International Journal of Ophthalmology and Clinical Research
New Diagnostic Criteria for Infantile Nystagmus. An Upgraded Nystagmus Clinical Approach
Nadim Nasser*
Department of Pediatrics, Clalit Health Organization services, Israel
*Corresponding author: Nadim Nasser, Department of Pediatrics, Clalit Health Organization services, Kofr Smei', (Kisra-Smei'), m. box: 201, area code 20138, Israel, Tel: 0144-9978585, +972 523 743 134, Fax: 01449570127, +01446882320, E-mail: nasser_nadim9@hotmail.com
Int J Ophthalmol Clin Res, IJOCR-2-042, (Volume 2, Issue 6), Original Article; ISSN: 2378-346X
Received: October 17, 2015 | Accepted: November 24, 2015 | Published: November 26, 2015
Citation: Nasser N (2015) New Diagnostic Criteria for Infantile Nystagmus. An Upgraded Nystagmus Clinical Approach. Int J Ophthalmol Clin Res 2:042. 10.23937/2378-346X/1410042
Copyright: © 2015 Nasser N. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: CEMAS group has classified nystagmus comprehensively in 2001. From that time, attempts to make the subject as uniform as possible, needed continuous upgrading. This manuscript is an upgraded clinical approach for diagnosis of Irreversible Congenital Nystagmus, which is in addition to its being one of the major clinical features of intrinsic ocular diseases, is also a sign of inborn errors of myelination.
Design: We will accompany the way to the diagnosis of congenital irreversible nystagmus of non-intrinsic eye disease origin, by highlighting all the symptoms and signs, which lead us to ascertain the exact etiologies, despite the important past classifications of nystagmus.
Methods: We reviewed the appropriate relevant medical literature and looked for all the diseases, syndromes and triads in which the congenital nystagmus is one of their main components. We brought here in addition, a case report of an infant suffering from nystagmus for further refreshment.
Results: In addition to its being a feature of several pediatric ophthalmic syndromes, congenital irreversible nystagmus (ICN) is also one of the exclusively important features in several non-ophthalmic diseases, but not necessarily the first to appear. It is a main component for the diagnosis of Multiple Sclerosis, as well as other Leukodystrophies diagnosis, and to a lesser frequency in other syndromes.
Conclusions: We recommend for Ophthalmologists to use the policy of "wait and see", in infants complaining of irreversible nystagmus, which is not caused by local ophthalmic diseases. The appearance of characteristics, such as deafness, axial hypotonia, tremor and staccato speech and so on, in parallel with hyperreflexia, is the core of our upgraded flow chart of nystagmus clinical approach.
Keywords
Triads in Ophthalmology, Tremor, Interrupted speech, Hypotonia and nystagmus, BERA test, Leukodystrophies triads, Leukoencephalopathy, Myelin, MNN Triad, Hypo-myelination, MS
Abbreviations
CNS: Central Nervous System, ICN: Irreversible Congenital Nystagmus, ABR: Auditory Brainstem Responses, BERA: Brainstem Evoked Response Audiometry
Introduction
Noticeable 'Congenital nystagmus', or 'infantile irreversible congenital nystagmus' (ICN), continues to be a broad and incompletely defined subject at all its supposed aspects [1]. The reason, apparently, is that previous proposed classifications for nystagmus diagnosis so far, are not enough to cover the whole of the existing etiologies. This demonstrates the need to upgrade protocols for the diagnosis of this medical issue.
The National Eye Institute's Classification of Eye Movement Abnormalities and Strabismus (CEMAS) was published in 2001, based on diagnosing nystagmus according to eye movements' recordings. This classification came after a long period of absence of unifying paradigm and lack of cross-disciplinary agreements about the definitions and about the variety of disorders, which might abbreviate the way to diagnose the etiology of nystagmus [1].
Sometimes, nystagmus could pass unnoticed until school age, and even until adult life [2,3]. In the other hand, Jayalakshmi et al. in a prospective study of fifty-two patients with infantile nystagmus published 1970; made the interesting observation that congenital nystagmus disappeared in approximately half the patients by five years of age, without mention of its cause [4].
Up to now, there are a number of facts pertaining congenital nystagmus: its overall incidence, prevalence of its various forms, characteristics, known causes and its prognosis, which we will list, as a preliminary step for discussing the upgraded approach for finding its etiology.
