International Journal of Neurology and Neurotherapy
The Prevalence of Spasticity in Veterans Living in a Long-Term Care Facility
Maxim Turchan, Taylor S Hudson, Chandler E Gill, Amanda D Currie, Anna L Molinari, Mallory L Hacker, Fenna T Phibbs, Christopher Tolleson, Sarah H Millan, Lauren E Heusinkveld, Candace J Grisham and David Charles*
Department of Neurology, Vanderbilt University Medical Center, Nashville, USA
*Corresponding author:
David Charles, Department of Neurology, Vanderbilt University Medical Center, Nashville, USA, Tel: 615-936-2596, E-mail: David.Charles@Vanderbilt.edu
Int J Neurol Neurother, IJNN-3-056, (Volume 3, Issue 5), Original Research; ISSN: 2378-3001
Received: February 24, 2016 | Accepted: July 31, 2016 | Published: September 01, 2016
Citation: Turchan M, Hudson TS, Gill CE, Currie AD, Molinari AL, et al. (2016) The Prevalence of Spasticity in Veterans Living in a Long-Term Care Facility. Int J Neurol Neurother 3:056. 10.23937/2378-3001/3/5/1056
Copyright: © 2016 Turchan M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To determine the prevalence of spasticity in a population of veterans residing in a long-term care facility. Design: Prospective population study.
Subjects/Patients: Forty-three male veterans of the United States Armed Forces residing in a long-term care facility.
Methods: All facility residents and their medical decision makers (if necessary) were approached for enrollment in this study. After consent was obtained, two neurologists specializing in movement disorders independently conducted a brief physical examination to ascertain the presence or absence of spasticity.
Results: The prevalence of spasticity in veterans residing in this long-term care facility was 33% (14/43). Forty-three percent (6/14) of subjects determined to have spasticity had at least one sign of spasticity documented in their medical record.
Conclusion: This study reports that one-third of veterans residing in a long-term care facility have spasticity. No signs of spasticity were documented in the medical records of 57% (8/14) of the subjects determined to have spasticity during the study, which suggests spasticity is greatly under-diagnosed in this population. These results highlight the need for more detailed observation of signs of spasticity for veterans in long-term care settings.
Keywords
Prevalence, Spasticity, Veterans, Movement disorders
Abbreviations
TBI: Traumatic Brain Injury
Introduction
Spasticity is a clinical manifestation of increased excitability of muscle stretch reflexes and a velocity-dependent increased resistance to passive stretch. The condition frequently causes pain and difficulty with ambulation and limb use [1]. Decreased strength and dexterity as well as abnormal limb positioning associated with spasticity can lead to social embarrassment, stigmatization, and reduced employability [2]. If left untreated, severe spasticity can cause contractures and increase the risk of skin breakdown, pressure ulcers, and urogenital infections [1,3]. These physical, social, and financial ramifications of spasticity significantly impact patients' quality of life [4,5]. When spasticity is effectively treated, there is significant improvement in both active (e.g. ambulation) and passive function (e.g. receiving hygienic care) as well as objective measures such as muscle tone [6-10]. Available therapies, which include oral medications, neurotoxin injections, intrathecal baclofen, and physical and occupational therapy, are safe, readily available, and covered by most public and private health insurance plans. However, despite the efficacy and availability of available therapeutic options, spasticity is frequently left untreated [1].
The incidence of traumatic brain injury (TBI) is high (22%) in soldiers who have returned from the wars in Iraq and Afghanistan [11]. Because typical survivors of combat-related TBI are previously healthy males in their twenties some patients with severe and irreversible TBI require long-term care in skilled nursing facilities, this population is expected to require comprehensive care for many decades [11]. Spasticity treatment is an important component of long-term care for survivors of combat-related TBI because it improves quality of life, restores function, and reduces medical complications [12]. This population study was designed to evaluate the prevalence of spasticity in a group of veterans residing in a long-term care facility in order to inform the provision of long-term care for returning soldiers.
