International Journal of Oral and Dental Health
Prevalence of Taurodontism in Mandibular Molars among Patients at a Dental Care Institution in Nigeria
Yemitan Tolulase Abosede* and Adediran Virginia Efunyemi
Department of Child Dental Health, Faculty of Dentistry, Lagos State University College of Medicine, Nigeria
*Corresponding author: Yemitan Tolulase Abosede, Department of Child Dental Health, Faculty of Dentistry, Lagos State University College of Medicine, Ikeja, Lagos, Lagos, Nigeria, E-mail: tolulaseyemitan@yahoo.com
Int J Oral Dent Health, IJODH-1-020, (Volume 1, Issue 4), Original Research Article; ISSN: 2469-5734
Received: October 12, 2015 | Accepted: November 16, 2015 | Published: November 18, 2015
Citation: Yemitan TA, Adediran VE (2015) Prevalence of Taurodontism in Mandibular Molars among Patients at a Dental Care Institution in Nigeria. Int J Oral Dent Health 1:020. 10.23937/2469-5734/1510020
Copyright: © 2015 Yemitan TA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: Taurodontism involves enlargement of the body of the tooth and shortening of the roots. This anomaly could be an isolated trait, or could be in association with other anomalies or may be a component of a syndrome. This study aims to determine the prevalence of taurodontism in mandibular molars of a Nigerian sample. The objectives are to test the hypotheses that there is no gender preference in taurodontism, and that there is no preference for either left or right side of the mandible.
Materials and Methods: A biometric method was used to assess taurodontism in mandibular molars from orthopantomograms obtained from dental patients in a specialist clinic in Lagos, Nigeria, between December 2013 and September 2014.
Results: The prevalence of taurodontism was 33% for individuals and 19.1% for mandibular molars. There were no statistically significant differences between gender groups. Taurodontism occurred unilaterally with no significant preference for either left (37.5%) or right (31.3%) mandibular side and bilaterally (31.3%). The most prevalent taurodont was hypotaurodonts (14.7%) followed by mesotaurodonts (4%), hypertaurodonts were the least with 0.4%.
Conclusion: The results indicate a moderate possibility of managing patients with taurodontic molars; therefore clinicians should be aware of its associated clinical implications.
Keywords
Dental anatomy, Dental morphology, Dental radiology, Epidemiology, Tooth development, Treatment planning
Introduction
Taurodontism is a developmental anomaly of a tooth characterized by the absence of constriction at the level of the cementoenamel junction (CEJ), with a more apically displaced floor of the pulp chamber and potential for additional root canal [1,2].
Taurodontism may present either as a single trait or as a component of syndromes such as hypophosphatasia, Klienfelter's syndrome, Down's syndrome, ectodermal dysplasia, and tricho-dento-osseous syndrome [3-8]. This anomaly may affect molars and premolars and may be seen in deciduous and permanent teeth [9].
The radiographic appearance of a taurodont is specific and characteristic; therefore the diagnosis and classification of taurodontism have been based on radiographic assessment and varying methods of biometric analysis [10-13].
As reported in the literatures, prevalence of taurodontism varies from one population to another. The frequency of taurodontism in normal Croatian population was 2.65% [14]. That of a group of Jordanian dental patients showed an overall prevalence of 8% [15]. Furthermore, a prevalence of 11.3% was reported for Saudi dental patients, while 5.6% prevalence was reported among Israeli dental patients, compared with 46.4% reported for young adult Chinese and 48% prevalence reported for a Senegalese population [13,16-18].
In view of the marked variation in prevalence reported in different ethnic groups, this study was undertaken to assess the frequency of taurodontism in a sample of Nigerian dental patients, with the objectives of testing the hypotheses of no gender preference in taurodontism, and no preference for either left or right side of the mandible.
Materials and Methods
This study was carried out on 97 randomly selected orthopantomograms of dental patients of both genders who had attended a specialist dental clinic in Lagos, Nigeria between December 2013 and September 2014. This study was carried out with approval from the Research and Ethics committee of the Lagos State University Teaching Hospital.
