International Journal of Neurology and Neurotherapy
Novel Aneurysm Grown from a PcomA Infundibulum after Embolization of a Contralateral Posterior Communicating Aneurysm in an Isolated Polycystic Hepatic Patient: A Case Report
Rui Zhao#, Chao Zou#, Chuan-Chuan Wang, Qing-Hai Huang and Jian-Min Liu*
Department of Neurosurgery, Stroke Center, Changhai Hospital, Second Military Medical University, China
#Equal Contribution
*Corresponding author:
Jian-Min Liu, Department of Neurosurgery, Stroke Center, Changhai Hospital, Second Military Medical University, 168 Changhai Road, Shanghai 200433, China, Tel: 8513916728169; E-mail: chstroke@163.com
Int J Neurol Neurother, IJNN-1-008, (Volume 1, Issue 1), Case Report; ISSN: 2378-3001
Received: October 05, 2014 | Accepted: October 22, 2014 | Published: October 27, 2014
Citation: Zhao R, Zou C, Wang CC, Huang QH, Liu JM (2014) Novel Aneurysm Grown from a PcomA Infundibulum after Embolization of a Contralateral Posterior Communicating Aneurysm in an Isolated Polycystic Hepatic Patient: A Case Report. Int J Neurol Neurother 1:008. 10.23937/2378-3001/1/1/1008
Copyright: ©2014 Zhao R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We present a case of a posterior communicating (Pcom) artery infundibulum that progressed to an aneurysm in a patient with Polycystic liver disease without kidney cyst after embolization of the controlateral Pcom aneurysm. The infundibulum had been documented on angiography 4 years earlier, while a ruptured controlateral Pcom aneurysm was embolised. The patient presented to the outpatient clinic with right postorbital headache and cranial ? nerve palsy. Head CT scan found no gross subarachnoid hemorrhage. Though MR angiography note a 7-mm pcom aneurysm at the previous site of Pcom infundibulum. The patient underwent coiling for the novel aneurysm with a good result. This case illustrates a small but growing number of cases which developed from infundibula into aneurysms. It suggests that polycystic disease may pose an additional risk for infundibulum while what has traditionally been considered as a benign lesion.
Keywords
Aneurysm, Infundibulum, Polycystic liver disease
Introduction
Intracranial Arterial Infundibulum (IAF) is frequently regarded as incidental normal anatomical variant which rarely causes severe events. Up to now, reports of IAF rupture and aneurysms arised from IAF were rare. We report a case of development of aneurysm from IAF at origin of Pcom artery with a recurrent contralateral Pcom Aneurysm and a history of Polycystic Liver Disease (PLD).
Case Report
A 48-year-old man suffered sudden onset of headache with nausea and vomiting. CT scanning revealed a diffused SAH. He had a Polycystic Liver Disease (PLD) without kidney cyst, a ten-year history of HBP, no history of cigarette smoking, no familial history of intracranial aneurysm or Autosomal dominant PLD (ADPLD). After admission, cerebral angiography disclosed a left Pcom Aneurysm (Figure 1A) with an infundibulum (Figure 1C) at the origin of right Pcom artery. The aneurysm measured 6.10mm in width, 2.86mm in neck, 5.50mm in height. The infundibulum is less than 3mm in diameter. The patient was discharged with no symptom after the aneurysm was embolized with GDC coils successfully. He has no neurologic symptom during two-year clinical follow-up at outpatient clinic and had no more follow-up until the 51st month after the treatment. The patient came back to our outpatient clinic and complained of paroxysmal right post-orbit headache with cranial ? nerve palsy symptom. We found out that he hadn't had a regular visit to a physician to control his blood pressure well after aneurysm embolization. CT scanning revealed no SAH. Follow-up cerebral angiography suggested neck recurrence of L-Pocm Aneurysm (Figure 1B) and a novel saccular aneurysm (Figure 1D) at the previous site of right Pcom infundibulum. The size of novel aneurysm is 7.53 mm in length, 3.33 mm in width.
