Journal of Dermatology Research and Therapy
Report a Case of Linear Iga Bullous Dermatosis During Gestation and Minor Review for Features of Different Subtypes and Differential Diagnosis
Xiaoyue Wang1, Pengfei Song2 and Jing Wang2*
1The College of Biological Science, University of California, USA
2Department of Dermatology, The Eighth Affiliated Hospital Sun Yat-sen University, China
*Corresponding author: Jing Wang, Ph.D, Professor of Dermatology, Director of Department of Dermatology, The Eighth Affiliated Hospital Sun Yat-sen University, China, Tel: 86-755-83982222, E-mail: jwangray@vip.tom.com
J Dermatol Res Ther, JDRT-3-043, (Volume 3, Issue 1), Case Report; ISSN: 2469-5750
Received: January 07, 2017 | Accepted: February 23, 2017 | Published: February 25, 2017
Citation: Wang X, Song P, Wang J (2017) Report a Case of Linear Iga Bullous Dermatosis During Gestation and Minor Review for Features of Different Subtypes and Differential Diagnosis. J Dermatol Res Ther 3:043. 10.23937/2469-5750/1510043
Copyright: © 2017 Wang X, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Linear IgA bullous dermatosis (LABD) is a rare auto-immune bullous disease occurring in adults or childhood. There are similarities and differences between these two subtypes of the disease. We report a twenty-seven-year-old patient with adult subtype of LABD. The disease started from the second month of gestation and remitted after the delivery.
We briefly reviewed literature about the features on epidemiological and clinical data, laboratory investigations for adult linear IgA disease and childhood IgA disease. Lesions in children are mainly localized to the lower abdomen and the perineal area but in adults, face and trunk are frequently affected. The disease may be either idiopathic or triggered by several medications and some other autoimmune diseases. Direct immunofluorescence in both forms of LABD shows linear IgA deposition on the basement membrane zone. T cells and other celluar immune components are involved in the IgA-mediated autoimmune disease.
Introduction
Linear IgA bullous dermatosis (LABD) is a rare autoimmune blistering disease characterized by the linear deposition of IgA at the dermoepidermal junction. It can be associated with some inflammatory bowel disorders, drug-induced, neoplasm or idiopathic [1]. We report a case of typical adult type of LABD probably induced by gestation.
Although the clinical features of this disorder can be difficult to distinguish from some other autoimmune blistering disease, such as dermatitis herpetiformis or cicatricial pemphigoid, the distinct immunopathologic findings in LABD and the absence of an associated gluten-sensitive enteropathy confirm the status of LABD as a distinct disease [2].
The disease occurs in both adults and children. The differences and similarities between these two subtypes of the diseases on clinical features and laboratory investigations will be compared and discussed in this review.
Case Reports
The patient is a 27-year-old woman who had severe itching erythema and blisters over her trunk and extremities for 3 months. The patient started to have blisters on her arms from the second month of her second gestation. She was given antihistamines, systemic corticosteroid and corticosteroid creams and the lesions increased gradually. She reminded a history of mild similar vesicles and itching skin problems during her first gestation two years ago. The lesion recovered after delivery without any medication. On examination, the patient was generally healthy except for the skin problems. Numerous vesicles or bullae and crusts on the red macules were seen on her shoulders, back and four extremities (Figure 1). The vesicles or bullae were generally tense and Nikolsky's sign was negative. No mucosa involvement was found.
