International Journal of Oral and Dental Health
Late Diagnosis of CGCL in a Noonan Syndrome Patient
N AlAsseri1 and M AlSaeed2*
1Oral and Maxillofacial Surgery Consultant in Prince Sultan Military Medical City, Riyadh, Saudi Arabia
2Senior Dental student at Riyadh Colleges of Dentistry and Pharmacy, Riyadh, Saudi Arabia
*Corresponding author:
M AlSaeed, Senior Dental student at Riyadh Colleges of Dentistry and Pharmacy, Riyadh, Saudi Arabia, E-mail: Monirah.AlSaeed@gmail.com
Int J Oral Dent Health, IJODH-2-030, (Volume 2, Issue 3), Case Report; ISSN: 2469-5734
Received: April 02, 2016 | Accepted: July 02, 2016 | Published: July 05, 2016
Citation: AlAsseri N, AlSaeed M (2016) Late Diagnosis of CGCL in a Noonan Syndrome Patient. Int J Oral Dent Health 2:030. 10.23937/2469-5734/1510030
Copyright: © 2016 AlAsseri N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Oral and maxillofacial surgery, Noonan syndrome, Pediatric dentistry, Central giant cell lesion, Coincidental finding, Awareness, Dentists
Abbreviations
CGCL: Central Giant Cell Lesion
Introduction
Noonan syndrome is an autosomal dominant condition with variable phenotypic expressions including short stature, distinct craniofacial features, congenital heart anomalies, and developmental delay. The main craniofacial features include hypertelorism with a downward slanting of the palpebral fissures, ptosis, low-set posteriorly rotated ears, deeply grooved philtrum and a high arched palate [1,2]. Furthermore, Noonan syndrome has been found to be closely linked to tumor development such as central giant cell lesions (CGCL) [3,4]. As in this report we present a case of an 11-year-old male diagnosed with Noonan syndrome and during his dental follow-up visit, a multilocular lesion on the right side of his mandible was discovered coincidentally in an orthopantomogram (OPG) and was diagnosed to be central giant cell lesion after the histopathological assessment following surgery.
Case Report
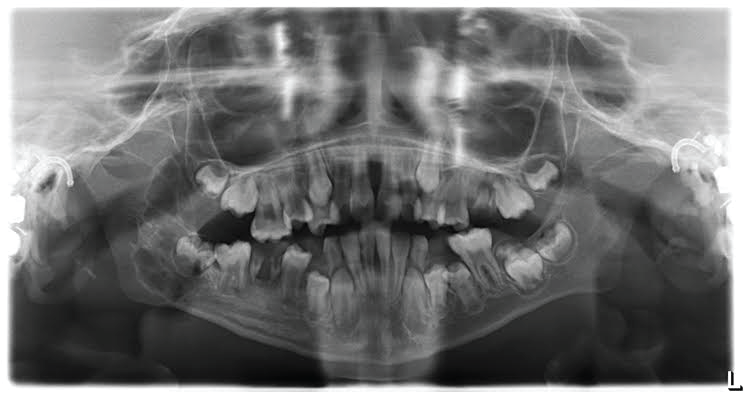
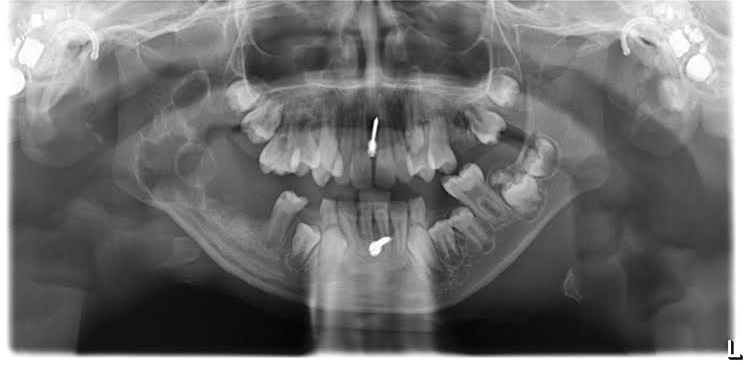
An 11-year-old Saudi male who was referred by a Pedodontist to the Oral and Maxillofacial Surgery Department, Prince Sultan Military Medical City, Riyadh, Saudi Arabia with a right mandibular lesion that was coincidentally found in an orthopantomogram (OPG) (Figure 1) during dental screening.