The most common form of congenital nystagmus, incidence per 10,000 infants, was the neurologic nystagmus (equals 6.8). Other forms of nystagmus associated with congenital cataracts and retinal diseases, were less incident (equals 4.2, 3.4 in the order) [5].
Intrinsic ocular diseases, which cause irreversible congenital nystagmus, are mostly bilateral (as shown in Table 1). The main causes are congenital cataracts, some forms of strabismus, aniridia, retinal coloboma, achromatopsia and foveal hypoplasia. Exceptionally, is the congenital nystagmus due to congenital cataract that can be reversible if surgical repair is early and successful [6,7].
![]()
Table 1: Presence of nystagmus in Ophthalmologic syndromes and triads
View Table 1
Reversible nystagmus, such as Periodic alternating, could also be caused, though less frequent in childhood, by labyrinths' disease, usage of drug and alcohol, head injuries and vitamin B deficiency and by Spasmus nutans [5,8,9].
Intermittent nystagmus is again a feature in some metabolic diseases like maple syrup urine disease, in chromosomal abnormalities such as Down syndrome [10]. It could also occur as a dominant or X-linked characteristic without obvious intrinsic eye disease [1].
When nystagmus is the only sole existing clinical feature, without evidence of intrinsic ocular diseases, and especially when some specific patterns of neurologic nystagmus, like the rapid pendular congenital nystagmus exist, Ophthalmologists could choose to start investigation directly by magnetic resonance imaging (MRI) of the brain, searching a brain defect [1,5,11-13].
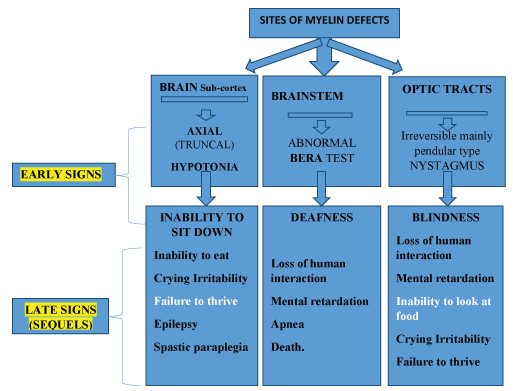
It is known that in certain neurological diseases, nystagmus, including the pendular type, is not necessarily the first sign to appear in the clinical follow up of a patient [14,15], though, it could be one of a variety of combinations of physical characteristics of those diseases, which are not typical to intrinsic ocular disease. Those characteristics, when together, could help locate the site of injury in the central nervous system, both the gray and the white matters (Figure 1).
Generally, congenital eye movement problems are rarely a manifestation of upper motor neuron disease, such as Tay-Sachs, cerebral/cerebellar cortical disease, and etcetera [8,16]. The diagnosis of these disorders is much easier when features as large head circumference, deteriorating mental functions and a macular cherry red spot are available (Table 1).
This paper will highlight the feasibility of bedside clinical diagnosis of one of the rare etiologies of irreversible congenital idiopathic nystagmus, or ICN, especially when it is associated with other major markers of brain white matter disorders [17].
Nystagmus associated with disease of central myelin, subject of this article, which are caused by hypo- or demyelinating of the sensory and motor central nervous system fibers, [14,18-21], are mainly the MS, Pelizaeus-Merzbacher, Cockayne's and Adrenoleukodystrophy peroxisomal disorders, in addition to dozens of hereditary Mitochondrial disorders [22-25].
The mitochondrial disorders have been considered among 'unknown etiologies' or as 'idiopathic' before 2001, until their disclosure during a couple of last decades, mainly by the introduction of MRI and DNA Sequencing and to a lesser extent by clinical bedside diagnosis.
List of the markers of the white matter disorders might be beneficial for our colleagues while dealing with the 'unknown' causes of nystagmus. In the field of ophthalmology, there is an increasing number of syndromes in which irreversible nystagmus is a major clinical marker within a combination of other features, although nystagmus is not always the first sign to appear prior to the other signs. Part of these syndromes are diseases of infancy and childhood.