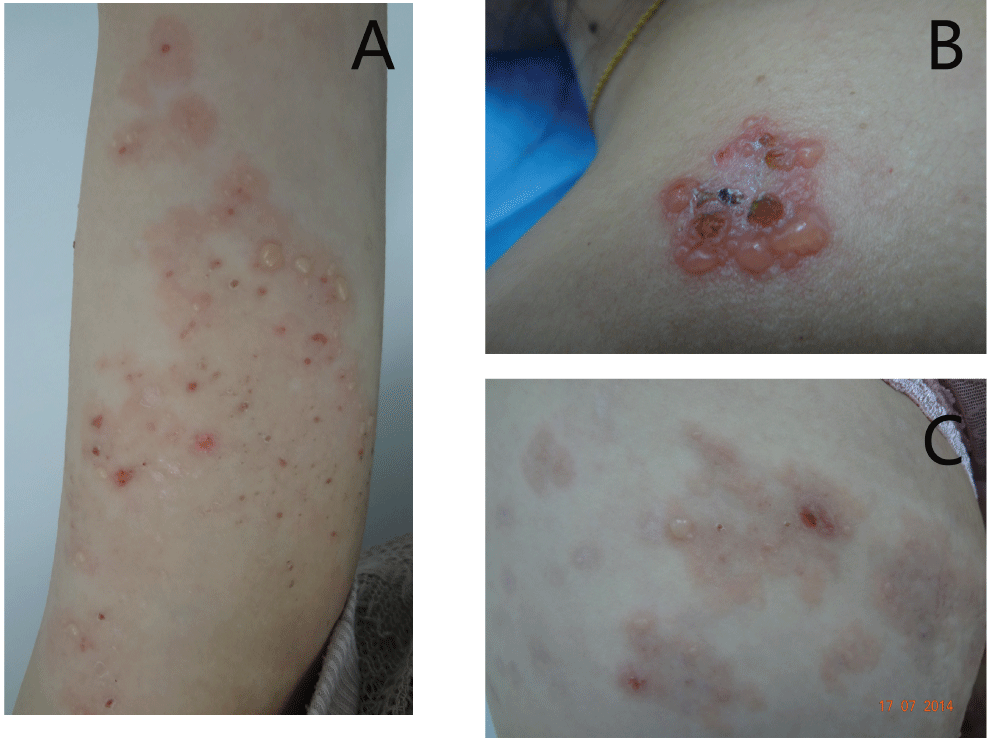
.
Figure 1: Clinical pictures of the patient lesions.
A) Patient's arm, vesicles, bullae and crusts were seen on the border of large reddish annular macules; B) The same patient. Cluster of vesicles on the shoulder. The vesicles or bullae were generally tense and Nikolsky's sign was negative; C) Annular erythema and bullae on the pregnant woman's abdomen.
View Figure 1
Histopathology for biopsy taken from the arm lesions showed subcutaneous blisters (Panel A, Figure 2) and large amount of infiltration of neutrophils and lymphatic cytes in upper dermis layer (Panel B, Figure 2). Direct immunofluorescence shows linear IgA deposition on the BMZ (Panel C, Figure 2) and negative C3 or IgG deposition. ELISA for Desmoglein1, Desomoglein3, Bp180 and Bp230 IgG antibodies were negative (Data not shown). Immunoblot using human epidermis protein stripes showed IgA antibodies from patient sera were reacted against 180-kDa, 120-kD, 100-kDa proteins (Panel D, Figure 2). Based on the histopathologic and immunological findings, we made a diagnosis of adult subtype of linear IgA bullous dermatosis. Herpes gestationis was excluded because the linear IgA deposition at BMZ, the sub-epidermal cleft was infiltrated with neutrophils, but eosinophils was rarely observed. The patient was successfully treated with low dose of daposone (100 mg daily) for 2 weeks. The skin lesions disappeared in one week and she remains well with maintenance dose of 50 mg every other day. However, the lesions recurred after she stopped medicine 4 weeks before the delivery. She came back 12 months after delivery to follow up, and reported that the lesions recovered in three weeks after delivery without any medication.
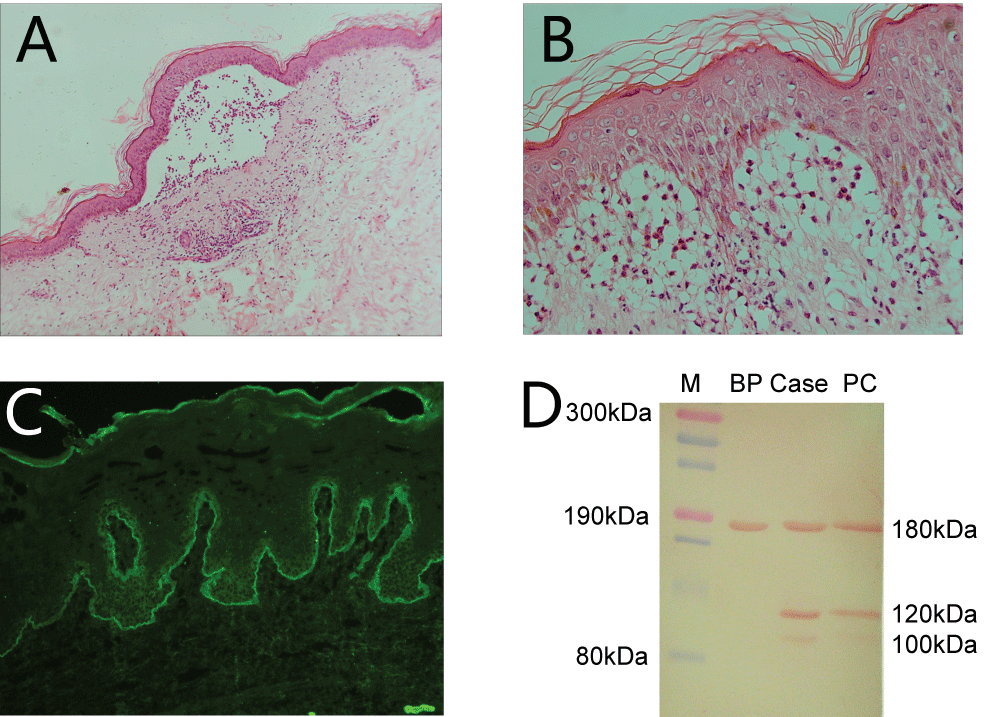
.
