International Journal of Neurology and Neurotherapy
Endoscopic Biopsy using High-Dose Fluorescein Sodium for Malignant Brain Tumors
Takeshi Okuda1*, Mitsugu Fujita2, Hiromasa Yoshioka1, Takayuki Tasaki1, Shuichi Izumoto1 and Amami Kato1
1Department of Neurosurgery, Kindai University Faculty of Medicine, Osaka-Sayama, Osaka, Japan
2Department of Microbiology, Kindai University Faculty of Medicine, Osaka-Sayama, Osaka, Japan
*Corresponding author:
Takeshi Okuda, Department of Neurosurgery, Kindai University Faculty of Medicine, 377-2 Ohno-Higashi, Osaka-Sayama, Osaka 589-8511, Japan, Tel: +81-72-366-0221, Fax: +81-72-365-6975, E-mail: okuda@med.kindai.ac.jp
Int J Neurol Neurother, IJNN-3-052, (Volume 3, Issue 4), Case Report; ISSN: 2378-3001
Received: May 19, 2016 | Accepted: June 18, 2016 | Published: July 01, 2016
Citation: Okuda T, Fujita M, Yoshioka H, Tasaki T, Izumoto S, et al (2016) Endoscopic Biopsy using High-Dose Fluorescein Sodium for Malignant Brain Tumors. Int J Neurol Neurother 3:052. 10.23937/2378-3001/3/4/1052
Copyright: © 2016 Okuda T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The utility of endoscopic biopsy using high-dose fluorescein sodium in biopsy of malignant brain tumors was investigated. The subjects were 9 patients with malignant brain tumors (malignant lymphoma 5, glioblastoma 3, metastatic brain tumor 1). After making an incision in the dura mater, 20 mg/kg fluorescein sodium were administered intravenously. A transparent sheath was then inserted under neuronavigation guidance. To observe the tumor, an endoscopic biopsy was performed using normal white light endoscopy with a rigid endoscope, without the use of an excitation filter or other special devices. Yellow staining of the tumor was confirmed with white light endoscopy in all cases, and the biopsies could be performed with reliable identification of the target and no complications. Endoscopic biopsy using high-dose fluorescein sodium for malignant brain tumors is a simple and highly useful surgical technique that improves the safety and reliability of biopsies.
Keywords
Endoscopic biopsy, Fluorescein sodium, Malignant brain tumor
Introduction
For some time, the main techniques for biopsy of brain tumors have been open biopsy or stereotactic needle biopsy. With the recent development of endoscopic surgical instruments, many reports on the usefulness of endoscopic biopsy have appeared, and it is reasonable to say that operative procedures for endoscopic biopsy of intraventricular brain tumors have already been established. The usefulness of endoscopic biopsy for intraparenchymal brain tumors has also been reported [1-3]. With endoscopic biopsy for intraventricular brain tumors, it is easy to identify the target intraoperatively because the anatomy within the ventricle can be observed and the tumor confirmed. For intraparenchymal brain tumors, however, a trans cylinder transcortical approach with an inserted sheath is used in the majority of cases, and so there are few anatomical landmarks until the tumor is reached. The target has to be identified within this limited, narrow visual field. Therefore, identification of the target is often done with the use of neuronavigation or stereotactic methods [2,3]. We therefore developed a method of endoscopic biopsy for malignant intraparenchymal brain tumors. With this method, the tumor is stained using fluorescein sodium, and the target can be easily identified, improving the reliability of the biopsy.
Methods
Patients' characteristics
Nine patients with malignant brain tumor who underwent endoscopic biopsy between February 2013 and September 2015 were analyzed. The tumor was malignant lymphoma in 5 patients, glioblastoma in 3 patients, and metastatic brain tumor in 1 patient. Six patients were male and three were female. Their mean age was 67.6 years (range 40-79 years). In all cases, the focus was confirmed with contrast enhancement on contrast-enhanced MRI, and the tumor was located in a deep portion of the brain, a mean of 30 mm (range 23-38 mm) from the brain surface. Patients' details are shown in table 1.
![]()
Table 1: Patients' clinical characteristics.
View Table 1
Operative technique
All surgery was performed under general anesthesia. A single burr hole was made directly on the route that could reach the target in a non-eloquent area in the shortest distance, and then enlarged to a diameter of 2 cm. After making an incision in the dura mater and achieving sufficient hemostasis, 20 mg/kg fluorescein sodium (Alcon Japan, Tokyo, Japan) were administered intravenously. Then, following a wait of 10-15 minutes, corticotomy was performed in the brain surface, and a transparent sheath was inserted under navigation (Brainlab, Feldkirchen, Germany) guidance. The endoscope used was a 0°, 2.7-mm rigid endoscope (EndoArm®, Olympus, Tokyo, Japan). For tumor observation, biopsy was done with regular white light endoscopy without the use of an excitation filter or other special instruments.