All selected radiographs are those of Nigerian subjects. The orthopantomograms were taken on Planmeca ProMax (Planmeca USA Inc, IL, USA) at an exposure of 68 KV, 13.0 mA and 15.8 s. Permanent mandibular right and left first and second molars with radiographic evidence of root completion were included in the study. Faulty radiographs with unclear or altered tooth morphology of the teeth were excluded. Permanent first and second molars with carious involvement, attrition, or those with restorations or orthodontic appliances were also excluded. Also excluded were those with co-existing genetic diseases or syndromes.
All radiographs were viewed in a dark room using an X-ray viewer (Slim-Line™ View Box, Select Dental Manufacturing Inc, NY, USA), and read by one experienced examiner (VA).
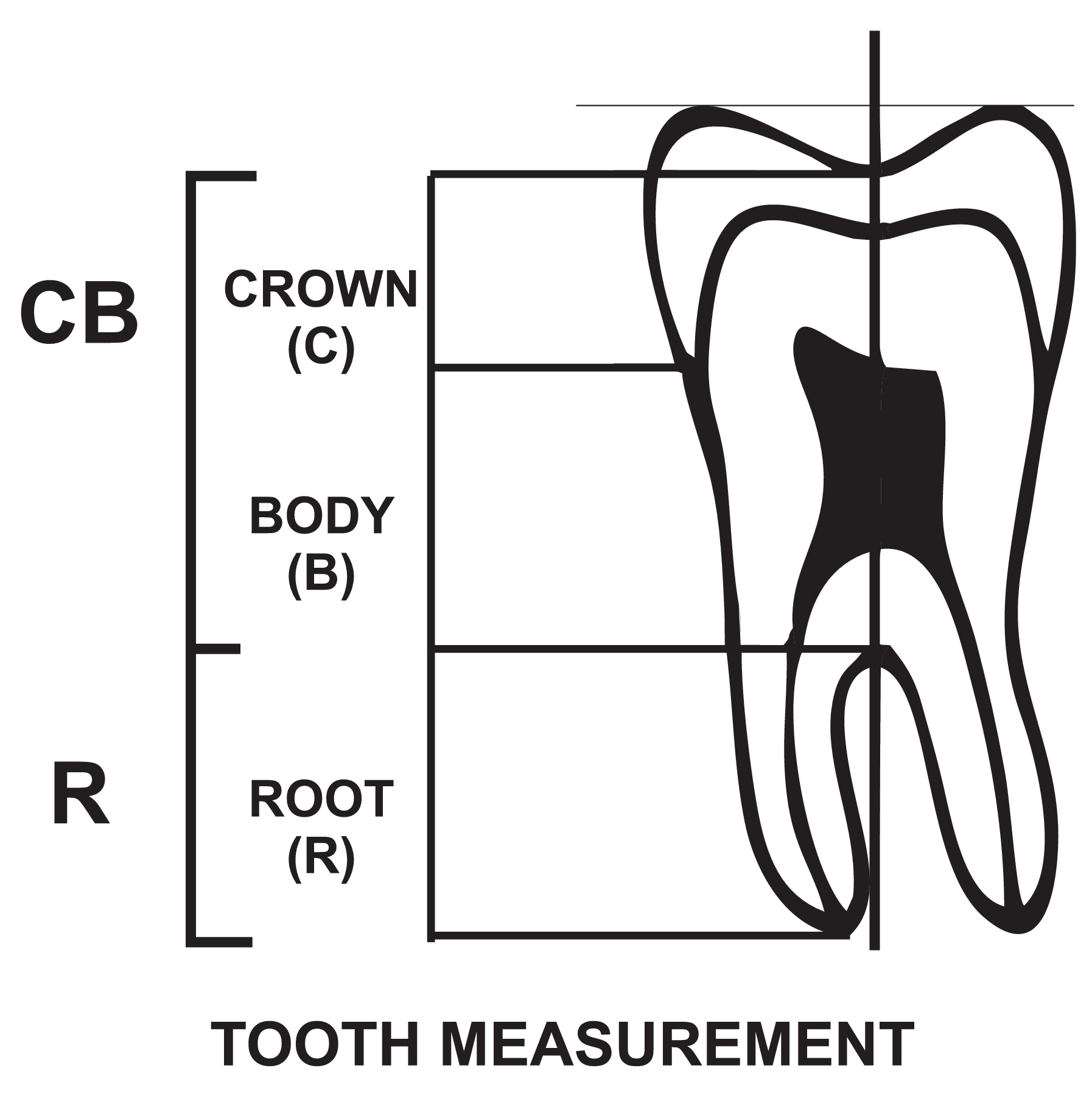
A biometric method used by Seow and Lai [19] was used to measure teeth from panoramic radiographs. The outlines of first and second permanent mandibular molar teeth were traced from the panoramic radiographs unto acetate tracing paper from which measurements were taken. The parts of each tooth were identified using the following landmarks:
Crown (C) - from the deepest part of occlusal surface to the cement-enamel junction (CEJ).