Figure 2: Histopathological findings and laboratory investigations of the disease.
A) Histopathology of biopsy taken from the abdomen lesion showed sub-epidermis blisters and large amount of neutrophil infiltration. Very few eosinophils are observed. (H&E, × 100); B) The same slide, sub-epidermal separation with large amount of neutrophils. (H&E, × 400); C) Direct immunofluorescence on normal skin outside the border of blistering lesions showed linear IgA deposition on the basement membrane zone; D) Immunoblot: IgA antibodies from patient sera were reacted against 180-kDa, 120-kDa 100-kDa proteins using human epidermis protein stripes. M for marker, BP for patient serum from bullous pemphigoid, Case for our reported case, PC for positive control.
View Figure 2
Discussion and Review of Articles
Linear IgA dermatosis is a rare immune-mediated blistering skin disease. It may affect people of all races and all ages, but actually two peaks have been predominantly observed: childhood-onset and adult-onset.
Clinical difference of childhood LABD and adult LABD
Since the late 1980s, the disorder designated as chronic bullous disease of childhood (CBDC) has been recognized as the Childhood linear IgA bullous dermatosis (LABD) [3]. Childhood LABD usually appears after 6 months old. Even though several newborns with LABD have been described, it usually remits by age 13 [4]. Besides the age of onset, another difference between the two types concerns the diversity of clinical features. Lesions in children are mainly localized to the lower abdomen and the perineal area, with a prominent anogenital involvement, where blisters commonly occur in an annular blisters pattern. New lesions often arise at the periphery of the older ones. Faces, hands, and feet are rarely involved in this type [4,5].
Adult linear IgA bullous dermatosis appears after puberty or later in life, generally after age 60. Epidemiologic data on its distribution and sex ratio worldwide are very heterogeneous [4]. It is also an acquired autoimmune blistering disease that may present with a clinical pattern of vesicles indistinguishable from dermatitis herpetiformis, or with vesicles and bullae in a bullous pemphigoid-like appearance [1,4]. In adults, annular lesions like our case are less common. The extensor extremities, face, and trunk are frequently affected but perianal or anogenital areas are seldom involved.
Clinical similarities of childhood LABD and adult LABD
Cutaneous manifestations on both types of LABD often mimic those on BP patients. These lesions may be distributed symmetrically or asymmetrically, appearing as clear or hemorrhagic vesicles or bullae. They are generally tense and can cause erosion, ulceration and scar [5]. The lesions may also show unusual features such as itchy, roundish excoriated papules, nodules that look like prurigo nodularis [6], contact dermatitis-like lesions [4], urticariod papules and erythema multiform [7]. The manifestation can also mimic toxic epidermal necrolysis [8]. Mucosal lesions, manifesting as inflammation, erosions, or scarring, may also occur in patients with LABD [4,5]. All mucous membranes may be involved and the most common affected mucosal sites are the oral cavity and eyes. If not promptly treated, these lesions may lead to blindness [4,9].
