Journal of Dermatology Research and Therapy
Topical Treatment of Cutaneous Leishmaniasis: Wound Reduction in Mice Using N-Methyl Glucamine from PVP and Nano Clay Membranes
Maria Jose Alves de Oliveira1*, Regina Maia2,3, Lucia Almeida Braz3, Ademar Benevolo Lugao1, Valdir Sabbaga Amato2,3 and Duclerc Fernandes Parra1
1Institute of Energy and Nuclear Research (IPEN), Cidade Universitária, Brazil
2Division of Infectious and Parasitic Diseases, São Paulo University School of Medicine, Brazil
3Institute of Tropical Medicine of São Paulo, Brazil
*Corresponding author:
Maria Jose Alves de Oliveira, Nuclear and Energy Reseach Institute, IPEN, CNEN/SP, Avenida Professor Lineu Prestes, 2242, Cidade Universitária, 05508-000, São Paulo-SP, Brazil, E-mail: mariajhho@yahoo.com.br
J Dermatol Res Ther, JDRT-2-036, (Volume 2, Issue 5), Original Article; ISSN: 2469-5750
Received: September 06, 2016 | Accepted: October 21, 2016 | Published: October 24, 2016
Citation: de Oliveira MJA, Maia R, Braz LA, Lugao AB, Amato VS, et al. (2016) Topical Treatment of Cutaneous Leishmaniasis: Wound Reduction in Mice Using N-Methyl Glucamine from PVP and Nano Clay Membranes. J Dermatol Res Ther 2:036. 10.23937/2469-5750/1510036
Copyright: © 2016 de Oliveira MJA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Biomaterials comprise a scientific field where synthetic materials are developed for pertinent biological functions, yielding a wide diversity of new products. Although widely employed, they still require improvement for therapeutic purposes. The objective of this study was to develop poly (N-2-vinyl-pyrrolidone) (PVP), poly (vinyl alcohol) (PVAl), clay, and N-methyl glucamine hydrogels for the treatment of cutaneous leishmaniasis. The characterization techniques used were tumescence, energy-dispersive X-ray (EDX) spectroscopy. Clay and antimoniate were found to disperse homogeneously in the polymeric matrix. Evaluation of cytotoxicity showed negative result. The in vivo tests used 12 BALB/c infected with 3 × 104 amastigotes/mL of L. (L.) amazonensis. All mice (4/4) from group II presented reduction of diameter wounds after submitted to PVP, PVAl, clay (1,5%), and N-methyl glucamine (2.3 mg) hydrogels, differently from group I PVP, PVAl, and N-methyl glucamine (1 mg) and III (control). Classified as results of a clinical cure, drug release was efficient and such a matrix is a potential contributor to an alternative human therapy capable of increasing the quality of life of a patient who cannot use medication in the conventional way because of side effects.
Keywords
Biomaterials, Cutaneous leishmaniasis, N-methyl glucamine, Hydrogels
Introduction
The global biomaterials market is estimated to move $28 billion each year, with an annual growth rate of 15% expected for the next few years. In these sense biomaterials are relevant importance for research and innovation. Although widely employed, they still require improvement and knowledge for therapeutic application purposes. Nevertheless, biomaterials significantly improve the quality of life of millions of people who, otherwise, would be condemned to limitations in the performance of activities or yet, ultimately, to death [1-3]. Prominent among these biomaterials are the nanogels, microgels, and macrogels making up the polymeric hydrogels, which, as drug carriers, have been the targets of research. Poly (vinyl alcohol) (PVAl) or poly (N-2-vinyl-pyrrolidone) (PVP) hydrogel has been prepared by using γ-irradiation and can be used as a biomaterial [4,5].
Besides the ionizing radiation process for modifying polymers, recently there has been the introduction of inorganic nanoparticles for obtain polymeric nanocomposites [6]. Especially the latter with both natural and synthetic clays have aroused several academic interests in the feasibility of new materials, including hydrogels [7]. These inorganic nanoparticles represent a rational alternative loading conventional polymers efficiently for example, in small percentage of clay polymers have improved their mechanical properties, greater thermal stability, and low permeability to gas [8].