Methods
Study design
This prospective population study was designed to determine the prevalence of spasticity among veterans with skilled nursing needs residing in a state-operated long-term care facility in Tennessee. Approval for the study was obtained from the Vanderbilt Institutional Review Board (IRB #110470, clinicaltrials.gov NCT018779449) and the host facility.
Study population
Eligibility for residence in the host facility was predicated upon the resident's, or their immediate family members', status as a veteran of the United States Armed Forces, their entitlement to medical treatment and/or other benefits from the United States Department of Veterans Affairs, and regional factors including at least one of, but not limited to, the following: 1) being born in the state the host facility is located, 2) being a resident of the state where the host facility is located at the time of admission to the facility, and/or 3) having a family member or legal guardian, residing in the same state as the host facility. There is no minimum amount of disability required for residence in the host facility, and as such the facility residents have a wide variety of medical conditions, both in scope and severity.
Study methods
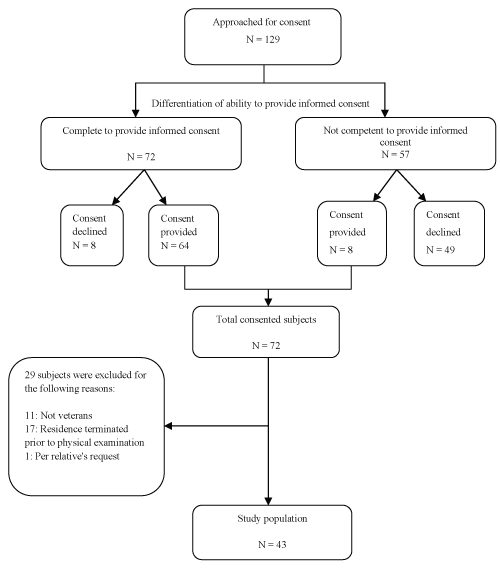
All residents who were deemed competent to make decisions regarding their own treatment by the facility physicians and the study principal investigator (PI) were asked to provide written informed consent to participate in the study. Consent forms were mailed to legally authorized medical decision-makers for residents who were not competent to provide consent. For this analysis, consent was assumed to be declined if the informed consent document was not returned. Figure 1 shows a schematic of the recruitment process.
Two neurologists with movement disorders expertise independently performed a brief physical and neurological examination on each subject. During the examination, the movement disorders specialists were instructed to look for the following cardinal signs of spasticity: 1) spastic postures, 2) increased muscle tone in the setting of weakness in the upper or lower extremities, 3) velocity dependant muscle tone, and 4) hyperreflexia. At the conclusion of the examination, each neurologist recorded their diagnostic impression, indicating whether spasticity was present, absent, or unable to be determined (for example, due to subject non-compliance with the exam). The examinations were conducted independently on the same day. If at least one of the two physicians reached a diagnosis of spasticity, the subject was considered to have spasticity.
To determine the rate at which signs of spasticity were previously detected in the long-term care facility, subjects' charts were reviewed for ICD-9 codes indicative of spasticity (342.2 family: hemiplegia and hemiparesis or 438.2 family: late effects of cerebrovascular disease). Subjects' medical records were also reviewed for therapeutic agents commonly used to treat spasticity (neurotoxin injections, baclofen, cyclobenzaprine, dantrolene, and tizanidine [11,12]).
Statistical analysis
Consent rates between residents who were competent to provide consent and the residents whose proxies were approached for consent on their behalf were compared by a chi-square test of independence. The average age at examination between groups was compared by a Wilcoxon-Mann-Whitney test, and the remaining demographic variables were compared by proportion z-tests.