Both types of LABD cases correlate with drugs and systemic diseases such as inflammatory bowel disorders, cancers or autoimmune disorders, and less frequently, with traumatic events such as burns and ultraviolet light exposure [1,10-13]. For drug-induced LABD, withdrawal of the medication is followed by a quick healing of the lesions. The most common reported drug is vancomycin. Some other antibiotics, non-steroid anti-inflammatory, anti-hypertensive and anti-epileptic drugs were also reported. However, a latest article concluded that although many drugs have been alleged to induce LABD over the past 30 years, this has been proven conclusively in very few cases [7,8,10].
Laboratory investigations for LABD
Humoral and cellular immune responses are both involved in the pathogenesis of both types of LABD. B cells and immunoglobulins are involved in mediating tissue injury, along with the antigen specific T cells which release cytokines, natural cellular immune cells, and specific pattern of proteolytic enzymes.
Investigations have identified different antigens involved in the pathophysiology of LABD. Direct immunofluorescence shows linear IgA deposition on the BMZ. Immunoblot and immunoelectron microscopy study reveals IgA auto-antibodies typically directed against a ectodomain of collagen XVII/BP180 antigen, LAD-1 (a 97 or 120 kD protein) antigen, 100-kDa and 145-kDa antigen, 285-kDa antigen, and other components of dermal-epidermal junction [3,14,15].
The patients were identified autoreactive CD4+ T helper (Th) cells against a specific NC 16A domain of BP180 and might be involved in type XVII collagen degradation. Plasminogen activation, factors released by keratinocytes and neutrophils, activation of pro-MMP-9 into MMP-9/gelatinase B and activation of neutrophil elastase, degranulation of mast cells, seem to be important factors involved in tissue injury and blister formation [16,17]. Recent animal models in mice have showed that passive transfer of monoclonal IgA against the 97-kDa LABD antigen into SCID mice can resulted in IgA deposits along the BMZ, a significant recruitment of neutrophils and a subepithelial detachment [18].
LABD in children and in adults have been revealed to carry an increased frequency of particular HLA genotypes (HLA B8,Cw7,B8,DR2,DR3 reported) and tumor necrosis factor-2 genotypes [4,19].
Differential diagnosis between LABD in pregnancy and pemphigoid gestationis
Our patient was probably gestation induced, which was a novel correlated factor of the disease. However, pemphigoid gestationis should be excluded before we confirm the diagnosis of LABD. Pemphigoid gestationis is the disease of bullous pemphigoid (BP) presented during the gestation. Antibodies from the BP patient serum included 180 and 230 KDa IgG antibodies is the main immunological character for the diagnosis confirmation. In LABD, as we described above, IgA auto-antibodies against BP180 antigen, 97 or 120 kD LAD-1 antigen, 100-kDa and 145-kDa antigen is the characters for immunological investigations. Western blot for the reported case showed positive IgA antibodies against 180-kDa, 120-kDa and 100-kDa proteins and direct immunofluorescence reported linear IgA deposition on the basement membrane zone. Therefore, we confirmed the diagnosis for LABD and excluded diagnosis for BP. Although Horvath, B et al described the presence of IgA directed against antigens of the dermal-epidermal junction in patients with pemphigoid gestationis, we believed LABD should be excluded for their patient [20]. The pathogenesis for LABD during the gestation remained unknown. We cannot exclude the hormone changes during gestation or some drug induced factors triggered the onset of LABD, and further investigations are needed.
Future Perspective
Up to the present, pathological, immunological and genetic studies have not explained why there are two subtypes of the disease with a similar pathophysiological aspect. The etiology or the underlying pathophysiologic mechanism that triggers the similar autoimmune response indifferent types of LABD still remains unknown. No strong genetic background to LABD has yet been reported. Further molecular and genetic study should focus on the explanation of the different epidemiological and clinical manifestations in LABD.
Conclusion
Besides the age of onset, the difference between adult and childhood type of LABD concerns diversity of clinical features and lesion distribution. Both forms of the disease are characterized by the linear deposition of IgA at BMZ. Recent study showed that T cells and other cellular immune components, such as nutrophils, eosinophils, mast cells etc., are involved in the IgA-mediated autoimmune disease. Gestation is probably correlated to the LABD for the reported case.
Acknowledgement
We thank Dr. Zhi Liu from Department of Dermatology, School of Medicine, University of North Carolina at Chapel Hill, USA for his important comments and useful suggestions.
References
-
Tiger JB, Rush JT, Barton DT, Danilov AV, Chapman MS (2015) Urticarial linear IgA bullous dermatosis (LABD) as a presenting sign of chronic lymphocytic leukemia (CLL). JAAD Case Rep 1: 412-414.
