International Journal of Neurology and Neurotherapy
Marburg's Variant of Multiple Sclerosis with Extensive Brain Lesions: An Autopsy Case Report
Ayumi Ludwig1, Jaclyn Duvall1, Jo Elle Peterson2 and Ryan Hakimi1*
1Department of Neurology, University of Oklahoma Health Sciences Center, USA
2Department of Pathology, University of Oklahoma Health Science Center, USA
*Corresponding author:
Ryan Hakimi, Assistant Professor, University of Oklahoma Health Sciences Center, Director of Critical Care Neurology, Medical Director of Oklahoma University Medical Center Neurosciences Intensive Care Unit, 920 Stanton L. Young Blvd, Suite 2040, Oklahoma City, OK 73104, USA, Tel: 405-271-4113, E-mail: Ryan-Hakimi@ouhsc.edu
Int J Neurol Neurother, IJNN-2-027, (Volume 2, Issue 1), Case Report; ISSN: 2378-3001
Received: March 24, 2015 | Accepted: April 08, 2015 | Published: April 10, 2015
Citation: Ludwig A, Duvall J, Peterson JE, Hakimi R (2015) Marburg's Variant of Multiple Sclerosis with Extensive Brain Lesions: An Autopsy Case Report. Int J Neurol Neurother 2:027. 10.23937/2378-3001/2/1/1027
Copyright: © 2015 Ludwig A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Marburg's variant of multiple sclerosis (MS) is a malignant and rapidly progressive form of MS that typically leads to deterioration or death within weeks to months. Here we present a case involving a 25-year-old woman who presented with fluctuating mild encephalopathy of one-week duration who progressed to coma and ultimately died on day seven after being admitted to our university-based neurosciences intensive care unit (NSICU). During her stay, numerous studies were completed including a brain MRI with gadolinium which revealed multiple diffuse ring-enhancing lesions consistent with a fulminant demyelinating process, which was later supported by autopsy findings. CSF studies and serologies were essentially unremarkable. The rarity of Marburg's variant of MS has resulted in a scarcity of literature and lack of clinical recommendations regarding management.
After review of literature and with consideration of our case report, even when marked improvement following IV methylprednisolone is observed, adjunct therapy with a second modality should be considered to improve the chance of survival.
Keywords
Marburg variant multiple sclerosis, Malignant multiple sclerosis, Fulminant demyelination, Acute demyelination
Introduction
Marburg's variant of MS can be considered the most fulminant, and often fatal form of MS variant that typically leads to deterioration or death within weeks to months [1]. Marburg first described a malignant form of MS in 1906 with three cases of fulminant demyelinating disease. The first patient he described was a previously healthy, 30-year-old woman who suddenly developed confusion associated with headaches, vomiting, gait abnormality, left hemiparesis, and hyperreflexia, and died within one year [2].
Clinical course is often characterized by a short duration of rapid progression of neurologic signs such as hemiplegia, hemianopsia, aphasia, seizures, and confusion [3]. Radiographically, lesions demonstrate cerebral edema and may overlap with findings seen in other demyelinating conditions such as tumefactive MS, acute demyelinating encephalomyelitis (ADEM), or Balo's concentric sclerosis. Currently there is no definitive consensus on the distinguishing features of these variants of MS [4]. A similar radiographic appearance can be seen in central nervous system (CNS) malignancies or infections, therefore a timely diagnosis of this often fatal disease can be challenging.