Results
Enrollment
Subject demographics are shown in table 1. Participants were recruited from May 2013 to March 2014. During this time 129 residents were approached for informed consent. Fifty-six percent (72/129) of residents were deemed capable of providing consent, and of these, 89% (64/72) elected to participate in this study (Figure 1). Forty-four percent (57/129) of the residents required consent from their legal decision-maker and only 14% (8/57) of those residents were included in the study. This represents a significant difference between subjects who self-consented versus requiring surrogate consent (χ2 = 72.28, p < 0.0001). Ultimately 72 subjects provided consent.
![]()
Table 1: Demographics.
View Table 1
Forty percent (29/72) of enrolled subjects were excluded from the final analysis for the following reasons (Figure 1): 59% (17/29) of subjects were no longer facility residents at the time of examination, 38% (11/29) were determined to be non-veterans after review of the medical record, and 4% (1/29) withdrew consent at the request of a relative. Ultimately, 43 veterans (100% male) were included in the final analysis.
Outcomes
Spasticity was detected in 33% (14/43) of residents of this long-term care facility. There were no significant differences in age, ethnicity, or duration of residence between subjects with and without spasticity. Of those determined to have spasticity on exam, 43% (6/14) of subjects had at least one sign of spasticity as defined by the ICD-9 diagnostic codes documented in the medical record (Table 2).
![]()
Table 2: Identification and treatment of spasticity.
View Table 2
Within the 67% (29/43) who did not have spasticity detected by either neurologist upon examination, there was evidence of spasticity recorded in the medical record of one subject. Two subjects were receiving treatment with anti-spasticity medications (baclofen and cyclobenzaprine, respectively) at the time of this study; however, neither of these subjects had a documented sign of spasticity, nor were they found to have spasticity on examination [13-15].
Discussion
In this study, the prevalence of spasticity was 33% among veterans residing in a long-term care facility. A recent study reported 21% of nursing home patients had spasticity [16]. If the prevalence of spasticity in this study is representative of other long-term care facilities, the burden of spasticity in veterans may be equivalent to or exceed that of the general nursing home population. A diagnosis of spasticity was made in 57% of veterans in this study without any prior indications of spasticity documented in their medical records, which suggests this condition is under-diagnosed among veterans. Thus, these patients were not receiving adequate care for spasticity despite the availability of numerous effective and commonly accepted methods of treatment [17,18]. The under-diagnosis and subsequent under-treatment of spasticity represents a critical need for residents of long-term care facilities.
Improving the rate at which spasticity is detected and treated among veterans could improve care for this population by increasing the ease of performing activities of daily living such as maintaining hygiene and increasing the ease of patient transfer from chair to bed and bed to chair. Further, improved detection and treatment of spasticity would lead to an improved quality of life, as the risk of contractures, skin breakdown, pressure ulcers, and urogenital infections would be lessened.
Study Limitations
This is the first study to report the prevalence of spasticity in a population of veterans residing in a long-term care facility. This study is limited by several factors. First, no women were included. This gender discrepancy was anticipated and unavoidable due to the predominantly male demographic of the U.S. military between the 1930s and 1960s [19]. The results of this analysis may not be representative of all long term care facilities for veterans as it was conducted at a single center. We encountered significantly different consent rates between subjects who provided consent on their own behalf and those for whom consent was provided by a medical decision maker. It is unknown if the differing rates of consent affected the results. Considering that TBI is a common cause of both spasticity and cognitive impairment, [20] it is possible that an under-representation of cognitively impaired subjects in our cohort would therefore underestimate the actual prevalence of spasticity in this population. Further investigations into this patient population would benefit from the inclusion of multiple long-term care facilities as well as better representation of those unable to provide consent on their own behalf.
Conclusion
These results highlight the need for more detailed observation of signs of spasticity for veterans living in long-term care settings. This study encourages additional research involving multiple veteran-oriented long-term care facilities to determine whether these findings are generalizable and to explore ways of improving access to spasticity treatment among veterans.
