Journal of Dermatology Research and Therapy
Imatinib-Induced Erythema Nodosum : A Case Report and Literature Review
Hanane Rachadi*, Nadia Ismaili and Badreddine Hassam
Department of Dermatology, Ibn Sina hospital, Rabat, Morocco
*Corresponding author:
Hanane Rachadi, Department of Dermatology, Ibn Sina hospital, Rabat, Morocco, Tel: 0021-2668-275106, E-mail: hananerachadi@gmail.com
J Dermatol Res Ther, JDRT-2-030, (Volume 2, Issue 4), Case Report; ISSN: 2469-5750
Received: June 24, 2016 | Accepted: July 18, 2016 | Published: July 20, 2016
Citation: Rachadi H, Ismaili N, Hassam B (2016) Imatinib-Induced Erythema Nodosum : A Case Report and Literature Review. J Dermatol Res Ther 2:030. 10.23937/2469-5750/1510030
Copyright: © 2016 Rachadi H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The list of drugs inducing an erythema nodosum is long and is extending. Their accountability is difficult to ascertain; rare observations relate positive drug provocation tests.
Observation: We report a case of subintrant push of erythema nodosum caused by the Imatinib (Glivec) administration in a patient aged of 34 years old and followed for chronic myeloid leukemia. The drug provocation test was positive. In front of the recurrent, the painful character, the significant functional deficiency caused by the lesions and the positive drug provocation test, the imatinib was finally stopped. The evolution was characterized by the absence of recurrence 2 years after stopping the treatment.
Conclusion: Even if it is exceptional, the Imatinib may induce severe erythema nodosum imposing its stopping. Such side effects should be reported to improve their monitoring and treatment.
Keywords
Erythema nodosum, Imatinib, Drug provocation test
Introduction
The list of drugs inducing erythema nodosum (EN) is long and extending. Their accountability is difficult to ascertain, rare observations relate positive provocation tests. The Imatinib is a chemotherapeutic means used to treat many diseases, including chronic myelogenous leukemia. Many skin reactions to this drug have been described in the literature, however, the EN caused by its use is rare. We report a case of EN induced by the Imatinib with a positive drug provocation test.