Case Report
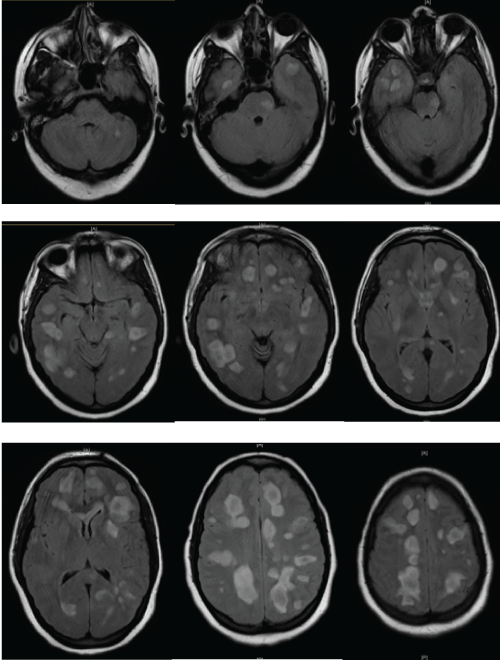
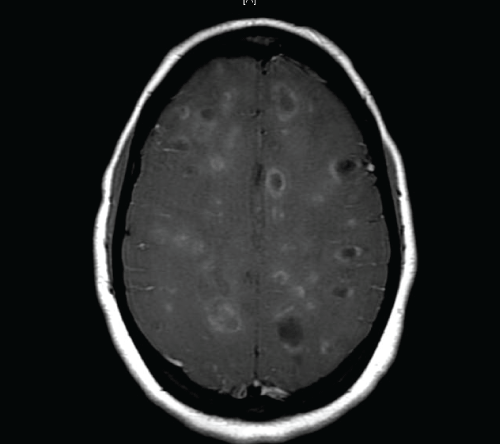
A 25-year-old woman without any known medical history presented to a referring emergency department with worsening confusion and was transferred to our university-based NSICU after a head CT was interpreted as diffuse cerebral edema. Nine days prior to admission, the patient was treated with a seven-day course of ciprofloxacin by her primary care physician for an upper respiratory tract infection and otitis media. The patient improved during this treatment. She then became progressively encephalopathic necessitating the emergency department visit. Upon presentation to our institution, she was oriented only to herself but otherwise had a non-focal neurological exam. MRI brain revealed numerous T2/FLAIR hyper intense lesions in the bilateral cerebral white and gray matter, brain stem, and cerebellum (Figure 1). These lesions also demonstrated incomplete ring-enhancement on T1 weighted imaging with gadolinium and peripheral restricted diffusion on DWI (Figure 2). There was not any mass effect noted. Urgent lumbar puncture revealed an opening pressure of 48mm H2O but was not consistent with an infectious process. She was started on methylprednisolone 1G IV daily for a suspected demyelinating process as well as broad-spectrum antimicrobials for a possible infectious etiology.
.
Figure 1: MRI brain, FLAIR sequence demonstrating multiple hyperintense lesions with surrounding vasogenic edema
View Figure 1
.
Figure 2: MRI brain, T1 with gadolinium, demonstrating multiple incomplete ring enhancing lesions
View Figure 2
Antibiotic and antiviral medications were discontinued as CSF cell and protein counts were normal and cultures were negative. CSF cytology was negative for malignancy.
With IV methylprednisolone therapy, her encephalopathy was improving to the point the patient could feed herself. However, on hospital day four she started showing signs of herniation. She was intubated and despite maximal medical management which included placement of an external ventricular drain and hyperosmolar therapy, she died on hospital day seven.
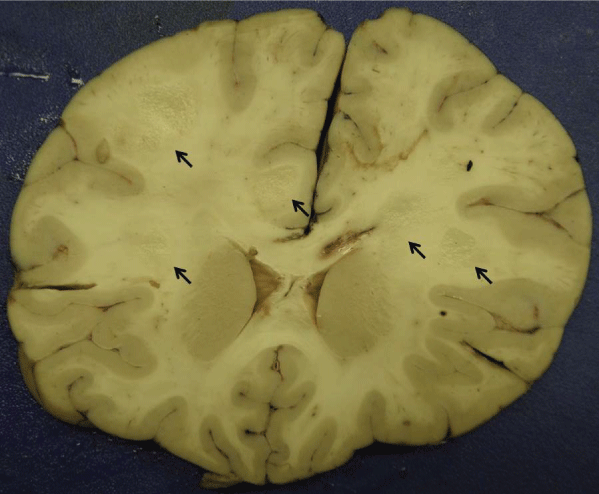
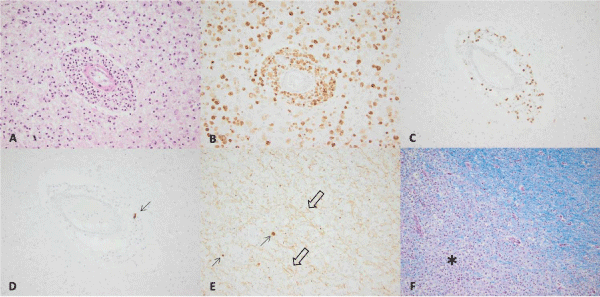
Autopsy was performed and showed numerous foci of subacute demyelination. Gross examination of the formalin-fixed brain showed marked and diffuse edema (brain weight - 1518g), with numerous white matter lesions involving all lobes of the cerebrum (Figure 3). The lesions were similar in appearance, with tan cut surfaces that were slightly sunken from the adjacent white matter. No cortical involvement by these lesions was seen, nor was involvement of the cerebellum, brainstem, or cranial nerves identified grossly. Microscopically, the lesions showed a prominent macrophage infiltrate, with perivascular chronic inflammation composed predominantly of T lymphocytes (Figure 4). Luxol fast blue/periodic acid Schiff (LFB/PAS) stain demonstrated marked loss of myelin and macrophages with collections of intracytoplasmic myelin. Axonal loss within these lesions ranged from mild to marked. Microglial activation was identified in the tissue surrounding the white matter lesions. Remyelination was not identified. Microscopically, lesions were demonstrated throughout the cerebrum, as well as a single focus within the pons. Additional neuropathologic findings included multiple acute infarcts, involving the left parietal, right occipital, and bilateral cerebellar hemispheres and a tract corresponding to the intraventricular pressure monitor placed prior to death.