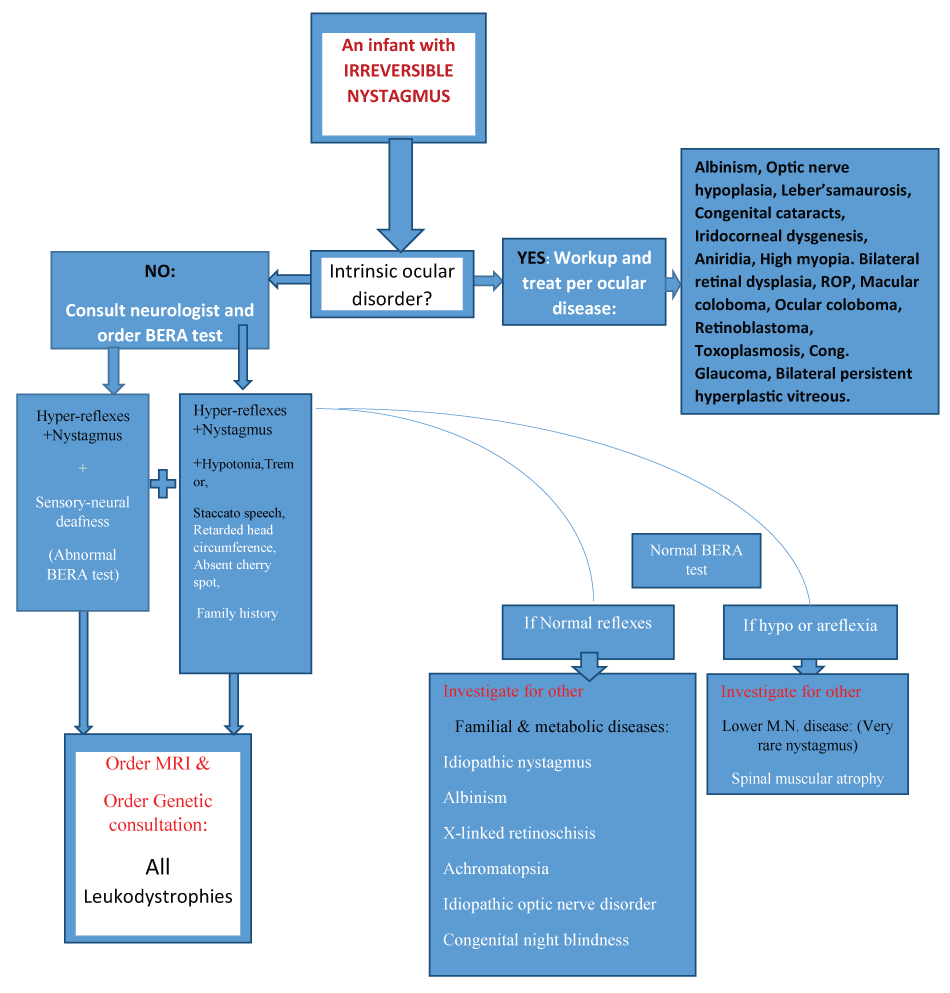
In figure 2, one could see that whenever nystagmus appears in a combination with hypotonia, abnormal BERA test, tremor and speech abnormality, the diagnosis of the white matter disorders, of every related mutation, will be the most probable etiology. Thereafter, the door will be opened for further investigation to prove the genetic defect [26,27].
In other words, in case of ICN, especially in the absence of intrinsic eye disease, Ophthalmologists should "wait and see" if other physical signs appear, which could point for the exact etiology that stands behind the existence of nystagmus.
In figure 1, we could learn not only the markers that together with nystagmus, lead to the diagnosis of myelin disorders, but also shows the systematic sequence of these features, according to the level of the injured site in the brain.
Accordingly, solid evidence supporting the upgraded flowchart for the diagnosis of ICN, according to accompanying clinical signs, will be brought through this manuscript.
Even though methods like MRI and Gene Sequencing for revealing the genetic basis of congenital neurological disorders are time saving and promising, we claim that together, both clinical diagnosis at the sick child bedside and those expensive tools, will enable the building of an upgraded diagram, as an enough useful tool for differential diagnosis of those clinical characteristics.
It is important to diagnose leukodystrophies as the etiology of ICN. These groups of brain disorders are characterized by the degeneration of the brain white matter and have yet a very bad prognosis [28,29].