Medical records of the patient revealed that he was diagnosed with Noonan syndrome by a genetic physician, with a history of atrial septal defect, which was managed conservatively. The patient is also being monitored by an ENT physician for his bilateral syndromic sensorineural hearing loss.
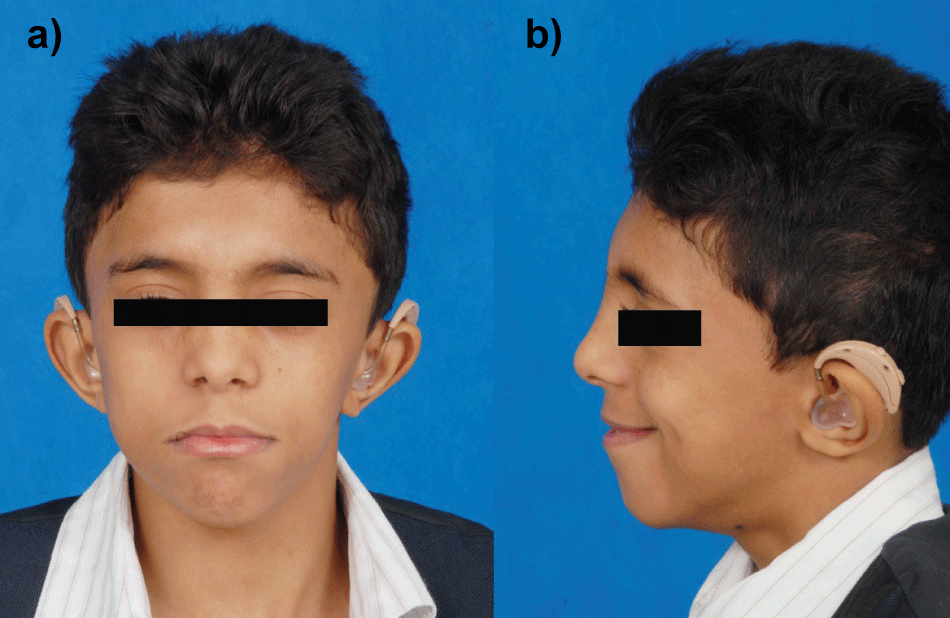
Upon clinical examination: he has short stature, webbed neck, facial asymmetry, deeply grooved philtrum, pectusexcavatum, bilateral hearing aids, in addition to an intellectual disability with articulation difficulties. Intraoral examination showed a high arched palate, poor tongue control, and swelling on the right side of the mandible (Figure 2).
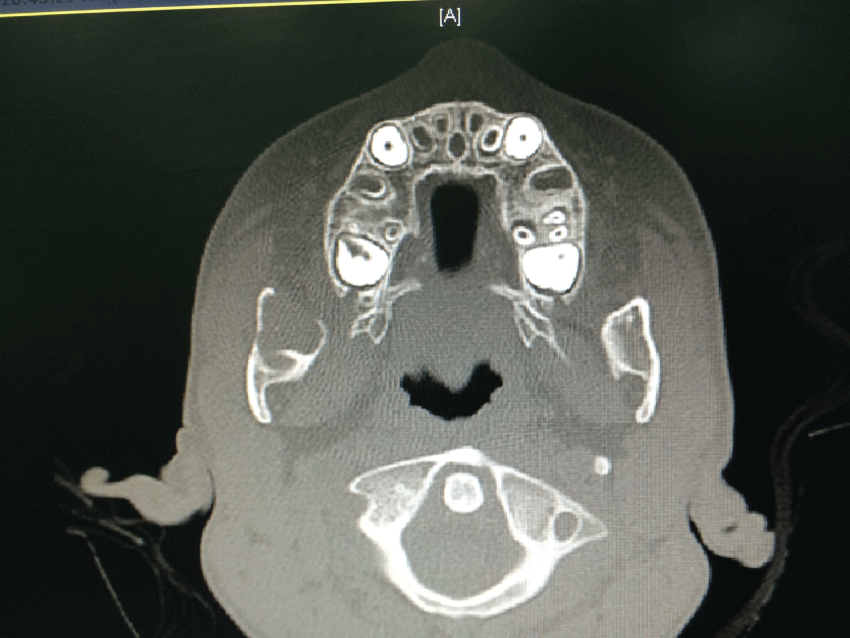
The radiographic interpretation of the OPG showed a multilocular radiolucent lesion involving the right angle of the mandible measuring 3 cm anteroposteriorly (Figure 1). Computed tomography (CT) was requested and reported an expansile lesion within the ramus of the right side of the mandible, causing remodeling and erosion (Figure 3).