.
Figure 3: Gross image of the formalin-fixed brain showing frontal lobes with severe edema, multiple foci of white matter demyelinating lesions (arrows), and the tract of the pressure monitor within the corpus callosum.
View Figure 3
.
Figure 4: H&E image (20X) of a demyelinating lesion with macrophage infiltration, perivascular macrophages, and scattered perivascular lymphocytes (A). CD68 immunostaining (20X) demonstrates immunoreactivity within macrophages, which are stained brown (B). CD3 immunostaining (20X) demonstrates perivascular T-lymphocytes, which are stained brown(C). CD20 immunostaining (20X) demonstrates scant perivascular B lymphocytes (arrow, D). Neurofilament immunostaining (20X) within a demyelinating lesion demonstrates retained axons (open arrows) as well as axonal spheroids (solid arrows) which represent axonal damage (E). Luxol-fast blue/periodic acid Schiff (LFB/PAS, 20X) special staining demonstrating retained myelin in the upper right and loss of myelin (∗) in the lower left, corresponding to a demyelinating lesion (F).
View Figure 4
Additional laboratory studies results became available after her death and are listed in Table 1. The only abnormal laboratory results included an elevated myelin basic protein (>120ng/mL, ref 0-1.2ng/mL), elevated erythrocyte sedimentation rate (46, ref 0 - 20) and elevated c-reactive protein (8.2, ref 0.2 - 8.0). However, oligoclonal bands were not present in the CSF. CSF IgG synthesis rate and ACE level were normal.
![]()
Table 1: Laboratory data
View Table 1
Discussion
Marburg's variant of MS is an uncommon entity. It is an acute malignant variant of MS characterized by extensive destructive lesions and a fulminant course usually resulting in death within one year [2,4]. Classical MS typically does not present with an encephalopathy, which is in contrast with Marburg's variant of MS [5]. Radiographically, the distribution of lesions in Marburg's variant of MS can be indistinguishable from classical MS, but unlike classical MS, CNS lesions with Marburg's variant of MS occur simultaneously in all affected areas, i.e. have a uniform radiographic appearance on brain MRI. In addition, these lesions are more destructive and widespread than classical MS [6]. CSF analysis in Marburg's variant of MS often reveals a normal to slightly elevated white blood cell count and increased protein [3]. Oligocolonal bands are less frequently detected than in classical MS [3]. Pathologically, the Marburg lesions are typically more destructive than those in classical MS and are associated with more severe axonal injury, edema, and necrosis. Dense cellular infiltrates that consist primarily of macrophages containing myelin breakdown products and enlarged astrocytes are also seen [6,7].
Characteristic radiographic findings were appreciated in our patient who had numerous lesions with gadolinium enhancement in the form of incomplete rings. The lesions were of the same age based on the fact that they all had the same degree of hyper intensity (Figure 2). However, the lesion burden of our patient was much greater than that described in the literature [1,5,6,8-18]. In addition, the incomplete ring enhancing lesions seen in our patient combined with the negative CSF studies supported the diagnosis of a demyelinating process and excluded an infection or malignancy [19]. Interestingly, our patient had normal CSF protein. The neuropathological findings of our patient were consistent with those described in the literature.
Here we present a case of Marburg's variant of MS whose lesion burden is much greater than that described in the literature. The clinical course, radiographic features, and post-mortem pathology of our patient were consistent with the diagnosis of this rare demyelinating disease. Our patient did not have any focal neurological signs, and her only presenting symptom was encephalopathy suggesting the importance of MRI brain imaging in making this diagnosis. Despite her good initial response to IV methylprednisolone, our patient's condition rapidly declined resulting in coma and eventually in death within a week of her admission. Although plasmapheresis, mitoxantrone, cyclophosphamide, and hemicraniectomy have been described in cases of Marburg's variant of MS where improvement is not seen following IV methylprednisolone, our patient's precipitous death following initial marked improvement with IV methylprednisolone suggests multimodal therapies should be considered even when the patient is improving with IV methylprednisolone therapy [5,8,9,16].
Conclusion
Data in the literature on Marburg's variant of MS is limited due to its rarity. The exact incidence of this condition is unknown, and to our knowledge there are only 17 cases that have been reported since Marburg's first description of the disease in 1906 [1,5,6,8-18,20-22]. Currently, consensus is lacking on the diagnostic criteria for Marburg's variant of MS or its optimal treatment. Despite improvement with standard initial treatment with IV methylprednisolone, our patient died within a week of admission. This suggests that even when marked improvement following IV methylprednisolone is observed, adjunct therapy with a second modality should be considered to improve the chance of survival. Future studies are needed to validate this hypothesis.