Questions from families of children suffering from these incurable diseases to physicians who treat their children, whether they have a medication to cure their child, are most frustrating, in time when their child is degenerating in their palms day after day. Physicians usually attempt to reassure the parents that their baby will be fed through a gastric tube, or will be protected from viral diseases, by giving appropriate vaccinations, and by treating every bacterial infections by antibiotics, etcetera, though necessary, are much more annoying for parents than just saying 'no, we have nothing to do'.
Anyway, we have a glimmer of hope in the therapeutic trial research currently underway in the USA, which was reported to have promising results by enhancing complete re-myelination and recovery of previously demyelinated myelin fibers [30]. The contribution of such research for humanity will be in finding an appropriate medication for those dying patients, especially when such therapy be started as soon as possible when the diagnosis of leukodystrophy is at hand through the outstanding sign of early infantile nystagmus. It remains the eternal proverb regarding preventing the birth of sick children, which is better than a pound of cure.
Methods
We reviewed the appropriate and relevant medical literature for the differential diagnosis of congenital nystagmus. All the diseases, syndromes and triads in which the congenital nystagmus is one of their main clinical characteristics are shown in a table 1.
The case report, one of our group of leukodystrophy infants with the same mutation D29G [2], highlights the importance of the policy of "wait and see" for other accompanying signs of infantile nystagmus, especially when nystagmus is the solitary presenting feature in a certain patient. The appearance of further symptoms and signs indicates that injuries had occurred to more than one organ in the brain, leading to a combination of more signs together, as an enough sensitive and specific factual pointing to the etiology of nystagmus.
Results
Table 1 Syndromes and triads with nystagmus.
.
Figure 1: The Symptoms and Signs in patients with leukodystrophy may present (According to level of injury)
View Figure 1
A patient case report
N. M a four months-old female, was born weighing 2600 g, after a full term uncomplicated pregnancy. Her Apgar score was 9/10 and she had mild physiologic jaundice with maximal total bilirubin level 12.2 mg percentage. Screening test for auditory brainstem responses (ABR) at birth was abnormal in the right side.
At this early age, her mother disclosed "something is going wrong with my baby: her meals necessitated longer times and she had probably a difficulty to move the head from side to side". N. M was the fifth child of consanguineous parents, fully immunized, carriers of the mutation for sickle cell anemia. Her brother, four years old, suffered from a nephrotic syndrome. The other brother and one sister suffered from mild asthma. Her brothers and sisters had a normal head growth. On physical examination, she was still had adequate weight for her age. She was without any dysmorphic body features, but had uncontrolled head movements and head lag.
Staring eyes, persistent Moro reflex and hyper-reflexes were observed. No enlargement of visceral organs. When she was five months old, brainstem evoked response audiometry (BERA test) was compatible with bilateral abnormal brainstem function. Nystagmus had appeared. It was clear then that we are facing a serious disease. N. M was not the first patient who was diagnosed in our group of patients. It was clear then that she suffered from the same white matter disease and it made sense already to establish the diagnosis soon by magnetic resonance imaging (MRI) of brain. MRI indeed showed diffuse hypo-myelination of the white matter. N.M. was homo-zygotic for the D29G mutation; a certain mitochondrial disorder of leukodystrophy [16].
The meanwhile, the Ophthalmologist confirmed the existence of nystagmus and the non-existence of a macular cherry red spot. He did not find any evidence of intrinsic eye diseases. He ordered genetic and neurologic consultations, but did not devote much time to discuss the differential diagnosis of the gaze abnormality, as he was expected to do.
From here to her death at twenty months of age, a chain of other signs had progressed sometimes simultaneously, or consecutively in the others: The loss of the social interest, absence of smile, failure to thrive, later, at the short run, spastic paraplegia, impossible swallowing, further decay of muscular tone were obvious and respiratory failure ensued.
The above flowchart is an applicable diagram, for clinical identification of a rare etiology of nystagmus, which has not been described in its specific details in previous classifications of infantile nystagmus. Please note that the starting point is [An infant with nystagmus] and the end-result in the chart is leukodystrophy. It is possible that at the end, we will reach a different etiology, depending on which symptoms and signs the patient has, and whether he has a family history of a certain hereditary disorder.
Figure 2: The flowchart.
.