Hydrogels may be differentiated in terms of physical and chemical interactions according to their crosslinks. They are used to release drugs and treat wounds and for dermatological purposes [9]. Usually remains in swollen state for specific applications [10]. Their composition supports until 80-90% of water [4]. If subjected to swelling, the volume may increase up to 300% in water or biological fluids without their structures breaking up or their physical properties changing. Swelling is essential feature for hydrogels both when used on dry wounds, as is the case with cutaneous leishmaniasis (CL), and when applied to wounds with exudate, because the hydrogels provide the conditions for controlling tumescence. The wound caused by CL has characteristics which differ from those of common wounds sourced by fungi and bacteria. Therapies for leishmaniasis are far from ideal and thus the development of new therapeutic strategies to treat CL has become a high priority [11]. This study has innovated by offering an alternative treatment of topical use for wounds caused by CL. This therapy controls the drug dose released into the bloodstream, thereby decreasing the cytotoxicity induced by the injectable dose.
American tegumentary leishmaniasis is caused by a protozoan of the Leishmania genus transmitted by a sand fly of the Phlebotomus subfamily [12]. It is a zoonosis regarded as a great public health problem and it represents a disease complex with a major clinical spectrum and epidemiological diversity. The World Health Organization estimates that 350 million people are at risk and there are approximately two million new cases every year of different forms of the disease: clinical, cutaneous, mucocutaneous, and visceral. No topical treatment, such as antibiotics or anti-inflammatory can be administered; the lesions must be washed with water. The patient with Leishmania-induced wounds along with renal or cardiac dysfunction may not undergo antimoniate - based treatments (N-methyl glucamine) - the wounds remain without dressing - due to the toxicity of the drug injected directly into the bloodstream. Given the complexity of the treatment for CL-engendered lesions, a proposal has come up to provide an alternative form of treatment by using a radiation-modified polymeric matrix as a vehicle for releasing antimoniate directly to the wound site. The matrix is a smooth, humid hydrogel which releases the drug slowly, thereby keeping the concentration of antimoniate in the bloodstream low and thus preventing a toxic buildup, harmful to the patient. Topical treatments using glucantime from PVP membranes to cure wounds caused by leishmaniasis supports our research of alternative treatment. BALB/c mice is a good model for cutaneous leishmaniasis infection [13]. The present study, therefore, assessed the drug action (PVP, PVAl, with or without clay (1.5%) and N-methyl glucamine (1-2.3 mg) carried in a hydrogel, in BALB/c mice infected with L. amazonensis. This experimental murine model was chosen because it is highly susceptible to the development of characteristic chronic lesions induced by the parasite [14].
Methods and Materials
Obtaining a hydrogel membrane and adding N-methyl glucamine
The reagents 10% PVP, 10% PVA1, 1.5% agar, and 0.5%, 1.0%, and 1.5% clay were solubilized in water and homogenized for 40 minutes. Next, under further homogenization, they were heated to 85 °C for 5 minutes. Then 1-2.3 mg of N-methyl glucamine was added to each dressing. The solution was placed on a pad, sealed, and sent to crosslinking and sterilization by ionizing radiation process with a 60 Co gamma source at dose of 25 kGy at 5 kGy h-1.
Swelling
After synthesis, the three samples were immersed in distilled water and weighed in periods of time until 60 h. The swelling was calculated according to the equation (A).
Swelling = (ms-md)/md*100
Where, ms is the mass of swelled polymer and md is the mass of the hydrogel.
Cytotoxicity
The sample extract obtained by immersion in cell culture medium (minimum Eagle's medium, Sigma Co. São Paulo, Brazil) during 24 h at 37 °C and serially diluted was exposed to NCTC L929 cell line, from mouse connective tissue purchased from American type culture collection (ATCC) tissue bank, cultured on 96 wells microplate. The cytotoxicity effect was evaluated by the cellular viability via the viable cells capacity of uptake neutral red and intracellular incorporation measured in spectrophotometer at 540 nm filter in the end of the assay.
