]. Kim, et al. [45] reported that their patient presented for evaluation and treatment of exposed bone in the right posterior mandible. Twelve years previous, tooth #31 was extracted. Past medical history was significant for medullary thyroid cancer that was treated by total thyroidectomy and intravenous zoledronic acid for bone metastases for the next five years. At the time of presentation, the patient was already taking everolimus 10 mg daily for the past 3-years. The authors speculate that because the last dose of zoledronic acid was prescribed six years ago, such a long interval without bisphosphonates may not have contributed to development of osteonecrosis of the mandible. Instead, the addition of everolimus could have precipitated the development of MRONJ. In the second case by Giancola, et al. [46] their patient was treated with zoledronic acid for two years for renal cancer that metastasized to the skeleton prior to initiating therapy with everolimus. In the case reported by Martini and colleagues [47], their patient was prescribed zoledronic acid for a period of six months for bone metastases from renal cell carcinoma before instituting sunitinib and everolimus therapy. Because the half-life of bisphosphonates in bone is greater than 10-years [48,49], we are of the opinion that in all three case reports, administration of intravenous zoledronic acid previous to everolimus therapy likely contributed to the development of MRONJ in their patient.
In our case report, we describe development of osteonecrosis of the mandible in a postmenopausal women on combined therapy with exemestane and everolimus after extraction of a mandibular molar tooth. To our knowledge, there are no case reports of MRONJ from exemestane. Several hypotheses have been presented in the literature to explain the pathogenesis of MRONJ due to bisphosphonates. Marx and colleagues hypothesize that bone turnover is effectively inhibited as the primary action of bisphosphonates is the inhibition of osteoclastic-mediated bone resorption [50,51]. The major toxic effect is cellular apoptosis of osteoclasts where coupling of osteoclastic and osteoblastic activity is disrupted resulting in suppression of bone turnover. A second theory is that MRONJ is due to the antiangiogenic effects of bisphosphonates that affects vascularization, inhibits angiogenesis and ultimately, delays wound healing [52]. Further, oral epithelial cells are exposed to localized increased toxic concentrations of bisphosphonates following trauma resulting in compromised epithelial wound healing, jaw exposure and development of osteonecrosis of the jaw [53]. Another theory is local tissue acidosis in the jaw due to bisphosphonates may elicit the onset of osteonecrosis [54]. Lee and Suzuki hypothesize that actinomyces may play a critical role in the pathogenesis of medication-related osteonecrosis of the jaws (MRONJ) [55]. Recognition of actinomycosis is critical. In the presence of signs and symptoms of soft tissue edema, erythema, exposed bone and suppuration, actinomycosis should be recognized as a primary diagnosis and specific treatment directed at eradication of this bacterial pathogen. The authors of this case report are of the opinion that the mechanism of osteonecrosis of the jaw from everolimus may involve all of the theories mentioned above due their effects on bone homeostasis, angiogenesis and the microbial pathogen, actinomyces. Therefore, new targeted cancer therapy with mTOR inhibitors may be another risk factor in the development of MRONJ.
Conclusion
The dental community must be aware that although medication-related osteonecrosis of the jaws is most commonly associated with intravenous bisphosphonates therapy for malignancies such as breast cancer, new targeted therapies in breast cancer treatment are potential risk factors for development of osteonecrosis of the jaw. For each dental practitioner that encounters the breast cancer patient, obtaining an up to date medical history is important especially if the patient is treatment planned for surgery involving the jaws. The risk of osteonecrosis of the jaw must always be considered and the patient must be educated on the potential risk from their cancer therapy. We recommend that oral health care providers follow the existing recommendations for the prevention of MRONJ.
Acknowledgments
The authors declare no financial interest in any of the products cited in this manuscript.
References
-
DeSantis C, Ma J, Bryan L, Jemal A (2014) Breast cancer statistics, 2013. CA Cancer J Clin 64: 52-62.
-
(2015) What are the key statistics about breast cancer? American Cancer Society.
-
Yager JD, Davidson NE (2006) Estrogen carcinogenesis in breast cancer. N Engl J Med 354: 270-282.
-
Clemons M, Goss P (2001) Estrogen and the risk of breast cancer. N Engl J Med 344: 276-285.
-
Yue W, Santen RJ, Wang JP, Li Y, Verderame MF, et al. (2003) Genotoxic metabolites of estradiol in breast: potential mechanism of estradiol induced carcinogenesis. J Steroid Biochem Mol Biol 86: 477-486.
-
Baselga J, Campone M, Piccart M, Burris HA 3rd, Rugo HS, et al. (2012) Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N Engl J Med 366: 520-529.
