International Journal of Oral and Dental Health
Treatment of Different Types of Bone Defects with Concentrated Growth Factor: Four Case Reports
Ceren Gökmenoğlu1, Mustafa Cihan Yavuz1, Elif Sadik2, Varol Çanakçi1 and Cankat Kara1*
1Department of Periodontology, Faculty of Dentistry, Ordu University, Ordu, Turkey
2Department of Oral and Maxillofacial Radiology, Faculty of Dentistry, Ordu University, Ordu, Turkey
*Corresponding author:
Cankat Kara, DDS, PhD, Associate Professor, Department of Periodontology, Faculty of Dentistry, Ordu University, 52100 Ordu, Turkey, Tel: +90 452 212 1283, E-mail: mcankat@hotmail.com
Int J Oral Dent Health, IJODH-2-029, (Volume 2, Issue 2), Case Series; ISSN: 2469-5734
Received: March 20, 2016 | Accepted: June 01, 2016 | Published: June 03, 2016
Citation: Gökmenoğlu C, Yavuz MC, Sadik E, Çanakçi V, Kara C (2016) Treatment of Different Types of Bone Defects with Concentrated Growth Factor: Four Case Reports. Int J Oral Dent Health 2:029. 10.23937/2469-5734/1510029
Copyright: © 2016 Gökmenoğlu C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Various techniques have been attempted in the past to truly regenerate the lost bone structures. Owing to its stimulatory effect on angiogenesis and epithelialization, concentrated growth factor (CGF) is an excellent material for enhancing bone healing. The purpose of these case series is to document the beneficial role of CGF in the healing of different bone defect areas. This report describes four female patients presented with (lateral cyst on tooth 22; periimplantitis on mandibular left first premolar area; furcation lesion on tooth 36, periapical abcess on teeth 11-21). Mucoperiosteal full-thickness flaps were elevated to perform the surgical debridement of lesion areas in all patients. Then, minced CGF mixed with bone graft was applied to fill the defects. Recall radiographs of cases showed satisfactory healing of the bony defects. According to the results obtained in these case reports, the positive clinical impact of additional application of CGF with bone graft materials in treatment of bone defects is based on significant radiographic defect bone fill.
Keywords
Concentrated growth factor, Bone defect, Oral surgery
Introduction
The bony defects can be developed by periodontal disease, tooth loss, trauma and infection. The aim of periodontal therapy is to eliminate inflammatory process, prevent the progression of periodontal disease and also to regenerate the lost periodontal tissues [1]. Various techniques and biomaterials have been attempted in the past to truly regenerate the lost bone structures in addition to autogenous and allogenic bone grafts, but complete and predictable reconstruction of periodontal tissues is still difficult to obtain.
Owing to its stimulatory effect on angiogenesis and epithelialization, concentrated growth factor (CGF) is an excellent material for enhancing bone healing. The use of CGF may be a simple and effective method of improving the healing of the bone defects. Its advantages over platelet-rich plasma include ease of preparation, ease of application, minimal expense, and lack of biochemical modification (no bovine thrombin or anticoagulant is required). It can also serve as a resorbable interpositional membrane [2,3]. The CGF layer avoids early invagination of the gingival epithelium, thereby serving as a barrier to epithelium migration [4]. The purpose of these case series is to document the beneficial role of CGF mixed with bone graft and CGF barrier membrane to accelerate bone formation in the healing of different bone defect areas.
Case Report
This report describes four female patients presented with (lateral cyst on tooth 22; periimplantitis on mandibular left first premolar area; furcation lesion on tooth 36, periapical abcess on teeth 11-21) (Table 1). All patients were informed about the treatment procedures, and were required to fill out a consent form and a medical history questionnaire. The current article was conducted in full accordance with the World Medical Association Declaration of Helsinki. Before the surgical procedures, patients were underwent oral hygiene instructions and scaling with ultrasonic and hand instruments to remove any local irritating factors that may have been responsible for the gingival inflammation at the surgical wound healing duration. In the surgery processes, mucoperiosteal full-thickness flaps were elevated to perform the surgical debridement of lesion areas in all patients under local anesthesia (articain 4% with 1:200.000 epinephrine†). 10 cc venous blood was taken to prepare 2 pieces of CGF with special centrifuge‡. One piece was mixed with 0.25-0.5 gr сancellous-cortical heterologous bone mix§ to accelerate new bone formation and the other was used as a barrier membrane. After complete removal of granulation tissue at lesion areas, minced CGF mixed with bone graft was gently tapped inside the osteotomy site with osteotomes and mallet to fill the defects. CGF barrier was covered on the graft to accelerate new bone formation and prevent soft tissue ingrowth (Figure 1). After repositioning the soft tissues, primary closure was attained using 4-0 silk suture. Analgesics and antibiotics were prescribed, and the patient was advised to use chlorhexidine mouthwash for a week.