Case Report
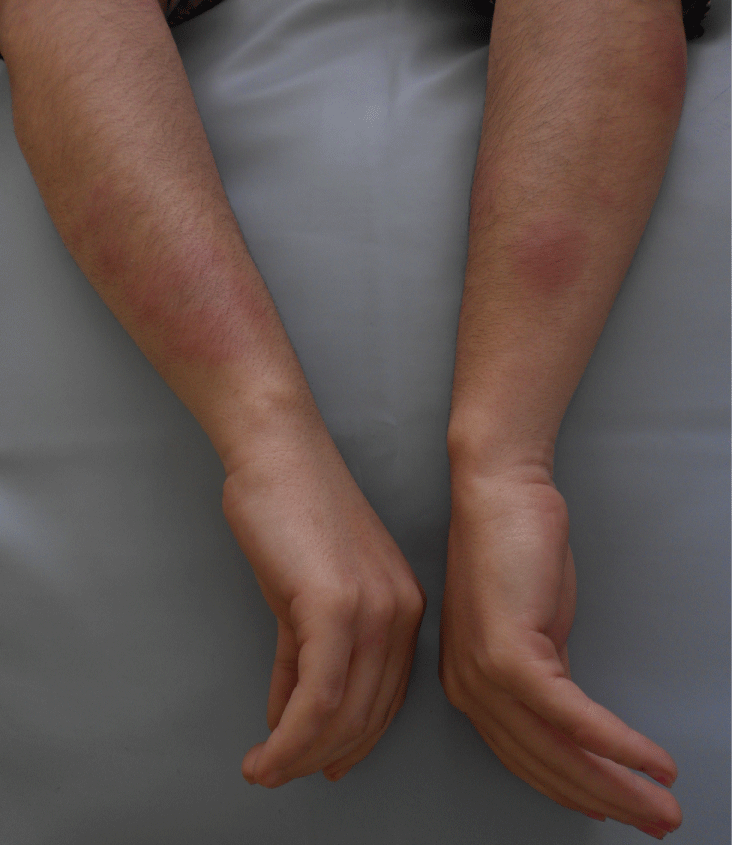
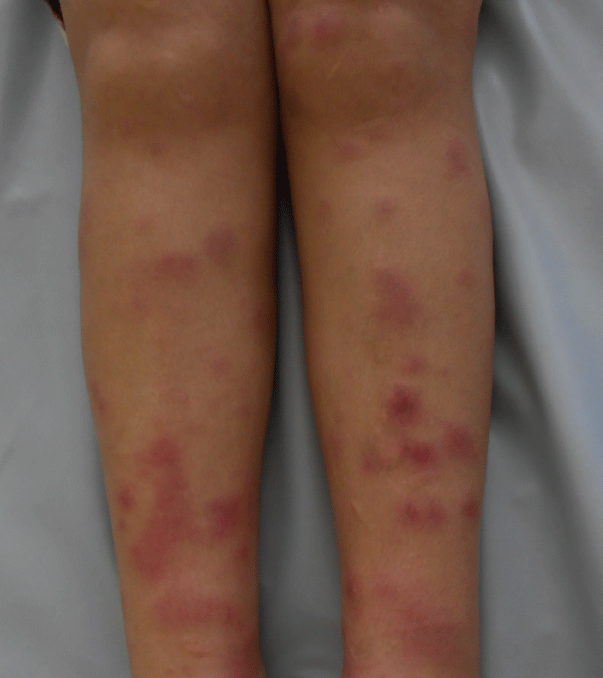
Ms. N.B, 34 years old, followed because of chronic myeloid leukemia, takes Imatinib (Glivec) 400 mg/day with a good clinical and hematologic reaction. The patient presented, one month after the start of the treatment with Imatinib, several erythematous and painful nodules, associated with arthralgia, in the upper and lower limbs,. The symptoms evolved by subintrant pushes that last 2 to 4 weeks without leaving any residual pigmentation. Her clinical examination at the admission showed various erythematous nodules embedded in the dermis, with variable size, from 1 to 3 cm, well limited, hot and painful spontaneously and on palpation, located in the upper limbs (Figure 1) and lower limbs (Figure 2). The skin biopsy revealed a mostly septal panniculitis without vasculitis, consistent with EN (Figure 3 and Figure 4). The erythrocyte sedimentation rate was augmented; the number of leukocytes was raised with neutrophilia and hyperfibrinosaemia. The Streptolysines antibodies anti-O, the C-reactive protein, the tuberculin skin test, the thorax radiography, the transaminases, the serological tests for viral hepatitis B and C and the parasitological examinations of feces were normal. The diagnosis of the EN induced by the Imatinib was strongly suspected. The patient was put under single oral prednisone (60 mg/day) then associated with azathioprine (100 mg/day) but this treatment decreased only moderately the symptoms. In front of the recurrence, the painful characteristics of the lesions and the significant functional deficiency, our decision was to stop the Imatinib and make a provocation test. After stopping the Imatinib for 3 months, it has been reintroduced in our patient. During the period of stopping the treatment, the patient didn’t present new lesion, whereas the retaking of the Imatinib had led to pushes of the EN a week after the start of the treatment. So, we conclude to positive drug provocation test. The final stopping of the Imatinib and its substitution by Nilotinib were decided. The evolution was marked by the absence of recurrence 2 years after stopping the Imatinib.
.