Endovascular treatment was considered as the first choice since the novel aneurysm was unrupture and the recurrent one had a small sac. The symptoms were assumed to be cause by the enlargement of right PcomAn. So, we performed the endovascular treatment of the novel aneurysm.
The novel aneurysm was successfully embolized by coiling with GDC coils. Post-embolization angiography suggested a neck residual (Figure 1E). Although the patient had no symptom, recurrence of R-Pcom aneurysm without obvious change of the left one was showed by 3-month follow-up angiography (Figure 1F). We considered retreating both aneurysms. But the patient refused and complained of no symptom in 4 more year's clinical follow-up.
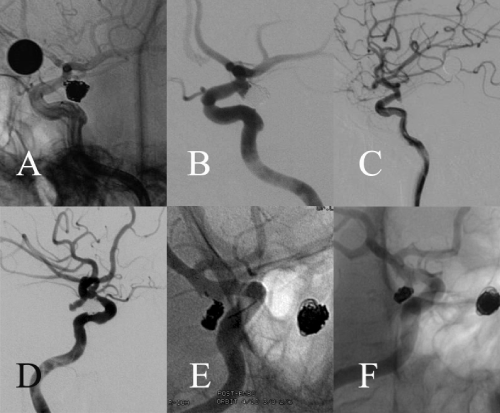 Figure 1: A: Postoperative angiogram of the ruptured left ICA PcomA
aneurysm. B: Four-year follow-up angiogram of the left ICA PcomA
aneurysm. C: A 3-mm, funnel-shaped infundibulum at conjunction of right
ICA-PcomA with a branch arises from whose tip. D: A novel aneurysm was
revealed at right PcomA at the site of prevous infundibulum at 4-yr-FU.
E: Postoperative angiogram of the novel right ICA PcomA aneurysm. F:
DSA follow-up at 3 months revealed neck recurrence of the right PcomA
aneurysm.
View Figure 1
Figure 1: A: Postoperative angiogram of the ruptured left ICA PcomA
aneurysm. B: Four-year follow-up angiogram of the left ICA PcomA
aneurysm. C: A 3-mm, funnel-shaped infundibulum at conjunction of right
ICA-PcomA with a branch arises from whose tip. D: A novel aneurysm was
revealed at right PcomA at the site of prevous infundibulum at 4-yr-FU.
E: Postoperative angiogram of the novel right ICA PcomA aneurysm. F:
DSA follow-up at 3 months revealed neck recurrence of the right PcomA
aneurysm.
View Figure 1
Discussion
IAFs have variously been described as funnel-shaped or triangular symmetrical widenings at origins of major cerebral arteries, principally occurring at the junction of the ICA and PComA [1]. IAFs are revealed on 7% to 25% of otherwise normal angiograms [2,3], and the incidence seems to be greater in cases of multiple [2] or familial [3] aneurysms.
The significance of IAF is under controversy. It is frequently regarded as normal anatomical variants that without pathogenetic significance [4], for its not too low occurrence in general population but generally resulting in no event. Some authors believe this lesion is preaneurysmal [3], requesting more attention and regular follow-ups for its higher incidence in patients with multiple, familial intracranial aneurysms than general people. Further more, histological examinations shew medial defect with a disrupted internal elastic lamina in some IAFs, just the same as that in normal artery bifurcations and aneurysms [5]. Up to now, six cases of IAF rupture without aneurismal formation were reported [3], and the authors suggested more attention be paid on some isolated IAF cases with SAH. These rare cases put us a not much too convincible but unoverlooked evidence of pathogenetic significance of IAF after all.
We present here the case of a man with isolated PLD and a history of HBP and a rupture left PCoA aneurysm in which a contralateral PCoA aneurysm developed at the site of a previously documented infundibulum during follow-up angiography. To our knowledge, this is the 14th known case about infundibulum growing into aneurysm in the literature [6-8]. According to our case and literature review the isolated PLD [9] and hypertensive may be risk factors for the novel ameurysm formation and recanalization of the embolized one. Cowan et al. [8] reported a case of progression from IAF to aneurysm in a man with Alagille Syndrome, a disease which may result in high incidence of systematic arterial lesions, including CVD.