Body (B) - from the CEJ to the root furcation.
Root (R) - from the root furcation to the apex.
Figure 1 shows these anatomical divisions.
The crown and body length (CB) was determined by drawing an occlusal line through the deepest part of occlusal surface which is parallel to another joining the cusp tips. The length CB was determined along a vertical axis drawn at right angle to the occlusal line measured from the deepest part of occlusal surface to the furcation (Figure 1). Similarly, the root length R was determined along the same vertical axis from the furcation to the root apex.
The intra-examiner reproducibility of the measurements was carried out by re-measuring 20 randomly selected orthopanthomograms by the same examiner. Correlation coefficient was used to assess the consistency of values within cases for reproducibility (r = 0.965; P < 0.001).
Teeth with a CB: R ratio of < 1.10 were considered normal (Cynodont), those with ratios of between 1.10 and 1.29 were designated as hypotaurodontic, those with ratio of between 1.30 and 2.00 were grouped as mesotaurodontic, and those with values which were > 2.0 were designated as hypertaurodontic.
Statistical analysis of data was performed using statistical package for social sciences program (SPSS version 17.0, Chicago, IL, USA). Frequency distribution for taurodontism was calculated. The Chi-square tests were used to compare the prevalence of taurodontism between groups. A p - value of less than .05 was deemed statistically significant.
Results
The study group comprised 41 (42.3%) males and 56 (57.7%) females with a mean age of 20.3 (SD 10.8) years and an age range of between 9 and 49 years.
Analysis of the data showed that 32 (33%) of the subjects had one or more teeth that were taurodonts with no significant difference between the two sexes, P > 0.05 (Table 1).
![]()
Table 1: Prevalence of Taurodontism among individuals.
View Table 1
The prevalence of taurodontism among the mandibular molars is shown in table 2.
![]()
Table 2: Prevalence of Taurodontism among mandibular molars
View Table 2
The most prevalent taurodont was hypotaurodonts (14.7%) followed by mesotaurodonts (4%), hypertaurodonts were the least with 0.4% (Table 3).
![]()
Table 3: Prevalence of Taurodontic mandibular permanent molars.
View Table 3
The occurrence of taurodontism in mandibular first and second molars in unilateral and bilateral cases is analyzed in Table 4. As shown, unilateral taurodontism was more prevalent than bilateral taurodontism. There was no significant preference of left (37.5%) or right side (31.3%).
![]()
Table 4: Occurrence of unilateral and bilateral taurodontism in mandibular molars.