Negative and positive controls are used to determine the reference limits of the results. Negative control (no toxic compound) used was HDPE (high density polyethylene) and positive control was natural rubber latex film, which causes toxic effect to the cells.
The cell viability percentage is plotted against extract concentration and the final graphic corresponds to the viability curves of each sample and controls. Sample with viability curve above 50% cell viability line is considered no cytotoxicity, as the negative control result. If viability curve is under or cross the 50% viability line is considered cytotoxicity, as the positive control. In the intersection is obtained the cytotoxicity index (IC50%), which is the extract concentration that injures or kills 50% of cell population in the assay.
Energy-dispersive X-ray (EDX) spectroscopy
Fracture surface of the hydrogel films were analyzed using the EDS technique for identification of ions. Identification of ions, mapping the distribution of chemical elements and the measurement of ions were performed using the EDS Hitachi Model TM -3000 SEM device.
Drug delivery in vitro
The drug delivery in vitro was done in triplicate from a membrane sample. The samples from hydrogels with a concentration of the drug were put on flasks with 40 mL of water. They were homogenized at 100 rpm, in 37 °C, during 48 h. After 3, 6, 9 12, 24, 36 and 48 h an aliquot of 2 ml was taken out for analyzing antimonials concentration. This aliquot was substitute for 2 mL of water.
Amastigotes determination for mice infection
Using a Neubauer chamber were quantified 104-105 parasites. It was sufficient for the infection of the BALB/c.
Mice infection
The project was submitted to the Ethic Committee of Instituto de Medicina Tropical - USP (CPE-IMT 2010/072). The animal care and handling were carried out according to the guidelines issued by the World Health Organization.
Ten BALB/c mice aged 6 to 10 weeks were used for intradermal (id) inoculation in the foot with 3 × 104 amastigotes/mL of L. (L.) amazonensis (strain L. amazonensis MHOM/BR/73/M2269). The age and number of animals were chosen according to the literature [12]. Four animals were used in each group as follows: Group I, 4 mice treated with PVA1/PVP/ N-methyl glucamine 1 mg; Group II, 4 mice treated with PVA1/PVP/clay/ N-methyl glucamine 2,3 mg; and Group III, 2 animals used for infection control. The membranes (PVA1/PVP/clay/ N-methyl glucamine or PVA1/PVP/ N-methyl glucamine) were placed on the cutaneous lesions for 48 hours and then taken off for 24 hours. This alternating procedure was maintained for 60 days. After this period 100 fields were examined microscopically (1000X) stained smears (Giemsa) from the foot of the control and treated mice.
Results
Swelling
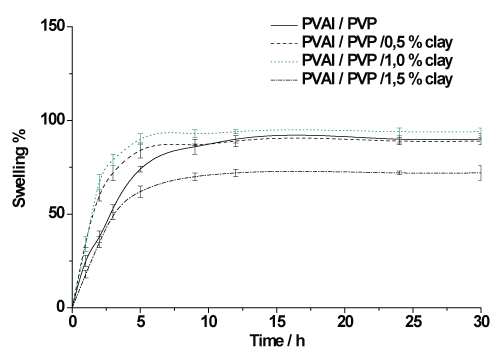
The PVAl/PVP and the PVAl/PVP 0.5% clay hydrogel membranes had similar swelling curves, while the PVAl/ PVP1% clay matrix showed higher swelling in figure 1. It can be seen that smaller clay amounts favor swelling when compared to the PVAl/PVP 1.5% clay matrix, which shows less swelling. It may also be observed that the time taken for the matrices to reach swelling equilibrium was 10 hours.