Results
Yellow staining of the tumor could be confirmed in all cases, and biopsy could be performed with reliable identification of the target. No difference in the degree of yellow staining was seen between the different types of tumor. There were no complications in any case, the biopsies could be performed accurately and safely, and a definitive pathological diagnosis was obtained. With administration of fluorescein sodium there was yellow coloring of the skin, mucous membranes, urine, and elsewhere, but it disappeared within 24 hours. There were no lasting side effects. Illustrative cases are presented in the following.
Illustrative cases
Case 2 (Figure 1): The patient was a 54-year-old woman diagnosed with primary central nervous system lymphoma. The tumor was present on both sides of the corpus callosum, and the approach was made via the left frontal lobe. The tumor was 32 mm from the brain surface and was confirmed to be colored yellow. Biopsy was performed, and a pathological diagnosis of diffuse large B-cell lymphoma was made.
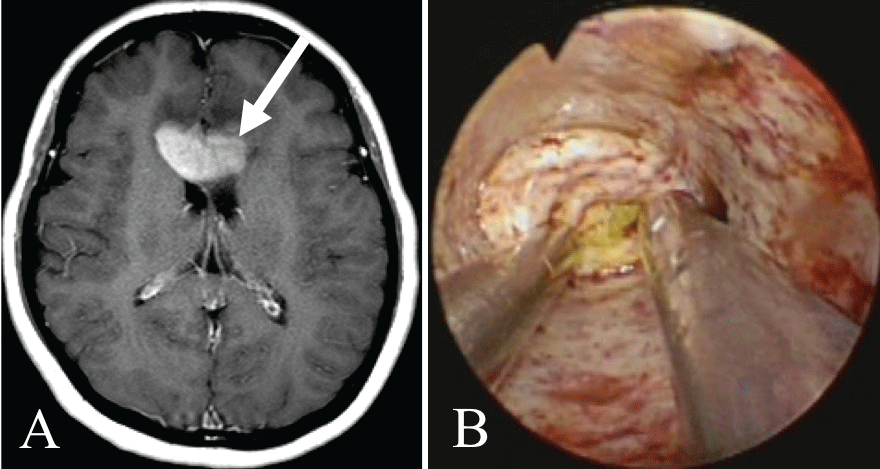
.
Figure 1: Illustrative case 2.
A) Axial T1-weighted gadolinium-enhanced MRI showing a primary central nervous system lymphoma in the corpus callosum. The white arrow shows the path of endoscope insertion;
B) The yellow-stained tumor can be seen under white light endoscopy.
View Figure 1
Case 6 (Figure 2): The patient was a 72-year-old man with glioblastoma. The approach was made via the right temporal lobe. The tumor was 38 mm from the brain surface and was confirmed to be colored yellow. Biopsy was performed, and a definitive pathological diagnosis of glioblastoma was made.
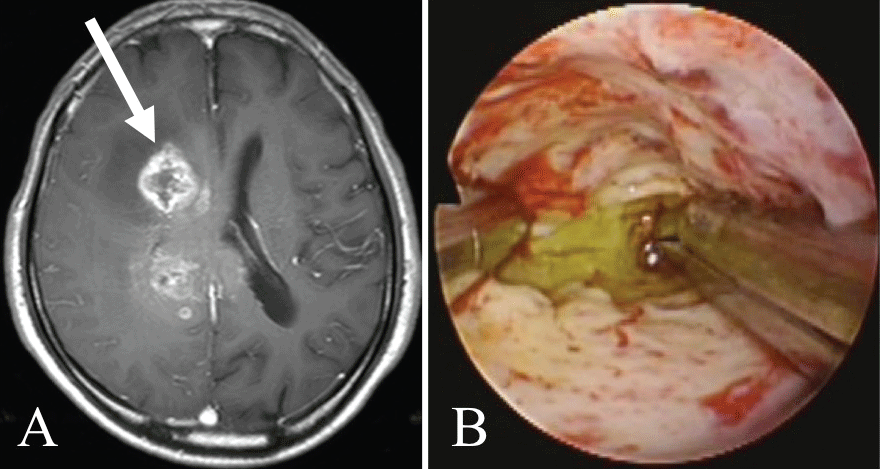
.
Figure 2: Illustrative case 6.
A) Axial T1-weighted gadolinium-enhanced MRI showing a glioblastoma in the right frontal lobe. The white arrow shows the path of endoscope insertion;
B) The yellow-stained tumor can be seen under white light endoscopy.