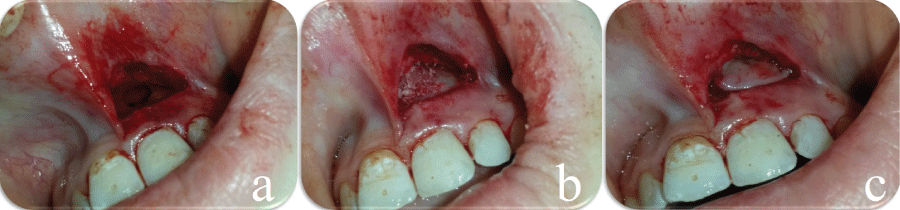
.
Figure 1: a) Intra-oral view of the lesion after removal of granulation tissue; b) Applying CGF mixed with bone graft; c) CGF barrier membrane.
View Figure 1
![]()
Table 1: Demographic details and clinical dental variables in patients.
View Table 1
At 1-week post-surgical follow-up the sutures were removed. The patients experienced mild postoperative pain and swelling. There were no significant complications, such as bleeding, severe pain, or delayed wound healing. Clinical healing was uneventful, with neither infectious episodes nor untoward clinical symptoms. Recall radiographs of cases at 3 months showed satisfactory healing of the bony defects (Figure 2). The patients were also satisfied with outcomes of the treatments.
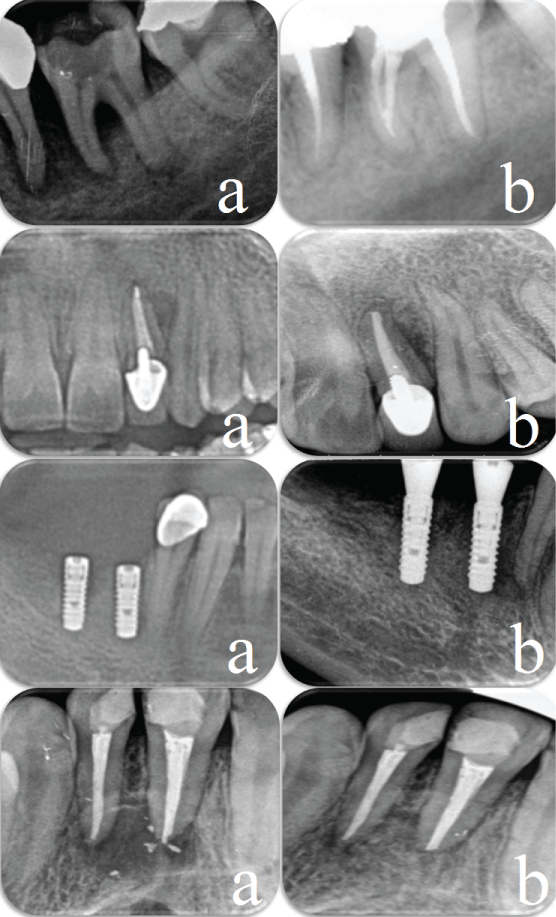
.
Figure 2: a) Pretreatment radiographic views showing bone defects; b) Periapical radiographic view 3 months after surgery showing evidence of bone healing.
View Figure 2
Discussion
Bony defects in the jaws occur for various reasons such as infections, periodontal diseases, tumors, or cysts. Each year a large number of teeth were extracted due to the bone defects.
† Ultracain D-S Forte; Aventis Pharma
‡ Medifuge, Silfradent srl, Sofia, Italy
§ OsteoBiol® Gen Os, Italy
Only a few of them are treated with bone regeneration materials. Therefore, bone regeneration becomes an important tool to prevent the loss of jaw bone, more generally, for all types of bone defects. From past to present many types bone graft materials and patterns of use were investigated in order to find better and inexpensive method for filling bony defects [5]. One of the recent approaches is to ameliorate the bone graft healing by growth factor enhancement. Growth factors are bioactive proteins which control the process of wound healing [6]. These growth factors are present in blood, within platelets and in plasma and platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and recently concentrated growth factor (CGF) have been used for reconstruction of bony defects and increasing the success rate of bone grafting therapy [7,8]. CGF is in the form of platelet gel and can be used in conjunction with bone grafts, which offers several advantages including promoting wound healing, bone growth and maturation, graft stabilization, hemostasis and improving the handling properties of graft materials [8]. The ability of CGF in accelerating bone healing has stimulated the research of its clinical applications in various areas of oral and maxillofacial surgery, which includes healing of extraction sockets including impacted tooth, implantology, ulcer management, and osteonecrosis of the jaw [9]. Therefore, in the present clinical cases, in the treatment of bone defects due to different reasons, the choice was to use CGF mixed with bone graft material, mainly because its biocompatibility, resilience, and availability. As a result, treatment of the bone defects with flap surgery plus CGF and bone mix provided significant defect fill as demonstrated by clinical and radiographic examinations at one-month follow-up.