View Table 4
Discussion
The results of the present study on a group of Nigerian dental patients have shown an overall prevalence of 33% for individuals (Table 1) and 19.1% for mandibular molars (Table 2). This overall prevalence is lower than reported prevalence of 46.4% for individuals and 21.7% for posterior teeth in a young adult Chinese sample, as well as 48% overall prevalence observed in a Senegalese population [17,18]. However, the overall prevalence of taurodontism in this present study is higher compared to prevalence of 5.6% for individuals and 1.5% for posterior teeth of Israeli dental patients, 2.65% overall prevalence reported for a Croatian sample, prevalence of 8% for individuals and 4.4% for posterior teeth of a group of Jordanian dental patients, prevalence of 11.3% for individuals of Saudi origin, as well as overall prevalence of 12% recorded among Pakistani dental patients [13-16,20].
This study showed no significant gender difference in prevalence of taurodontism (Table 1), even though there was lower prevalence in males (13.4%) than in females (19.6%). Previous studies obtained similar results [13,15,16,20]. Conversely, some results of chromosomal studies on males with an extra X chromosome such as 47XXY males (Klinefelter's syndrome) suggested that X chromosome may contain gene(s) that influence the development of taurodont teeth [21-23]. This however implies a gender preference with a higher frequency of taurodontism in normal females than in their male counterparts. This trend was observed in previous studies [17,20,23].
A possible explanation for the high prevalence of taurodontism in this population may be as a result of metric analysis of the teeth which diagnosed the mildest forms of taurodontism rather than use of visual radiographic assessment. Also, the inclusion of the mandibular second molars may have increased the prevalence as previous studies reported the mandibular second molars the most affected by taurodontism [11,12,24]. It may however be a reflection of ethnic diversity.
Of great interest is the finding that unilateral taurodontism was more prevalent than bilateral taurodontism (Table 4), in contrast to previous findings in which bilateral was more predominant than unilateral taurodontism [13,24]. Previous studies also reported unilateral taurodontism more prevalent than bilateral taurodontism [25,26]. There was no significant preference of the left or right side, although there were more on the left side, but it was not statistically significant. This is in agreement with previous report by Seow and Lai [19].
Taurodontism presents some challenges to the clinical practice of dentistry. One of such challenges is the increased risk of pulp exposure either from dental caries or during dental procedures. In the practice of endodontics, root canal instrumentation and obturation may be difficult as a result of the complexity of the configuration of the root canal of a taurodont. Also, a taurodont may offer reduced stability because of its smaller root surface area if used as an abutment either for prosthetic or orthodontic purposes. Meanwhile, extraction of a taurodont may be less difficult because of its reduced root surface area implanted within the alveolus, especially if the roots are not greatly divergent [3].
The periodontal treatment of a taurodont may offer good prognosis because the progression of periodontal disease to furcation involvement in a taurodont may be slower than in normal teeth as greater periodontal loss is expected in a taurodont before the furcation area is affected [27].
The mandibular molars were selected for measurement because their entire outline is usually obvious clearly on the orthopantomogram. This is in contrast to the maxillary molars where the root apices are usually overshadowed by the zygomatic bone, making it difficult to assess the root length.
The exclusion of the maxillary molars and the use of a two-dimensional radiographic method which can view tooth only in mesiodistal plane may have led to shortcomings in this study. Further study could do a volumetric assessment using a three-dimensional radiographic method such as cone beam computed tomography to overcome these limitations.
In conclusion, this study revealed a 33% incidence of taurodontism in a Nigerian sample with no gender predilection and no preference for either side of the mandible.
Acknowledgements
The authors hereby acknowledge the contribution of Mr. Debo Adediran towards the development of the figure.
The study was funded by the authors.
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
References
-
Jafarzadeh H, Azarpazhooh A, Mayhall JT (2008) Taurodontism: a review of the condition and endodontic treatment challenges. Int Endod J 41: 375-388.
-
Manjunatha BS, Kovvuru SK (2010) Taurodontism - A Review on its etiology, prevalence and clinical considerations. J Clin Exp Dent 2: 187-190.
-
Durr DP, Campos CA, Ayers CS (1980) Clinical significance of taurodontism. J Am Dent Assoc 100: 378-381.
-
Houpt MI, Kenny FM, Listgarten M (1970) Hypophosphatasia: case reports. ASDC J Dent Child 37: 126-137.