Energy-dispersive X-ray (EDX) spectroscopy
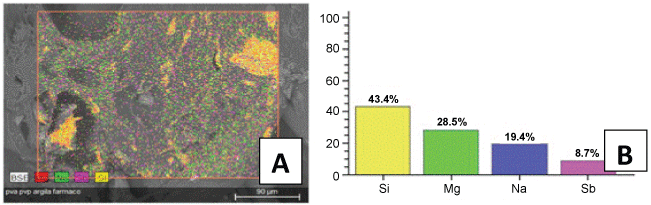
Identification and quantitation of sodium, magnesium, silicon, and antimoniate can be observed in EDS results at figure 2. In some regions of the sample was observed larger clay clusters. Antimoniate distribution was homogeneous on the fracture surface of the hydrogel sample.
.
Figure 2: (A) Micrograph of EDX; (B) graph with the percentage of ions Si, Mg, Sb, and Na dispersed in the PVAl/PVP/ 1.5% clay and N-methyl glucamine hydrogel membranes.
View Figure 2
Cytotoxicity
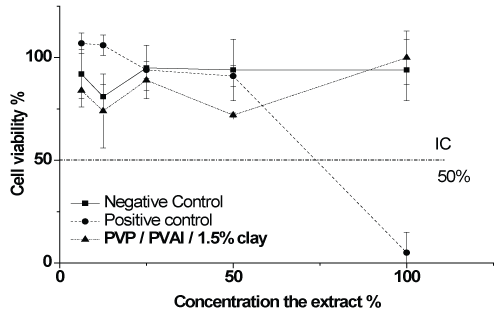
The cytotoxicity tests, in figure 3, showed identical results for samples and negative control. Therefore, the hydrogels synthetized by gamma crosslinking in this study do not harm the mammalian cells and in consequence they were characterized as noncytotoxicity.
.
Figure 3: Cell viability curve in the cytotoxicity assay by the neutral red incorporation method of the hydrogel membrane obtained from in vitro PVAl/PVP/clay tests.
View Figure 3
In vitro drug delivery
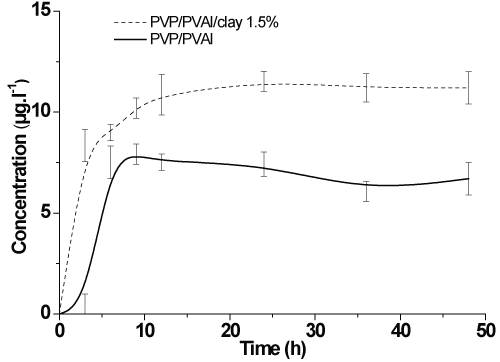
Figure 4 shows that, after 15 hours, the release of PVR/PVA/1.5% clay is steady, at higher level of release than that of the PVP/PVA1 system.
.
Figure 4: Release of N-methyl glucamine from the PVP/PVAl/1.5% clay and PVP/PVAl hydrogels.
View Figure 4
In vivo release of drug and reduction in the wounds
The curative membranes were applied on the foot lesions of the mice, which appeared 30 to 40 days after inoculation. As seen in figure 5, there was a lesion reduction in the treated animal B (Group II), comparatively with the animal A, control (Group III). The observed diameter of wounds showed results of the clinical cure, table 1.

.
Figure 5: Foot wound in the mouse A, from group III (controls without treatment), vs. wound reduction in the mouse B, from group II (treatment with PVA1/PVP/1.5% clay/ N-methyl glucamine) and C from group I (treatment with PVA1/PVP/N-methyl glucamine).
View Figure 5
![]()
Table 1: Composition of the hydrogels and evaluation of wound diameter form the BALB/c tested in vivo.
View Table 1
Discussion
Biomaterials still require improvement for therapeutic purposes like developing of membranes for topical treatment of cutaneous leishmaniasis.