Acknowledgement
This work was done at University of Oklahoma Health Sciences Center.
Ethical Statement
Authorization for autopsy was obtained from the patient's mother.
Contribution Statement
Ayumi Ludwig MD and Jaclyn Duvall MD conducted the chart review, literature search, and drafted the article. Jo Elle Peterson MD performed the autopsy, drafted the autopsy report, and provided the pathology images. Ryan Hakimi DO, MS is the guarantor, responsible for concept, design, and revisions of the manuscript.
References
-
Johnson MD, Lavin P, Whetsell WO Jr (1990) Fulminant monophasic multiple sclerosis, Marburg's type. J Neurol Neurosurg Psychiatry 53: 918-921.
-
Marburg O (1906) Die sogenannte 'acute multiple sklerose'. J Psychiatr Neuro l27: 211-321.
-
Capello E, Mancardi GL (2004) Marburg type and Balò's concentric sclerosis: rare and acute variants of multiple sclerosis. Neurol Sci 25: S361-363.
-
Rahmlow MR, Kantarci O (2013) Fulminant demyelinating diseases. Neurohospitalist 3: 81-91.
-
Turatti M, Gajofatto A, Rossi F, Vedovello M, Benedetti MD (2010) Long survival and clinical stability in Marburg's variant multiple sclerosis. Neurol Sci 31: 807-811.
-
Suzuki M, Kawasaki H, Masaki K, Suzuki SO, Terada T, et al. (2013) An autopsy case of the Marburg variant of multiple sclerosis (acute multiple sclerosis). Intern Med 52: 1825-1832.
-
Roncaroli F (2005) Neuropathology of multiple sclerosis. Adv Clin Neurosci Rehabil 5: 16-22.
-
Jeffery DR, Lefkowitz DS, Crittenden JP (2004) Treatment of Marburg variant multiple sclerosis with mitoxantrone. J Neuroimaging 14: 58-62.
-
González Sánchez JJ, Nora JE, de Notaris M, Arboix JR, García CG, et al. (2008) A case of malignant monophasic multiple sclerosis (Marburg's disease type) successfully treated with decompressive hemicraniectomy. J Neurol Neurosurg Psychiatry 81: 1056-1057.
-
Elenein RG, Sharer LR, Cook SD, Pachner AR, Michaels J, et al. (2011) A second case of Marburg's variant of multiple sclerosis with vasculitis and extensive demyelination. Mult Scler 17: 1531-1538.
-
Giubilei F, Sarrantonio A, Tisei P, Gasperini C, Salvetti M (1997) Four-year follow-up of a case of acute multiple sclerosis of the Marburg type. Ital J Neurol Sci 18: 163-166.
-
Jasperson J, Jones AG (1998) A case of rapid deterioration: acute multiple sclerosis of the Marburg type. J Neurosci Nurs 30: 350-355.
-
Shields DC, Avgeropoulos NG, Banik NL, Tyor WR (2000) Acute multiple sclerosis characterized by extensive mononuclear phagocyte infiltration. Neurochem Res 25: 1517-1520.
-
Walid MS, Sanoufa M (2010) The diagnosis of Marburg disease is course-dependent. Ger Med Sci 8: Doc06.
-
Niebler G, Harris T, Davis T, Roos K (1992) Fulminant multiple sclerosis. AJNR Am J Neuroradiol 13: 1547-1551.
-
Nozaki K, Abou-Fayssal N (2010) High dose cyclophosphamide treatment in Marburg variant multiple sclerosis A case report. J Neurol Sci 296: 121-123.
-
Bitsch A, Wegener C, da Costa C, Bunkowski S, Reimers CD, et al. (1999) Lesion development in Marburg's type of acute multiple sclerosis: from inflammation to demyelination. Mult Scler 5: 138-146.
-
Letournel F, Cassereau J, Scherer-Gagou C, Bernard I, Mercat A, et al. (2008) An autopsy case of acute multiple sclerosis (Marburg's type) during pregnancy. Clin Neurol Neurosurg 110: 514-517.
-
Masdeu JC, Quinto C, Olivera C, Tenner M, Leslie D, et al. (2000) Open-ring imaging sign: highly specific for atypical brain demyelination. Neurology 54: 1427-1433.
-
Wood DD, Bilbao JM, O'Connors P, Moscarello MA (1996) Acute multiple sclerosis (Marburg type) is associated with developmentally immature myelin basic protein. Ann Neurol 40: 18-24.
-
Mendez MF, Pogacar S (1988) Malignant monophasic multiple sclerosis or "Marburg's disease". Neurology 38: 1153-1155.
-
Talab R, Kundrata Z (2011) Marburg variant multiple sclerosis - a case report. Neuro Endocrinol Lett 32: 415-420.