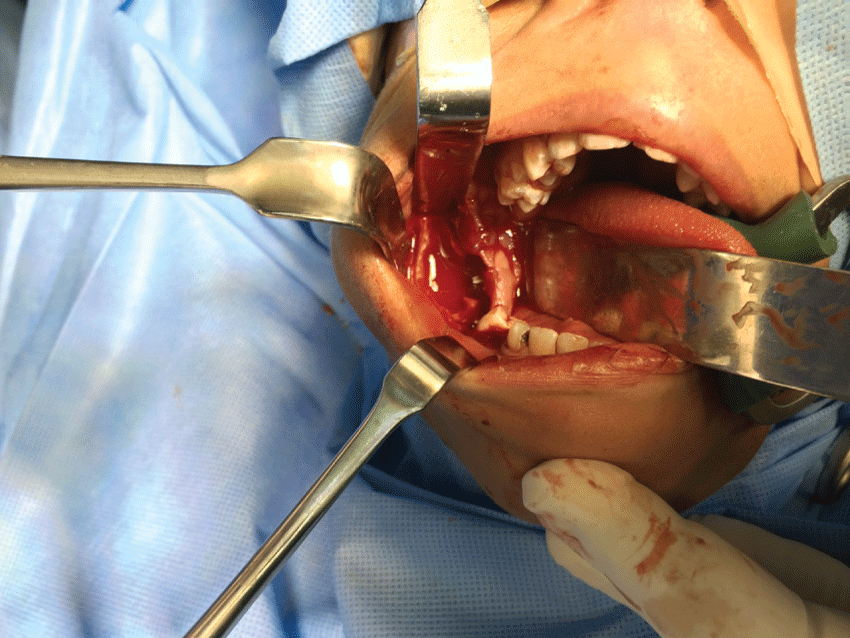
The patient was managed under general anesthesia with surgical excision of the whole lesion and aggressive curettage (Figure 4 and Figure 5). Specimen was sent for histopathological assessment and the result came back as central giant cell lesion. Because of the large defect of the mandible after removal of the lesion, the patient was kept under intermaxillary fixation (IMF) for 3 weeks to prevent the risk of mandibular fracture and promote initial bone healing.
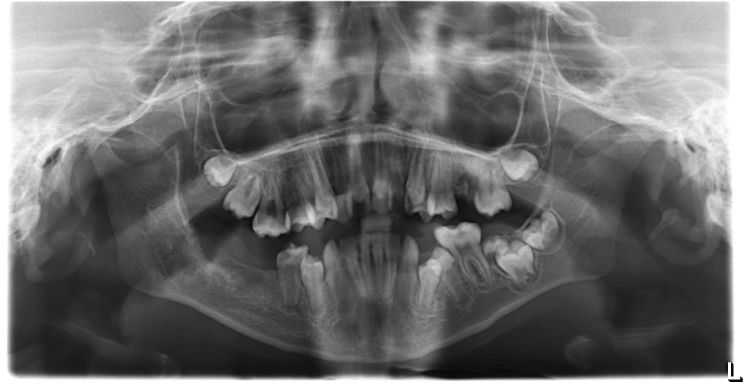
After the surgical intervention, the patient was seen several times in the OMFS clinic for a follow-up. The latest visit was a year later, showed complete soft tissue healing, good bone remodeling and a dramatic reduction in the size of the bony defect (Figure 6).
Discussion
Since the syndrome is of a multisystemic nature and is caused by mutations in the RAS-MAPK signaling pathway; which is linked to tumor development [3,5], it requires a multidisciplinary approach and regular check-ups. Particularly that 11-year-old male patient diagnosed with Noonan Syndrome; was referred to us after a coincidental finding in the OPG of a multilocular lesion on the right side of his mandible during his dental visit. When we revealed the dental history of this patient we found that he was seen several times but no OPG screening radiograph was taken to rule out any bony lesion in such syndrome.
An important part in the management of CGCL is the differential diagnosis, which includes brown tumor of primary or secondary hyperparathyroidism, cherubism, fibrous dysplasia or other types of giant cell lesions [6].