About the procedure for preparation of PVP membrane used it can be seen that smaller amounts of clay promote tumescence, since the PVA1/PVP/1.5% clay matrix curve, which is the least swollen, is set against the other swelling curves. The polymer performance with respect to the mobility of molecular chains is related to the functional groups present in the chains. According to Oliveira [15], the addition of particles with positive or negative charges tends to increase swelling, for it strengthens affinity with the aqueous solution. In general, the sustainability of hydrogel tumescence is dependent on the rearrangement of molecules after crosslinking and clay-polymer interaction. The cytotoxicity behavior of the hydrogel matrix was similar to that found in previously published studies, which also assessed the in vitro cytotoxicity of PVP hydrogels [16,17]; see figure 2. Clay macrodispersion by EDX spectroscopy was used to observe the composition of clay ions on the surface of samples. As silicon is the basis of silicates (clays), its percentage is the highest all throughout the sample. The homogenous distribution of antimoniate on the sample surface points to a contact between glucantime and the wound, as can be seen in figure 2A. The release of compounds, possibly influenced by the hydrophobicity on the clay surface as well as the amount of water added to the formula, is what causes the wound to heal [1]. According to Ikada [18] the area available for diffusion of the solute is the free space which exists between the macromolecular chains. The more a hydrogel is cross-linked, the smaller the free spaces become. When these are filled with water or biological fluids, they swell to equilibrium, the molecular chains lengthen, and the smaller particles diffuse out of the hydrogel matrix. Hydrogels are used to release drugs and treat wounds and for dermatological purposes. In the field of nanoparticles [19] reported that a suitable strategy for the treatment of chronic lesions consists of the topical administration of rhEGF - loaded lipid nanoparticles (recombinant human epidermal growth factor), namely solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC).
The necessity for topical treatment has occurred because the patient with leishmanial - caused lesions and with renal or cardiac dysfunctions may not undergo an antimoniate - based treatment (N-methyl glucamine) due to the toxicity of the drug injected directly into the bloodstream. The patient wounds are thus not treated. When N-methyl glucamine is not effective in treating leishmaniasis, amphotericin B (AmB) is the second drug of choice, but it has harmful side effects as well [13]. Administered a topical treatment for CL in mice with cantharidin ointment they demonstrated an effective and simple method for treating CL caused by L. tropical. Cantharidin may cause pain, tingling, itching, or a burning sensation for a few hours. In our study the therapeutic progress observed with the wound dressings demonstrated the applicability of hydrogel membranes containing encapsulated drugs in the topical treatment of leishmaniasis in accordance with the in vivo test results. Also, [20] in their work conducted with topical applications of lipid-based formulas containing AmB, showed preliminary results attesting to the significant therapeutic effect of the formulations on CL wounds in adults.
Conclusions
Cutaneous lesions engendered by Leishmania amazonensis were treated by the application of hydrogel membranes in the form of in vivo wound dressings. The wound dresses have evidently contributed to wound reduction and the achievement of clinical cure in BALB/c mice. This study is a potential support for further research into alternative human therapies with the purpose of increasing the quality of life of the patient who cannot use medication in the conventional way because of side effects. Additionally, there is the benefit of lower public health costs.
Acknowledgments
We would like to express our gratitude to the following institutions: CAPES, FAPESP, CNPq, Dr. Pablo Vasquez and Paulo Silva (CTR-IPEN-CNEN/SP) for irradiation, IMT/USP and Dr. Sizue Rogero for citotoxicity tests.
References
-
Holzapfel BM, Reichert JC, Schantz JT, Gbureck U, Rackwitz L, et al. (2013) How smart do biomaterials need to be? A translational science and clinical point of view. Adv Drug Deliv Rev 65: 581-603.
-
Onuki Y, Bhardwaj U, Papadimitrakopoulos F, Burgess DJ (2008) A review of the biocompatibility of implantable devices: current challenges to overcome foreign body response. J Diabetes Sci Technol 2: 1003-1015.
-
Centro de Gestão e Estudos Estratégicos (2010) Materiais Avançados para Saúde Médiaca-odontológica. Ciência, Tecnologia e Inovação 15: 297-331.