-
Lawley TJ, Strober W, Yaoita H, Katz SI (1980) Small intestinal biopsies and HLA types in dermatitis herpetiformis patients with granular and linear IgA skin deposits. J Invest Dermatol 74: 9-12.
-
Wojnarowska F, Marsden RA, Bhogal B, Black MM (1988) Chronic bullous disease of childhood, childhood cicatricial pemphigoid, and linear IgA disease of adults. A comparative study demonstrating clinical and immunopathologic overlap. J Am Acad Dermatol 19: 792-805.
-
Giulio FM, Peter M (2012) Linear immunoglobulin A bullous dermatosis. Clinics in Dermatology 30: 38-50.
-
Freedberg (2003) Fitzpatrick's Dermatology in General Medicine. (6th edn), McGraw-Hill.
-
Antiga E, Bellandi S, Bianchi B, Bianco ED, Pierini I, et al. (2012) A further case of subacute prurigo-like linear IgA bullous dermatosis: growing evidence of a new subset. Int J Dermatol 51: 1500-1501.
-
Armstrong AW, Fazeli A, Yeh SW, Mackool BT, Liu V (2004) Vancomycin-induced linear IgA disease manifesting as bullous erythema multiforme. J Cutan Pathol 31: 393-397.
-
Khan I, Hughes R, Curran S, Marren P (2009) Drug-associated linear IgA disease mimicking toxic epidermal necrolysis. Clin Exp Dermatol 34: 715-717.
-
Talhari C, Althaus C, Megahed M (2006) Ocular linear IgA disease resulting in blindness. Arch Dermatol 142: 786-787.
-
Fortuna G, Salas-Alanis JC, Guidetti E, Marinkovich MP (2012) A critical reappraisal of the current data on drug-induced linear immunoglobulin A bullous dermatosis: a real and separate nosological entity? J Am Acad Dermatol 66: 988-994.
-
Usmani N, Baxter KF, Child JA, Sheehan-Dare R (2004) Linear IgA disease in association with chronic lymphocytic leukaemia. Br J Dermatol 151: 710-711.
-
Zhao CY, Chiang YZ, Murrell DF (2016) Neonatal Autoimmune Blistering Disease: A Systematic Review. Pediatr Dermatol 33: 367-374.
-
He C, Xu H, Xiao T, Geng L, Chen HD (2007) Localized linear IgA dermatosis induced by UV light-treatment for herpes zoster. Int J Dermatol 46: 500-502.
-
Marinkovich MP, Taylor TB, Keene DR, Burgeson RE, Zone JJ (1996) LAD-1, the linear IgA bullous dermatosis autoantigen, is a novel 120-kDa anchoring filament protein synthesized by epidermal cells. J Invest Dermatol 106: 734-738.
-
Allen J, Wojnarowska F (2003) Linear IgA disease: the IgA and IgG response to the epidermal antigens demonstrates that intermolecular epitope spreading is associated with IgA rather than IgG antibodies, and is more common in adults. Br J Dermatol 149: 977-985.
-
Liu Z, Li N, Diaz LA, Shipley M, Senior RM, et al. (2005) Synergy between a plasminogen cascade and MMP-9 in autoimmune disease. J Clin Invest 115: 879-887.
-
Hofmann SC, Voith U, Schonau V, Sorokin L, Bruckner-Tuderman L, et al. (2009) Plasmin plays a role in the in vitro generation of the linear IgA dermatosis antigen LADB97. J Invest Dermatol 129: 1730-1739.
-
Zone JJ, Egan CA, Taylor TB, Meyer LJ (2004) IgA autoimmune disorders: development of a passive transfer mouse model. J Investig Dermatol Symp Proc 9: 47-51.
-
Collier PM, Wojnarowska F, Welsh K, McGuire W, Black MM (1999) Adult linear IgA disease and chronic bullous disease of childhood: the association with human lymphocyte antigens Cw7, B8, DR3 and tumour necrosis factor influences disease expression. Br J Dermatol 141: 867-875.
-
Horvath B, Niedermeier A, Podstawa E, Muller R, Hunzelmann N, et al. (2010) IgA autoantibodies in the pemphigoids and linear IgA bullous dermatosis. Exp Dermatol 19: 648-653.