View Figure 2
Case 7 (Figure 3): The patient was a 40-year-old man with glioblastoma. The tumor was located on the medial side of the inferior horn of the right lateral ventricle, and the approach was made via the right temporal lobe. The tumor was 27 mm from the brain surface in the inferior horn of the lateral ventricle, and the choroid plexus was confirmed. This was separated to reveal the yellow-colored tumor. Biopsy was performed, and a definitive pathological diagnosis of glioblastoma was made.
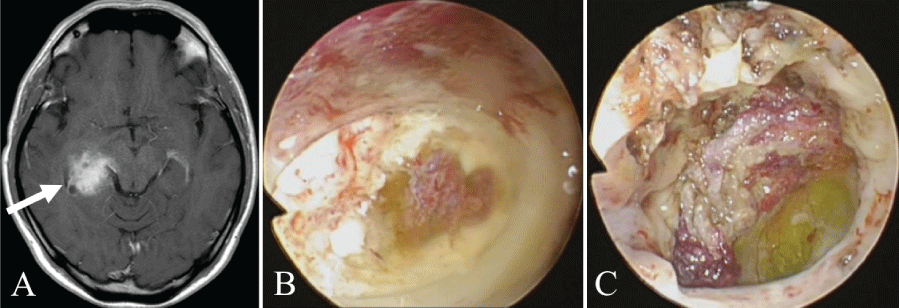
.
Figure 3: Illustrative case 7.
A) Axial T1-weighted gadolinium-enhanced MRI showing a glioblastoma in the right inferior horn of the lateral ventricle. The white arrow shows the path of endoscope insertion;
B) With penetration to the inferior horn of the lateral ventricle, the choroid plexus can be seen;
C) The yellow-stained tumor can be seen behind the choroid plexus.
View Figure 3
Case 8 (Figure 4): The patient was a 72-year-old man with glioblastoma. The tumor was located near the pars triangularis of the right lateral ventricle. There was also intrathecal dissemination. The tumor was approached via the right posterior temporal lobe. The ventricular wall of the pars triangularis of the lateral ventricle was 23 mm from the brain surface. As a result of biopsy of the ventricular wall at this site, a definitive pathological diagnosis of glioblastoma was made. After the biopsy, the ventricle was penetrated, and the yellow-colored choroid plexus was confirmed. With further observation of the inferior horn from the pars triangularis of the lateral ventricle, the yellow-colored ventricular wall was confirmed.
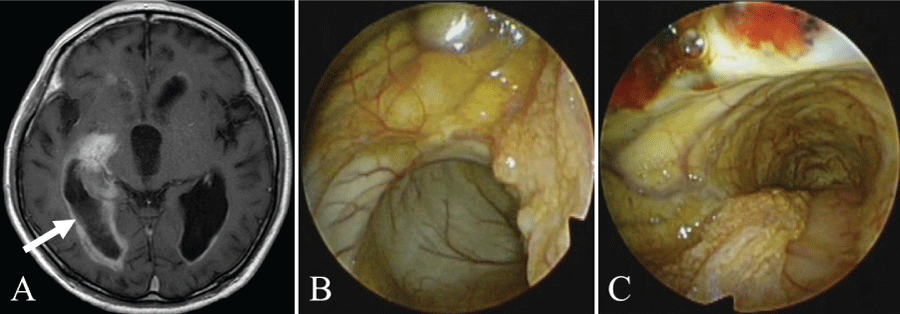
.
Figure 4: Illustrative case 8.
A) Axial T1-weighted gadolinium-enhanced MRI showing a glioblastoma in the right atrium of the lateral ventricle. The white arrow shows the path of endoscope insertion;
B) With penetration to the posterior horn of the lateral ventricle, the yellow-stained choroid plexus can be seen on observation of the pars triangularis of the lateral ventricle;
C) The yellow-stained ventricular wall can be seen when the endoscope moves in the direction of the inferior horn of the lateral ventricle.
View Figure 4
Discussion
Fluorescein sodium leaks through breaks in the blood-brain barrier and stains tissue yellow. The simple staining principle results in a sensitivity of 100% but specificity of 0% [4]. Since the staining corresponds to the area of contrast enhancement on images, this fluorescent stain is highly useful for patients with malignant brain tumors, high-grade glioma, malignant lymphoma, and metastatic brain tumors, of which major portions are contrast-enhanced.
As a fluorescent stain in brain tumor surgery, fluorescein sodium was initially used mostly in glioma surgery and reported to be effective [5]. With this method, the coloring from the fluorescein sodium needed to be observed with the use of an excitation filter, but Shinoda et al. reported that, by doubling the dose, the yellow coloration could be observed with regular white light microscopy without an excitation filter [6]. This method is simple because it does not require any special instruments, and today it is also used in metastatic brain tumor surgery, where good results have been reported [7,8]. In this study, this doubled dose was applied to endoscopic biopsy, and it was demonstrated that the yellow stain of the tumor could even be seen with regular white light endoscopy.