-
Boulay A, Rudloff J, Ye J, Zumstein-Mecker S, O'Reilly T, et al. (2005) Dual inhibition of mTOR and estrogen receptor signaling in vitro induces cell death in models of breast cancer. Clin Cancer Res 11: 5319-5328.
-
Coombes RC, Hall E, Gibson LJ, Paridaens R, Jassem J, et al. (2004) A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med 350: 1081-1092.
-
Lewiecki EM, Miller PD, McClung MR, Cohen SB, Bolognese MA, et al. (2007) Two-year treatment with denosumab (AMG 162) in a randomized phase 2 study of postmenopausal women with low BMD. J Bone Miner Res 22: 1832-1841.
-
Tanvetyanon T, Stiff PJ (2006) Management of the adverse effects associated with intravenous bisphosphonates. Ann Oncol 17: 897-907.
-
Ruggiero SL, Dodson TB, Assael LA, Landesberg R, Marx RE, et al. (2009) American Association of Oral and Maxillofacial Surgeons position paper on bisphosphonate-related osteonecrosis of the jaws. 2009 Update. J Oral Maxillofac Surg 67: 2-12
-
Ruggiero SL, Dodson TB, Fantasia J, Goodday R, Aghaloo T, et al. (2014) American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw- 2014 Update. J Oral Maxillofac Surg 72: 1938-1956.
-
Marx RE (2003) Pamidronate (aredia) and zoledronate (zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg 61: 1115-1117.
-
Gnant M, Baselga J, Rugo HS, Noguchi S, Burris HA, et al. (2013) Effect of everolimus on bone marker levels and progressive disease in bone in BOLERO-2. J Natl Cancer Inst 105: 654-663.
-
Seeman E, Delmas PD (2006) Bone quality- the material and structural basis of bone strength and fragility. N Engl J Med 354: 2250-2261.
-
Canalis E, Giustina A, Bilezikian JP (2007) Mechanisms of anabolic therapies for osteoporosis. N Engl J Med 357: 905-916
-
Riggs BL, Khosla S, Melton LJ 3rd (2002) Sex steroids and the construction and conservation of the adult skeleton. Endocr Rev 23: 279-302.
-
Hofbauer LC, Schoppet M (2004) Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. JAMA 292: 490-495.
-
Jensen EV, Jordan VC (2003) The estrogen receptor: a model for molecular medicine. Clin Cancer Res 9: 1980-1989.
-
Goss PE (2003) Breast cancer prevention- clinical trials strategies involving aromatase inhibitors. J Steroid Biochem Mol Biol 86: 487-493.
-
Smith IE, Dowsett M (2003) Aromatase inhibitors in breast cancer. N Engl J Med 348: 2431-2442.
-
Goss PE, Ingle JN, Alés-Martínez JE, Cheung AM, Chlebowski RT, et al. (2011) Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med 364: 2381-2391.
-
Osborne C, Tripathy D (2005) Aromatase inhibitors: rationale and use in breast cancer. Annu Rev Med 56: 103-116.
-
Yamnik RL, Digilova A, Davis DC, Brodt ZN, Murphy CJ, et al. (2009) S6 kinase 1 regulates estrogen receptor alpha in control of breast cancer cell proliferation. J Biol Chem 284: 6361-6369.
-
Efeyan A, Sabatini DM (2010) mTOR and cancer: many loops in one pathway. Curr Opin Cell Biol 22: 169-176.
-
Bjornsti MA, Houghton PJ (2004) The TOR pathway: a target for cancer therapy. Nat Rev Cancer 4: 335-348.
-
Campbell RA, Bhat-Nakshatri P, Patel NM, Constantinidou D, Ali S, et al. (2001) Phosphatidylinositol 3-kinase/AKT-mediated activation of estrogen receptor alpha: a new model for anti-estrogen resistance. J Biol Chem 276: 9817-9824.
-
Santen RJ, Song RX, Zhang Z, Kumar R, Jeng MH, et al. (2005) Adaptive hypersensitivity to estrogen: mechanisms and clinical relevance to aromatase inhibitor therapy in breast cancer treatment. J Steroid Biochem Mol Biol 95: 155-165.
-
Yue W, Fan P, Wang J, Li Y, Santen RJ (2007) Mechanisms of acquired resistance to endocrine therapy in hormone-dependent breast cancer cells. J Steroid Biochem Mol Biol 106: 102-110
-
Marx RE, Carlson ER, Eichstaedt RM, Schimmele SR, Strauss JE, et al. (1998) Platelet-rich plasma: Growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 85: 638-646.
-
Marx RE (2004) Platelet-rich plasma: evidence to support its use. J Oral Maxillofac Surg 62: 489-496.
-
Lee CY, David T, Nishime M (2007) Use of platelet-rich plasma in the management of oral biphosphonate-associated osteonecrosis of the jaw: a report of 2 cases. J Oral Implantol 33: 371-382.