A clinical trial suggested that the combination of bone graft along with the growth factors in the platelet-rich fibrin (PRF) may be suitable to enhance the bone density [10]. Mazor et al. [11] stated that the use of PRF as the filling material during sinus lift and implantation procedure had stabilized a good amount of regenerated bone in the subsinus cavity. Combined use of PRF and bone graft with good results has also been reported for combined periodontic-endodontic furcation defect [12]. Chang et al. [13] reported that infrabony defects exhibited pocket reduction and clinical attachment gain after six months after PRF application by increasing extracellular signal-regulated protein kinase phosphorylation and osteoprotegerin in periodontal ligament fibroblasts and up-regulation of alkaline phosphatase activity. Our results suggested that the growth factors in CGF had large effects on nearly every stage of bone graft healing.
PRF can serve as a resorbable membrane, which can be used in oral surgery to cover bone augmentation site, it is much less expensive than commercial membrane [10]. Since the surface of PRF membrane is smoother, it can cause superior proliferation of human periosteal cells thereby enhancing bone regeneration [14]. Shivashankar et al. [15] showed rapid rate of bone formation in the treatment of large periapical lesion by using PRF barrier membrane. In the present cases, CGF barrier was used with the goals of promoting bone regeneration and soft tissue healing improving bonding between bone and bone graft.
According to the results obtained in these case reports, the positive clinical impact of additional application of CGF with bone graft materials in treatment of bone defects is based on significant radiographic defect bone fill. CGF barrier is effective to regenerate bone formation associated with GBR and GTR procedure. In addition, the mixture of CGF and bone graft could improve healing compared to conventional GBR procedure. We also prepare a prospective trial comparing different techniques such as PRP, PRF, CGF, GBR.
References
-
Preeja C, Arun S (2014) Platelet-rich fibrin: Its role in periodontal regeneration. Saudi J Dent Res 5: 117-122.
-
Gupta S, Banthia R, Singh P, Banthia P, Raje S, et al. (2015) Clinical evaluation and comparison of the efficacy of coronally advanced flap alone and in combination with platelet rich fibrin membrane in the treatment of Miller Class I and II gingival recessions. Contemp Clin Dent 6: 153-160.
-
Sharma A, Pradeep AR (2011) Autologous platelet-rich fibrin in the treatment of mandibular degree II furcation defects: a randomized clinical trial. J Periodontol 82: 1396-1403.
-
Tanya J, Thomas BS (2012) Platelet rich fibrin membrane for recession coverage. e-Journal of Dentistry 2: 223-227
-
Palti A, Hoch T (2002) A concept for the treatment of various dental bone defects. Implant Dent 11: 73-78.
-
Clark RA (2001) Fibrin and wound healing. Ann N Y Acad Sci 936: 355-367.
-
Bhanot S, Alex JC (2002) Current applications of platelet gels in facial plastic surgery. Facial Plast Surg 18: 27-33.
-
Kim TH, Kim SH, Sándor GK, Kim YD (2014) Comparison of platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and concentrated growth factor (CGF) in rabbit-skull defect healing. Arch Oral Biol 59: 550-558.
-
Pal US, Mohammad S, Singh RK, Das S, Singh N, et al. (2012) Platelet-rich growth factor in oral and maxillofacial surgery. Natl J Maxillofac Surg 3: 118-123.
-
Elgendy EA, Abo Shady TE (2015) Clinical and radiographic evaluation of nanocrystalline hydroxyapatite with or without platelet-rich fibrin membrane in the treatment of periodontal intrabony defects. J Indian Soc Periodontol 19: 61-65.
-
Mazor Z, Horowitz RA, Del Corso M, Prasad HS, Rohrer MD, et al. (2009) Sinus floor augmentation with simultaneous implant placement using Choukroun's platelet-rich fibrin as the sole grafting material: a radiologic and histologic study at 6 months. J Periodontol 80: 2056-2064.
-
Kanakamedala A, Ari G, Sudhakar U, Vijayalakshmi R, Ramakrishana T, et al. (2009) Treatment of a furcation defect with a combination of platelet rich fibrin and bone graft-A case report. ENDO 3: 127-135.
-
Chang YC, Zhao JH (2011) Effects of platelet-rich fibrin on human periodontal ligament fibroblasts and application for periodontal infrabony defects. Aust Dent J 56: 365-371.
-
Gassling V, Douglas T, Warnke PH, Açil Y, Wiltfang J, et al. (2010) Platelet-rich fibrin membranes as scaffolds for periosteal tissue engineering. Clin Oral Implants Res 21: 543-549.
-
Shivashankar VY, Johns DA, Vidyanath S, Sam G (2013) Combination of platelet rich fibrin, hydroxyapatite and PRF membrane in the management of large inflammatory periapical lesion. J Conserv Dent 16: 261-264.