Another fluorescent stain that has been proposed for fluorescence-guided surgery is 5-aminolevulinic acid (ALA). The utility of this fluorescent stain has already been established in glioma surgery [9]. In recent years, imaging systems for 5-ALA have been established for endoscopic biopsy as well, and it is reported to be useful in cases of high-grade glioma [10,11]. 5-ALA has high sensitivity and specificity in glioma surgery, but its color development rate in other malignant tumors is uneven, and both its sensitivity and specificity are said to be low [12].
As for the surgical method of biopsy for the brain tumor, stereotactic biopsy is a standard method. Tumor biopsy is possible in local anesthesia and burr hole surgery in stereotactic biopsy. Therefore, stereotactic biopsy is less invasive surgery more than our surgical technique. In addition, the indication of our surgical technique for the non-contrasting lesion and deep sited lesion like the basal ganglia is difficult. However, our surgical technique has the effectiveness for the visual confirmation and the hemostasis of the lesion and can expect it for a future expandability. Furthermore, we accumulate cases and examine the effectiveness of our surgical technique.
A drawback to fluorescein sodium is that, during tissue collection, the fluorescein sodium leaks from the stump of the collected tissue, decreasing the staining properties. This drawback occurs in aggressive resections, but it is not thought to be a problem in biopsies. Yellow coloration of the skin, mucous membranes, and urine is seen as a side effect in all cases. However, since this is a temporary side effect that disappears within 24 hours, it is not particularly problematic. Anaphylactic shock is reported, but it is rare [13], and fluorescein sodium is very safe even when compared with other agents.
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper. This study has no financial support.
References
-
Ishikawa E, Yamamoto T, Matsuda M, Akutsu H, Zaboronok A, et al. (2015) Intraparenchymal brain lesion biopsy guided by a rigid endoscope and navigation system. Surg Neurol Int 6: 149.
-
Nagahisa S, Watabe T, Sasaki H, Nishiyama Y, Hayashi T, et al. (2013) Surgical navigation-assisted endoscopic biopsy is feasible for safe and reliable diagnosis of unresectable solid brain tumors. Neurosurg Rev 36: 595-600.
-
Tanei T, Nakahara N, Takebayashi S, Hirano M, Nagatani T, et al. (2012) Endoscopic Biopsy for Lesions Located in the Parenchyma of the Brain: Preoperative Planning Based on Stereotactic Methods. Neurol Med Chir (Tokyo) 52: 617-621.
-
Okuda T, Yoshioka H, Kato A (2012) Fluorescence-guided surgery for glioblastoma multiforme using high-dose fluorescein sodium with excitation and barrier filters. J Clin Neurosci 19: 1719-1722.
-
Kuroiwa T, Kajimoto Y, Ohta T (1998) Development of a Fluorescein Operative Microscope for use During Malignant Glioma Surgery: a technical note and preliminary report. Surg Neurol 50: 41-48.
-
Shinoda J, Yano H, Yoshimura S, Okumura A, Kaku Y, et al. (2003) Fluorescence-guided resection of glioblastoma multiforme by using high-dose fluorescein sodium. J Neurosurg 99: 597-603.
-
Okuda T, Kataoka K, Taneda M (2007) Metastatic brain tumor surgery using fluorescein sodium: technical note. Minim Invasive Neurosurg 50: 382-384.
-
Okuda T, Kataoka K, Yabuuchi T, Yugami H, Kato A (2010) Fluorescence-guided surgery of metastatic brain tumors using fluorescein sodium. J Clin Neurosci 17: 118-121.
-
Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, et al. (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7: 392-401.
-
Ritz R, Feigl GC, Schuhmann MU, Ehrhardt A, Danz S, et al. (2011) Use of 5-ALA fluorescence guided endoscopic biopsy of a deep-seated primary malignant brain tumor. J Neurosurg 114: 1410-1413.
-
Tamura Y, Kuroiwa T, Kajimoto Y, Miki Y, Miyatake S, et al. (2007) Endoscopic identification and biopsy sampling of an intraventricular malignant glioma using a 5-aminolevulinic acid-induced protoporphyrin IX fluorescence imaging system. J Neurosurg 106: 507-510.
-
Marbacher S, Klinger E, Schwyzer L, Fischer I, Nevzati E, et al. (2014) Use of fluorescence to guide resection or biopsy of primary brain tumors and brain metastases. Neurosurg Focus 36: E10.
-
Dilek O, Ihsan A, Tulay H (2011) Anaphylactic reaction after fluorescein sodium administration during intracranial surgery. J Clin Neurosci 18: 430-431.