Autosomal Dominant PLD (ADPLD), a rare inherited Mendelian disorder (with ADPLD gene mutation of PRKCSH [10] and SEC63 [11] ) that is characterized by development of multiple hepatic cysts, is genetically different from Autosomal-Dominant Polycystic Kidney Disease (ADPKD). The latter is a hereditary systemic connective tissue disease involving cardiocerebral vessels [12], especially intracranial aneurysm [13]. ADPLD has been reported significantly relative with cerebral aneurysms at autopsy [14] and patients [15]. The occurrence of vascular type in ADPLD patients is much higher than those in the general population [16]. We can't diagnose this patient ADPLD, because we did not perform the gene analysis. As our case is concerned, the vessel wall abnormality caused by polycystic disease may be a risk factor of aneurysmal occurrence and relapsing after occlusion the sac. So we suggest that patients with polycystic disease should accept screening and regular follow-ups for cerebrovascular disease, especially for intracranial aneurysm.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (81000495).
References
-
Yoshimoto T, Suzuki J (1974) Surgical treatment of an aneurysm on the funnel-shaped bulge of the posterior communicating artery. J Neurosurg 41: 377-379.
-
Leblanc R, Worsley KJ, Melanson D, Tampieri D (1994) Angiographic screening and elective surgery of familial cerebral aneurysms: A decision analysis. Neurosurgery 35: 9-18.
-
Coupe NJ, Athwal RK, Marshman LA, Brydon HL (2007) Subarachnoid hemorrhage emanating from a ruptured infundibulum.Case report and literature review. Surg Neurol 67: 204-206.
-
Edelsohn L, Caplan L, Rosenbaum AE (1972) Familial aneurysms and infundibular widening. Neurology 22: 1056-1060.
-
Archer CR, Silbert S (1978) Infundibula may be clinically significant. Neuroradiology 15: 247-251.
-
Marshman LA, Ward PJ, Walter PH, Dossetor RS (1998) The progression of aninfundibulum to aneurysm formation and rupture: case report andliterature review. Neurosurgery 43: 1445-1449.
-
Martins C, Macanovic M, Costa e Silva IE, Griz F, Azevedo-Filho HR (2002) Progression of an arterial infundibulum to aneurysm: case report. Arq Neuropsiquiatr 60: 478-480.
-
Cowan JA Jr, Barkhoudarian G, Yang LJ, Thompson BG (2004) Progression of a posterior communicating artery infundibulum into an aneurysm in a patient with Alagille syndrome. Case report. J Neurosurg 101: 694-696.
-
Schievink WI, Spetzler RF (1998) Screening for intracranial aneurysms in patients with isolated polycystic liver disease. J Neurosurg 89: 719-721.
-
Drenth JP, TeMorsche RH, Smink R, Bonifacino JS, Jansen JB (2003) Germlinemutations in PRKCSH are associated with autosomal dominantpolycystic liver disease. Nat Genet 33: 345-347.
-
Davila S, Furu L, Gharavi AG, Tian X, Onoe T, et al. (2004) Mutations in SEC63 cause autosomal dominant polycystic liver disease. Nat Genet 36: 575-577.
-
Arnaout MA (2000) The vasculopathy of autosomal dominant polycystic kidney disease:Insights from animal models.Kidney Int 58: 2599-2610.
-
Niemczyk M, Gradzik M, Niemczyk S, Bujko M, Golebiowski M, et al. (2013) Intracranial aneurysms in autosomal dominant polycystic kidney disease. AJNR Am J Neuroradiol 34: 1556-1559
-
Geevarghese SK, Powers T, Marsh JW, Pinson CW (1999) Screening for cerebral aneurysm in patients with polycystic liver disease. South Med J 92: 1167-1170.
-
Schievink W, Spetzler RF (1998) Screening for intracranial aneurysms in patients with isolated polycystic liver disease. J Neurosurg 89: 719-721.
-
Qian Q, Li A, King BF, Kamath PS, Lager DJ, et al. (2003) Clinical profile of autosomal dominant polycystic liverdisease. Hepatology 37: 164-171.