-
Lee CY, Suzuki JB (2015) Medication-related osteonecrosis of the jaws from once per year intravenous zoledronic acid (Reclast): report of 4 cases. Implant Dent 24: 227-231.
-
Rochefort GY, Pallu S, Benhamou CL (2010) Osteocyte: the unrecognized side of bone tissue. Osteoporos Int 21: 1457-1469
-
Subramanian G, Cohen HV, Quek SYP (2011) A model for the pathogenesis of bisphosphonate-associated osteonecrosis of the jaw and teriparatide's potential role in its resolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 112: 744-753.
-
Pozzi S, Raje N (2011) The role of bisphosphonates in multiple myeloma: mechanisms, side effects, and the future. Oncologist 16: 651-662.
-
Marx RE (2003) Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: A growing epidemic. J Oral Maxillofac Surg 61: 1115-1117.
-
Marx RE, Sawatari Y, Fortin M, Broumand V (2005) Bisphosphonate-induced exposed bone (osteonecrosis-osteopetrosis) of the jaws: risk factors, recognition, prevention, treatment. J Oral Maxillofac Surg 63: 1567-1575.
-
Aghaloo TL, Felsenfeld AL, Tetradis S (2010) Osteonecrosis of the jaw in a patient on Denosumab. J Oral Maxillofac Surg 68: 959-963
-
Malan J, Ettinger K, Naumann E, Beirne OR (2012) The relationship of denosumab pharmacology and osteonecrosis of the jaws. Oral Surg Oral Med Oral Pathol Oral Radiol 114: 671-676.
-
Pichardo SE, Kuypers SC, van Merkesteyn JP (2013) Denosumab osteonecrosis of the mandible: a new entity? A case report. J Craniomaxillofac Surg 41: e65-69.
-
Fleissig Y, Regev E, Lehman H (2012) Sunitinib related osteonecrosis of jaw: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol 113: e1-3.
-
Santos-Silva AR, Belizário Rosa GA, Castro Júnior Gd, Dias RB, Prado Ribeiro AC, et al. (2013) Osteonecrosis of the mandible associated with bevacizumab therapy. Oral Surg Oral Med Oral Pathol Oral Radiol 115: e32-36.
-
Pilanci KN, Alco G, Ordu C, Sarsenov D, Celebi F, et al. (2015) Is administration of trastuzumab an independent risk factor for developing osteonecrosis of the jaw among metastatic breast cancer patients under zoledronic acid treatment? Medicine (Baltimore) 94: e671.
-
Kim DW, Jung YS, Park HS, Jung HD (2013) Osteonecrosis of the jaw related to everolimus: a case report. Br J Oral Maxillofac Surg 51: e302-304.
-
Giancola F, Campisi G, Lo Russo L, Muzio LL, Di Fede O (2013) Osteonecrosis of the jaw related to everolimus and bisphosphonate: a unique case report? Ann Stomatol (Roma) 4: 20-21.
-
Martini V, Bonacina R, Mariani U (2014) Osteonecrosis of the jaw in a patient treated with zoledronic acid and everolimus: a case report. Annali di Stomatologia 2: 27.
-
Fleisch H (2003) Bisphosphonates in osteoporosis. Eur Spine J 12: S142-146.
-
Diab DL, Watts NB (2013) Bisphosphonate drug holiday: who, when and how long. Ther Adv Musculoskelet Dis 5: 107-111.
-
Marx RE, Sawatari Y, Fortin M, Broumand V (2005) Bisphosphonate-induced exposed bone (osteonecrosis/osteopetrosis) of the jaws: risk factors, recognition, prevention, and treatment. J Oral Maxillofac Surg 63: 1567-1575.
-
Marx RE, Cillo JE Jr, Ulloa JJ (2007) Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg 65: 2397-2410.
-
Fournier P, Boissier S, Filleur S, Guglielmi J, Cabon F, et al. (2002) Bisphosphonates inhibit angiogenesis in vitro and testosterone-stimulated vascular regrowth in the ventral prostate in castrated rats. Cancer Res 62: 6538-6543.
-
Landesberg R, Cozin M, Cremers S, Woo V, Kousteni S, et al. (2008) Inhibition of oral mucosal cell wound healing by bisphosphonates. J Oral Maxillofac Surg 66: 839-847.
-
Otto S, Hafner S, Mast G, Tischer T, Volkmer E, et al. (2010) Bisphosphonate-related osteonecrosis of the jaw: is pH the missing part in the pathogenesis puzzle? J Oral Maxillofac Surg 68: 1158-1161
-
Lee CY, Pien FD, Suzuki JB (2011) Identification and treatment of bisphosphonate-associated actinomycotic osteonecrosis of the jaws. Implant Dent 20: 331-336