Figure 2: A flowchart for the work-up of an infant with irreversible congenital nystagmus (ICN).
A flowchart for the diagnosis of congenital nystagmus. After ruling out ocular disease or causes of reversible nystagmus such as labyrinthitis, for example, order a neurologic consultation for disclosing truncal hypotonia and other signs, and order a BERA test for disclosing an injury in the brainstem level. The BERA test is an executive marker for upper neuron disease by itself. (U.M.N. = upper motor neuron, L.M.N. = lower motor neuron disease, LD = leukodystrophy or white matter disease). Nadim Nasser, Sept. 2015.
View Figure 2
Discussion
The early manifestation of irreversible congenital nystagmus (ICN) of infancy and childhood could be a challenging issue for diagnosis.
In certain neurological diseases, nystagmus is infrequently the sole manifestation of the disease, but one of a variety of combinations of signs, and not always the first sign that appears. Therefore, previous classifications of nystagmus that do not recognize the entire spectrum of neurological diseases need further upgrading, according to accompanying clinical features.
In any case, ICN, especially the pendulum type, should alert the ophthalmologist for the existence of serious diseases of the central nervous system (CNS), particularly, white matter disorders; the more so when it is accompanied by intention tremor, interrupted or staccato speech, or by truncal hypotonia and sensory-neural deafness, etcetera [31].
We advise to search insistently these accompanying features, and insert them in the flow charts of differential diagnosis of nystagmus.
When a combination of several signs is clear-cut, it becomes directly into a very sensitive and highly specific combination of characteristics that directs us to defects of white matter of the CNS.
It is important to diagnose leukodystrophies as the etiology of ICN. These groups of brain disorders are characterized by the degeneration of the brain white matter and have yet a very bad prognosis, and no cure.
While writing this article, we met with the clinical trial team from the USA, and has the opportunity to discuss the available treatment of children suffering from nystagmus due to white matter disorders. It was pleasant to hear that there is certainly a glimmer of hope in the clinical trial currently underway, when the head of the team reported that they have promising results of enhancing complete re-myelination and recovery of previously demyelinated fibers in lab animals with MS.
We hope that the drug will be approved for treatment in the coming years, including all these disorders including the healing of ICN. The contribution of such research, when it will be available for humanity will be in finding an appropriate medication for those dying patients, especially when such therapy be started as soon as possible.
At last, all accounts should be taken during the search for the causes of nystagmus.
Acknowledgement/Disclosure
Thanks to fortune, nature and children who are part of nature, whom I have the respect and pleasure to care for their health, and who showed me the same things that others saw but could not catch. Thanks to medical student Mr. Minaem Nasser at Sackler medical faculty, in Tel-Aviv University, for his endeavor and his generous contribution for preparing the tables for this article without any cost. His E-mail: spike.2021@hotmail.com
References
-
Gresty M, Page N, Barratt H (1984) The differential diagnosis of congenital nystagmus. J Neurol Neurosurg Psychiatry 47: 936-942.
-
Nasser N (2014) "Mais-Nadim Nasser Triad", a Useful Marker for Leukodystrophies Diagnosis. J Genet Syndr Gene Ther 5: 5.
-
Cogan DG (1967) Congenital nystagmus. Can J Ophthalmol 2: 4-10.
-
Jayalakshmi P, Scott TF, Tucker SH, Schaffer DB (1970) Infantile nystagmus: a prospective study of spasmus nutans, and congenital nystagmus, and unclassified nystagmus of infancy. J Pediatr 77: 177-187.
-
Leigh RJ, Zee DS (2015) The neurology of the eye movements (2015) 5th (edn). Oxford university press, USA.
-
Schiav C, Fresina M (2009) A Brief Review, Eur Ophthalmol Rev 2: 53-54.
-
Godde'-Jolly D (1973) Larmande A: Nystagmus congenital. In: Les nystagmus, vol 1. Paris, Masson.
-
Gottlob I, Zubcov AA, Wizov SS, Reinecke RD (1992) Head nodding is compensatory in spasmus nutans. Ophthalmology 99: 1024-1031.
-
Burke JP, O'Keefe M, Bowell R, Naughten ER (1991) Ophthalmic findings in maple syrup urine disease. Metab Pediatr Syst Ophthalmol (1985) 14: 12-15.