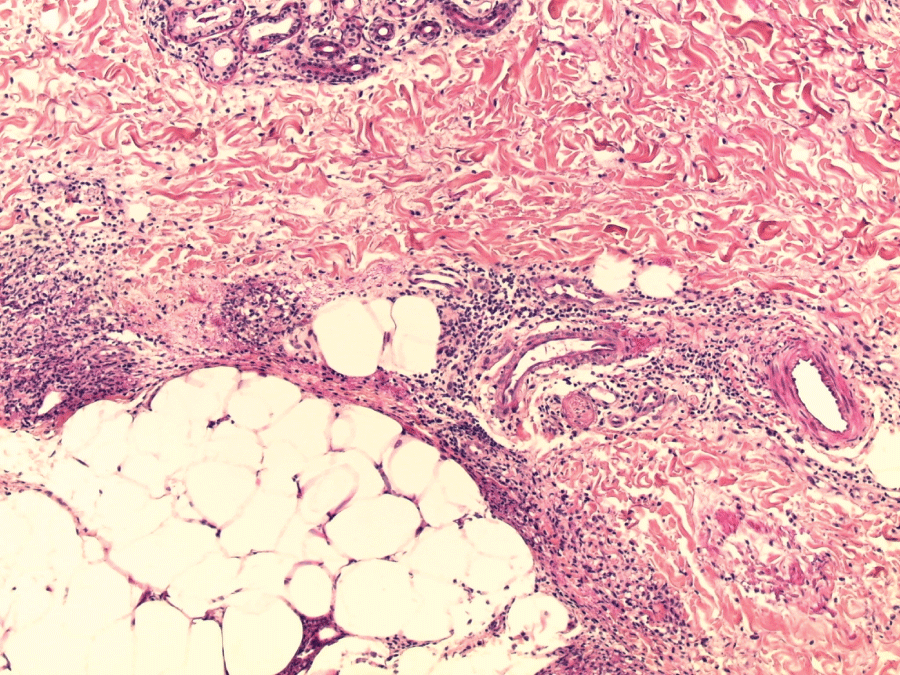
Figure 3: Inflammation of the septa between intact fat lobules (skin histology : H&E stain; ×100).
View Figure 3
.
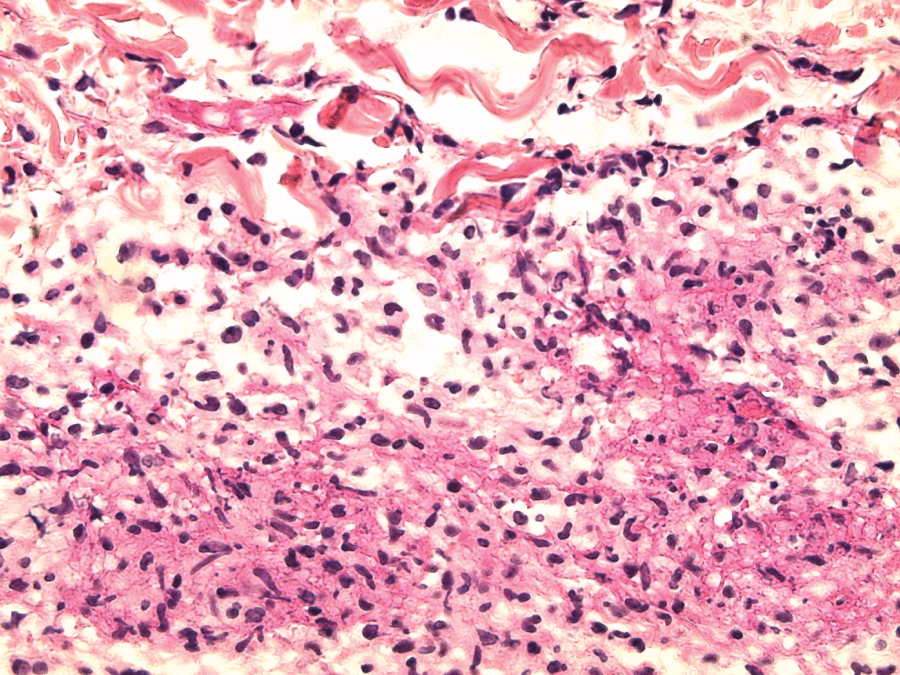
Figure 4: Septal inflammatory infiltrate containing lymphocytes, histiocytes, polynuclear neutrophils nuclear debris and fibrin (skin histology: H&E stain; ×400).
View Figure 4
Discussion
The EN is the most frequent and the best individualized among the panniculitides. The pathogenesis of EN is not fully elucidated. It is likely a delayed hypersensitivity reaction, which can be triggered by a variety of infectious and non-infectious antigenic stimuli including drugs like it would be the case with your patient [1].
The EN is clinically translated by the unexpected appearance of painful, warm, erythematous, slightly raised nodules, measuring one to five centimeters in diameter and may coalesce to form plaques. Usually, these lesions undergo a bruise-like transformation and disappear after two to eight weeks without leaving scars [2,3].
Habitually, the lesions reach bilaterally and symmetrically the extension faces of the lower limbs. However, all body parts can be affected as the ankles, the thighs, the forearms, the face, the neck and the trunk [4,5].
Nevertheless the etiology, the nonspecific symptoms may occur. They include: malaise, low-grade fever, and arthralgia with or without arthritis [6].