-
ABD El-Mohdy HL, Ghanem S (2009) Biodegradability, antimicrobial activy and properties of PVA/PVP hydrogels prepared by γ-irradiation. J Polym Res 16: 1-10.
-
Amaral RH, Sizue RO, Cruz ÁS, Roger JRO, Lugão AB (2016) Immobilization and release from hydrogel membranes, COLOAB. The Latin American Congress of Artificial Organs and Biomaterials 1-12.
-
Oliveira MJA, Silva EO, Braz LM, Maia R, Amato VS, et al. (2014) Influence of chitosan/clay in drug delivery of glucantime from PVP membranes. Radiat Phys Chem 94: 194-198.
-
Lee JH, Siddaramaiah, Li P, Kim H, Yoo GH (2008) Poly(acrylamide/laponite) nanocomposite hydrogels: Swelling and cationic dye adsorption properties, J Applied Polym Sci 111: 1786-1798.
-
Fonseca MG, Airoldi C (2003) Híbridos inorgãnicos-Orgânicos derivados da reação de filossilicatos com organossilanos. Quím Nova 26: 5.
-
Fernández F, Ciurlizza C, Lázaro R, Parra A, Clares B (2014) Semisolid formulations containing cetirizine: human skin permeation and topical antihistaminic evaluation in a rabbit model, Arch Dermatol Res 306: 711-717.
-
Okrasa L, Kozanecki M, Boiteux G, Rosiak J, Ulanski J, et al. (2007) Molecular relaxations in radiationally crosslinked poly(vinyl methyl ether) hydrogels. J Non-Cryst Solids 353: 47-51.
-
de Menezes JP, Guedes CE, Petersen AL, Fraga DB, Veras PS (2015) Advances in Development of New Treatment for Leishmaniasis. Biomed Res Int 2015: 815023.
-
Tuon FF, Neto VA, Amato VS (2008) Leishmania: origin, evolution and future since the Precambrian. FEMS Immunol Med Microbiol 54: 158-166.
-
Ghaffarifar F (2010) Leishmania major: in vitro and in vivo anti-leishmanial effect of cantharidin. Exp Parasitol 126: 126-129.
-
Lopes CT, de Paula DM, Cury PM, Valero-Lapchik VB, Bueno V (2010) Leishmania (Leishmania) amazonensis infection in mice treated with FTY720. Transplant Proc 42: 578-581.
-
Oliveira MJA, Parra DF, Amato VS, Lugão AB (2013) Hydrogel membranes of PVAl/ clay by, gamma radiation. Radiat Phys Chem 84: 111-114.
-
Lugão AB, Higa OZ, Rogero SO, Machado LDB, Mathor MB (1999) Biocompatibility study for PVP wound dressing obtained in different conditions. Radiat Phys Chem 55: 705-707.
-
Lopérgolo LC, Lugão AB, Catalani LH (2003) Direct UV photocrosslinking of poly(N-vinyl-2-pyrrolidone) (PVP) to produce hydrogels. Polym 44: 6217-6222.
-
Burczak K, Fujisato T, Hatada M, Ikada Y (1994) Protein permeation through poly(vinyl alcohol) hydrogel membranes. Biomaterials 15: 231-238.
-
Gainza G, Pastor M, Aguirre JJ, Villullas S, Pedraz JL, et al. (2014) A novel strategy for the treatment of chronic wounds based on the topical administration of rhEGF-loaded lipid nanoparticles: In vitro bioactivity and in vivo effectiveness in healing-impaired db/db mice. J Control Release 185: 51-61.
-
Layegh P, Rajabi O, Jafari MR, Emamgholi Tabar Malekshah P, Moghiman T, et al. (2011) Efficacy of Topical Liposomal Amphotericin B versus Intralesional Meglumine Antimoniate (Glucantime) in the Treatment of Cutaneous Leishmaniasis. J Parasitol Res 2011: 656523.