-
Keeler C (1973) Taurodont molars and shovel incisors in Klinefelter's syndrome. J Hered 64: 234-236.
-
Jaspers MT (1981) Taurodontism in the Down syndrome. Oral Surg Oral Med Oral Pathol 51: 632-636.
-
Stenvik A, Zachrisson BU, Svatun B (1972) Taurodontism and concomitant hypodontia in siblings. Oral Surg Oral Med Oral Pathol 33: 841-845.
-
Lichtenstein J, Warson R, Jorgenson R, Dorst JP, McKusick VA (1972) The tricho-dento-osseous (TDO) syndrome. Am J Hum Genet 24: 569-582.
-
Cichon JC, Pack RS (1985) Taurodontism: review of literature and report of case. J Am Dent Assoc 111: 453-455.
-
Shaw JC (1928) Taurodont Teeth in South African Races. J Anat 62: 476-498.
-
Keene HJ (1966) A morphologic and biometric study of taurodontism in a contemporary population. Am J Phys Anthropol 25: 208-209.
-
Blumberg JE, Hylander WL, Goepp RA (1971) Taurodontism: a biometric study. Am J Phys Anthropol 34: 243-255.
-
Shifman A, Chanannel I (1978) Prevalence of taurodontism found in radiographic dental examination of 1,200 young adult Israeli patients. Community Dent Oral Epidemiol 6: 200-203.
-
Hrvoje Brkic, Ivana Bagic, Denis Vojvodic (1992) The Prevalence of Taurodontism in Croatian Population. Acta Stomatologica Croatica 26: 79-84.
-
Darwazeh AM, Hamasha AA, Pillai K (1998) Prevalence of taurodontism in Jordanian dental patients. Dentomaxillofac Radiol 27: 163-165.
-
Ruprecht A, Batniji S, el-Neweihi E (1987) The incidence of taurodontism in dental patients. Oral Surg Oral Med Oral Pathol 63: 743-747.
-
MacDonald-Jankowski DS1, Li TT (1993) Taurodontism in a young adult Chinese population. Dentomaxillofac Radiol 22: 140-144.
-
Toure B, Kane AW, Sarr M, Wone MM, Fall F (200) Prevalence of Taurodontism at the level of the molar in the black Senegalese population 15-19 years of age. Odontostomatol Trop 23: 36-39.
-
Seow WK, Lai PY (1989) Association of taurodontism with hypodontia: a controlled study. Pediatr Dent 11: 214-219.
-
Munir MB, Sajjad T, Sajid M (2013) Prevalence of taurodontism in mandibular second molars. Pakistan Oral Dent J 33: 528-530.
-
Komatz Y, Tomoyoshi T, Yoshida O, Fujimoto A, Yoshitake K (1978) Taurodontism and Klinefelter's syndrome. J Med Genet 15: 452-454.
-
Jaspers MT, Witkop CJ Jr (1980) Taurodontism, an isolated trait associated with syndromes and X-chromosomal aneuploidy. Am J Hum Genet 32: 396-413.
-
Varrela J, Alvesalo L (1988) Taurodontism in 47, XXY males: an effect of the extra X chromosome on root development. J Dent Res 67: 501-502.
-
Shah D, Garcha V, Garde J, Ekhande D (2015) Prevalence of taurodontism among the patients visiting a dental teaching Hospital in Pune, India: A retrospective orthopantomogram study. J Indian Assoc Public Health Dent 13: 83-86.
-
Pillai KG, Scipio JE, Nayar K, Louis N (2007) Prevalence of taurodontism in premolars among patients at a tertiary care institution in Trinidad. West Indian Med J 56: 368-371.
-
Bronoosh P, Haghnegahdar A, Dehbozorgi M (2012) Prevalence of taurodontism in premolars and molars in the South of iran. J Dent Res Dent Clin Dent Prospects 6: 21-24.
-
Shifman A, Buchner A (1976) Taurodontism. Report of sixteen cases in Israel. Oral Surg Oral Med Oral Pathol 41: 400-405.