The diagnosis of the EN is usually clinical. The skin biopsy is not required in typical forms; when we performed it, it objectives, habitually, septal panniculitis without vasculitis [7,8].
The etiologies of the EN are numerous including: infections, malignancies, autoimmune diseases, systemic illnesses, drugs, and pregnancy [9]. Our patient had two conditions that can induce the EN: the leukemia and taking the Imatinib, but she had not made any push to the EN before the taking of this drug and the provocation test was positive which advocates its accountability.
The Imatinib is an oral inhibitor of the tyrosine kinase BCR-Abl, c-KIT and platelet-derived growth factor receptor, successfully used in the treatment of several diseases such as chronic lymphocytic leukemia, gastrointestinal stromal tumor, hypereosinophilic syndrome, chronic myelomonocytic leukemia, systemic mastocytosis, dermatofibrosarcoma protuberans and melanoma.
Despite its enormous therapeutic effects, the Imatinib has very frequent side effects including digestive disorders (nausea, vomiting, diarrhea), asthenia, myalgia, cramps, fluid retention and lowered resistance to infection (due to neutropenia) [10].
Dermatological side effects of the Imatinib have also been very frequent such as macular-papular rash, superficial edema, lichenoid reactions, psoriasis and psoriasiform rash, pityriasisrosea-like rash, acute generalized exanthematous pustulosis, stevens-johnson syndrome, neutrophilic dermatosis, toxic epidermal necrolysis, photosensitivity, fungoid-like mycosis, cutaneous porphyria cutanea tarda, pseudoporphyria and skin fragility and blistering [11].
Only one case of Imatinib-induced EN has-been described in the literature by Drummond et al. This was a patient with tender subcutaneous nodules that were clinically and histologically consistent with EN, not controlled with oral prednisilone and azathioprine, ultimately required stop of the therapy [12].
Our patient had a severe form of E.N, recurrent and moderately relieved by symptomatic treatment, hence, the decision to substitute the Imatinib by another treatment, considered as effective but less provider of severe adverse effects.
Conclusion
Even if it is exceptional, the Imatinib may induce severe EN imposing its stopping and its substitution by another therapeutic alternative.
References
-
Blake T, Manahan M, Rodins K (2014) Erythema nodosum - a review of an uncommon panniculitis. Dermatol Online J 20: 22376.
-
Bohn S, Buchner S, Itin P (1997) Erythema nodosum: 112 cases. Epidemiology, clinical aspects and histopathology. Swiss Med Weekly 127: 1168-1176.
-
Requena L, Yus ES (2008) Erythema nodosum. Dermatol Clin 26: 425-438.
-
Mana J, Marcoval J (2007) Erythema nodosum. Clin Dermatol 25: 288-294.
-
Brodell RT, Mehrabi D (2000) Underlying causes of erythema nodosum: lesions may provide clue to systemic disease. Postgrad Med 108: 147-149.
-
Mert A, Ozaras R, Tabak F, Pekmezci S, Demirkesen C, et al. (2004) Erythema nodosum: an experience of 10 years. Scand J Infect Dis 36: 424-427.
-
Requena L, Sánchez Yus E (2001) Panniculitis. Part II. Mostly septal panniculitis. J Am Acad Dermatol 45: 325-361.
-
Thurber S, Kohler S (2006) Histopathologic spectrum of erythema nodosum. J Cutan Pathol 33: 18-26.
-
Acosta KA, Haver MC, Kelly B (2013) Etiology and therapeutic management of erythema nodosum during pregnancy: an update. Am J Clin Dermatol 14: 215-22.
-
Romeo U, Palaia G, Fantozzi PJ, Tenore G, Bosco D (2015) A Rare Case of Melanosis of the Hard Palate Mucosa in a Patient with Chronic Myeloid Leukemia. Case Rep Dent 2015: 817094.
-
Pretel-Irazabal M, Tuneu-Valls A, Ormaechea-Pérez N (2014) Adverse skin effects of imatinib, a tyrosine kinase inhibitor. Actas Dermosifiliogr 105: 655-662.
-
Drummond A, Micallef-Eynaud P, Douglas WS, Hay I, HolyoakeTL, et al. (2003) A spectrum of skin reactions caused by thetyrosine kinase inhibitor imatinib mesylate (STI571, Glivec). Br J Haematol 120: 911-913.