-
Coffeen CM, McKenna CE, Koeppen AH, Plaster NM, Maragakis N, et al. (2000) Genetic localization of an autosomal dominant leukodystrophy mimicking chronic progressive multiple sclerosis to chromosome 5q31. Oxford Journals. Hum Mol Genet 9: 787-793.
-
Weber T, Kohler W (2010) [Demyelinating disorders]. Nervenarzt 81: 471-496.
-
Vanderver A, Tonduti D, Schiffmann R, Schmidt J, van der Knaap MS (2014) Leukodystrophy Overview. GeneReviews.
-
Pouwels PJ, Vanderver A, Bernard G, Wolf NI, Dreha-Kulczewksi SF, et al. (2014) Hypomyelinating leukodystrophies: translational research progress and prospects. Ann Neurol 76: 5-19.
-
Poser CM, Paty DW, Scheinberg L, McDonald WI, Davis FA, et al. (2004) New diagnostic criteria for multiple sclerosis: Guidelines for research protocols. Ann Neurol 13: 227-231.
-
Scott EO, Denis H. Laura P.S (2007) Disorders of the Eye Movement and alignment. In: Nelson textbook of Pediatrics. 18th (edn). 2582, Sounders Elseviers.
-
Magen D, Georgopoulos C, Bross P, Ang D, Segev Y, et al. (2008) Mitochondrial hsp60 chaperonopathy causes an autosomal-recessive neurodegenerative disorder linked to brain hypomyelination and leukodystrophy. Am J Hum Genet 83: 30-42.
-
Neuhann T, Rautenstrauss B (2013) Genetic and phenotypic variability of optic neuropathies. Expert Rev Neurother 13: 357-367.
-
Traboulsi EI (2012) Genetic diseases of the eye. In: Traboulsi EI, (ed). Oxford monographs on medical genetics. Oxford university Press (OUP) USA.
-
Gordon HB, Letsou A, Bonkowsky JL (2014) The leukodystrophies. Semin Neurol 34: 312-320.
-
Perlman SJ, Mar S (2012) Leukodystrophies. Adv Exp Med Biol 724: 154-171.
-
Ahmed RM, Murphy E, Davagnanam I, Parton M, Schott JM, et al. (2014) A practical approach to diagnosing adult onset leukodystrophies. J Neurol Neurosurg Psychiatry 85: 770-781.
-
Parikh S, Bernard G, Leventer RJ, van der Knaap MS, van Hove J, et al. (2015) A clinical approach to the diagnosis of patients with leukodystrophies and genetic leukoencephelopathies. Mol Genet Metab 114: 501-515.
-
Kohlschutter A1, Eichler F (2011) Childhood leukodystrophies: a clinical perspective. Expert Rev Neurother 11: 1485-1496.
-
Hobson GM, Garbern JY (2012) Pelizaeus-Merzbacher disease, Pelizaeus-Merzbacher-like disease 1, and related hypomyelinating disorders. Semin Neurol 32: 62-67.
-
Tanigawa A, Komiyama A, Hasegawa O (1998) Truncal muscle tonus in progressive supranuclear palsy. J Neurol Neurosurg Psychiatry 64: 190-196.
-
Bernot A (2001) Genome Transcriptome and Proteome Analysis, Wiley publishers. Sample book reference: 2001. Volpe JJ. Neurology of the newborn. 4th (edn). Philadelphia: Saunders.
-
Gahl WA, Markello TC, Toro C, Fajardo KF, Sincan M, et al. (2012) The National Institutes of Health Undiagnosed Diseases Program: insights into rare diseases. Genet Med 14: 51-59.
-
Gandevia SC1, Plassman BL (1988) Responses in human intercostal and truncal muscles to motor cortical and spinal stimulation. Respir Physiol 73: 325-337.
-
Ochs R, Markand ON, DeMyer WE (1979) Brainstem auditory evoked responses in leukodystrophies. Neurology 29: 1089-1093.
-
Sha Mi (2015) Blocking LINGO-1 for CNS remyelination and repair: From discovery to clinical trials. J Brain Disord Ther, Biogen.
-
Pouget J (2004) Molecular diagnosis of hereditary neuropathies such as Charcot-Marie-Tooth disease. Rev Neurol (Paris) 160: 181-187.