For this reason, the preoperative laboratory test was done for this patient showed within normal calcium, phosphate and parathyroid hormone levels excluding hyperparathyroidism which causes brown tumor and by rule it out from the differential diagnosis.
CGCL and cherubism cannot be differentiated histopathologically. Therefore CGCL can be easily misdiagnosed with cherubism [7]. Recently gene testing for known mutations in the PTPN11 gene [8-10] and the SOS1 gene [11] were found in patients with Noonan syndrome.
Typical radiographic presentation of cherubism is to have the lesion bilaterally of the mandible and/or the maxilla. In this case, the lesion was found on one side of the mandible and size was not large as commonly seen in cherubism [6]. Suggested treatment options include curettage or complete surgical excision. There is also the alternative method of Intralesional injections of steroids, interferons or calcitonin where it should be taken on a daily basis for 6 months in order to shrink the tumor; however, it is not highly recommended owing to its long treatment period [12].
Therefore, our treatment of choice was surgical excision and aggressive curettage, considering the size of the lesion and to avoid further bone resorption. And due to our prior knowledge of the syndrome's relation to giant cell lesions, plus the radiographic and clinical interpretation, the surgical intervention was carried out then followed by the histopathological assessment to confirm the expected diagnoses, which was CGCL.
After undertaking the procedure the patient was placed in intermaxillary fixation for 3 weeks to prevent mandibular fracture. The latest follow-up which was a year after the intervention, showed complete healing of the soft tissue and good bone remodeling.
Conclusion/Recommendations
The reason we reported this case is to raise awareness among dental clinicians regarding the syndrome and its characteristics. The diagnosis of the syndrome tends to be overlooked due to the wide variation of phenotypic expressions; which can be presented in a mild form and age receding facial features [13].
Because Noonan syndrome patients have a high incidence of jaw abnormalities including CGCL, They should be screened clinically and radiographically during the first dental visit to rule out the presence of giant cell lesion that could be misdiagnosed and cause further jaw expansion before it can be detected and treated.
References
-
Ineke Van der Burgt (2007) Noonan Syndrome. Orphanet J Rare Dis 4: 1-6.
-
JE Allanson (1987) Noonan Syndrome. J Med Genet 1: 9.
-
Moskovszky L, Idowu B, Taylor R, Mertens F, Athanasou N, et al. (2013) Analysis of Giant Cell Tumour of Bone Cells for Noonan Syndrome/Cherubism-Related Mutations. J Oral Pathol Med 42: 95-98.
-
Hussain MR, Baig M, Mohamoud HS, Ulhaq Z, Hoessli DC, et al. (2015) BRAF gene: From human cancers to developmental syndromes. Saudi J Biol Sci 22: 359-373.
-
Moos D, Droitcourt C, Rancherevince D, Marec Berard P, Skowron F (2012) Atypical Granular Cell Tumor Occurring in an Individual with Noonan Syndrome Treated with Growth Hormone. Pediatr Dermatol 29: 665-666.
-
Papadaki ME, Lietman SA, Levine MA, Olsen BR, Kaban LB, et al. (2012) Cherubism: best clinical practice. Orphanet J Rare Dis 7: S6.
-
Cohen MM Jr, Gorlin RJ (1991) Noonan-like/multiple giant cell lesion syndrome. Am J Med Genet 40: 159-166
-
Lee SM, Cooper JC (2005) Noonan syndrome with giant cell lesions. Int J Paediatr Dent 15: 140-145.
-
Tartaglia M, Zampino G, Gelb BD (2010) Noonan syndrome: clinical aspects and molecular pathogenesis. Mol Syndromol 1: 2-26.
-
Lee JS, Tartaglia M, Gelb BD, Fridrich K, Sachs S, et al. (2005) Phenotypic and genotypic characterisation of Noonan-like/ multiple giant cell lesion syndrome. J Med Genet 42: e11.
-
Beneteau C, Cavé H, Moncla A, Dorison N, Munnich A, et al. (2009) SOS1 and PTPN11 mutations in five cases of Noonan syndrome with multiple giant cell lesions. Eur J Hum Genet 17: 1216-1221.
-
George D, Bywaters LC (2008) Illustrated Lecture Notes in Oral and Maxillofacial Surgery. Quintessence Publishing Co. Ltd, UK.
-
Roberts AE, Allanson JE, Tartaglia M, Gelb BD (2013) Noonan syndrome. Lancet 381: 333-342.