Conflict of Interest Disclosure
Vanderbilt University receives income in excess of $10,000 from grants or contracts from Allergan, Ipsen, Merz, and Medtronic for research or educational programs led by David Charles. David Charles receives income from Allergan, Ipsen, Merz, and Medtronic for education or consulting services. Fenna T Phibbs has done consulting work for Medtronic, Boston Scientific, and St. Jude Medical. Christopher Tolleson receives income from Medtronic for education and consulting services. There are no conflicts of interest for Mallory L Hacker, Amanda D Currie, Taylor S Hudson, Chandler E Gill, Anna L Molinari, Maxim Turchan, Sarah M Millan, Lauren E Heusinkveld, Candace J Grisham.
References
-
Pfister AA, Roberts AG, Taylor HM, Noel-Spaudling S, Damian MM, et al. (2003) Spasticity in adults living in a developmental center. Arch Phys Med Rehabil 84: 1808-1812.
-
Andersson C, Mattsson E (2001) Adults with cerebral palsy: a survey describing problems, needs, and resources, with special emphasis on locomotion. Dev Med Child Neurol 43: 76-82.
-
Albright AL (1996) Spasticity and movement disorders in cerebral palsy. J Child Neurol 11 Suppl 1: S1-4.
-
Rizzo MA, Hadjimichael OC, Preiningerova J, Vollmer TL (2004) Prevalence and treatment of spasticity reported by multiple sclerosis patients. Mult Scler 10: 589-595.
-
Henze T (2007) What is new in symptom management? Int MS J 14: 22-27.
-
Kita M, Goodkin DE (2000) Drugs used to treat spasticity. Drugs 59: 487-495.
-
Beard S, Hunn A, Wight J (2003) Treatments for spasticity and pain in multiple sclerosis: a systematic review. Health Technol Assess 7: 1-111.
-
O'Brien CF (2002) Treatment of spasticity with botulinum toxin. Clin J Pain 18: S182-190.
-
Guillaume D, Van Havenbergh A, Vloeberghs M, Vidal J, Roeste G (2005) A clinical study of intrathecal baclofen using a programmable pump for intractable spasticity. Arch Phys Med Rehabil 86: 2165-2171.
-
Cardoso E, Pedreira G, Prazeres A, Ribeiro N, Melo A (2007) Does botulinum toxin improve the function of the patient with spasticity after stroke? Arq Neuropsiquiatr 65: 592-595.
-
Okie S (2005) Traumatic brain injury in the war zone. N Engl J Med 352: 2043-2047.
-
Graham LA (2013) Management of spasticity revisited. Age Ageing 42: 435-441.
-
Irdesel J, Aydiner SB, Akgoz S (2007) Rehabilitation outcome after traumatic brain injury. Neurocirugia (Astur) 18: 5-15.
-
Kheder A, Nair KP (2012) Spasticity: pathophysiology, evaluation and management. Pract Neurol 12: 289-298.
-
Chou R, Peterson K, Helfand M (2004) Comparative efficacy and safety of skeletal muscle relaxants for spasticity and musculoskeletal conditions: a systematic review. J Pain Symptom Manage 28: 140-175.
-
Chandler E Gill, James Bryant, Philip D Charles, John F Schnelle, Sandra F Simmons (2008) Poster 100: Prevalence and Impact of Spasticity in a Single Nursing Home. Arch Phys Med Rehabil 89: e52.
-
Charles D, Gill CE (2010) Neurotoxin injection for movement disorders. Continuum (Minneap Minn) 16: 131-157.
-
Furr-Stimming E, Boyle AM, Schiess MC (2014) Spasticity and intrathecal baclofen. Semin Neurol 34: 591-596.
-
Segal DR, Segal MW (2004) America's Military Population. Popul Bull 59: 28-32.
-
Eapen BC, Allred DB, O'Rourke J, Cifu DX (2015) Rehabilitation of moderate-to-severe traumatic brain injury. Semin Neurol 35: e1-3.