Journal of Dermatology Research and Therapy
Relapsing Non Melanoma Skin Cancer in a Patient with Ulcerative Colitis Exposed to Long-Term Therapy with Thiopurines: Need for Regular Dermatological Screening
Jiménez-García Victoria Alejandra1*, Silla-Prosper María Magdalena2, Argüelles-Arias Federico1, Caunedo-Álvarez Ángel1, Moreno-Ramírez David2 and Herrerías-Gutiérrez Juan Manuel1
1Clinical Management Unit of Gastroenterology, University Hospital Virgen Macarena, Seville, Spain
2Clinical Management Unit of Dermatology, University Hospital Virgen Macarena, Seville, Spain
*Corresponding author:
Dr. Victoria Alejandra Jiménez García, Clinical Management Unit of Gastroenterology, University Hospital Virgen Macarena, Avenida Dr Fedriani 3, 41009 Seville, Spain Tel: +34-955-00-88-01, Fax: 955-00-88-05, E-mail: drajimenezg@hotmail.com
J Dermatol Res Ther, JDRT-2-027, (Volume 2, Issue 3), Case Report; ISSN: 2469-5750
Received: April 13, 2016 | Accepted: June 02, 2016 | Published: June 04, 2016
Citation: Jiménez-García VA, Silla-Prosper MM, Argüelles-Arias F, Caunedo-Álvarez Á, Moreno-Ramírez D, et al. (2016) Relapsing Non Melanoma Skin Cancer in a Patient with Ulcerative Colitis Exposed to Long-Term Therapy with Thiopurines: Need for Regular Dermatological Screening. J Dermatol Res Ther 2:027. 10.23937/2469-5750/1510027
Copyright: © 2016 Jiménez-García VA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
An increase of risk of malignancies such as lymphoma and non-melanoma skin cancer has been reported with the chronic use of immunomodulators in inflammatory bowel disease, particularly thiopurines. A case of a patient with ulcerative colitis after several years in immunosuppressive therapy developing multiple non-melanocytic skin neoplasms is described below.
Keywords
Immunosuppressants, Ulcerative colitis, Malignancies, Nonmelanoma
Introduction
In the management of inflammatory bowel disease (IBD) there are a variety of immunosuppressive drugs that have been shown to achieve control of symptoms and keep patients in remission, which is the primary goal of treatment, thus avoiding the continued use of corticosteroids.
One such Immunomodulators drug is Azathioprine, which is an analog of purines and consequently is incorporated into cellular DNA by the active metabolite, Mercaptopurine. This achieves inhibition of multiple pathways in the biosynthesis of nucleic acids and to a lesser extent causes some "damage" to the DNA through the incorporation of purine thio-analogues. Chronic use of Immunomodulator drugs has been reported to increase the risk of developing some tumors, which in turn increases the risk per se of IBD patients having other malignancies.
Clinical Case
A 78-year-old male farmer (he has worked for over 40 years), frequent smoker with the following personal history: arterial hypertension, cerebrovascular disease, pacemaker since 2010, chronic obstructive pulmonary disease and esophagectomy due to perforation because of a foreign body.
The patient was diagnosed with ulcerative colitis in 1997. Since then, he was treated with Azathioprine 100 mg/24 hours and Mesalazine 3 g with success; however he stopped attending check-ups at the Digestive Department.
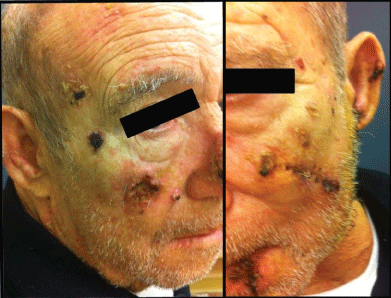
Since 2011 the patient´s case was followed at the department of Dermatology due to keratotic and ulcerated skin lesions in the facial, head region and elbows (Figure 1 and Figure 2). He was prescribed with surgical resection and occasional treatment with cryotherapy on lesions proving to be basal cell carcinomas and squamous.
.
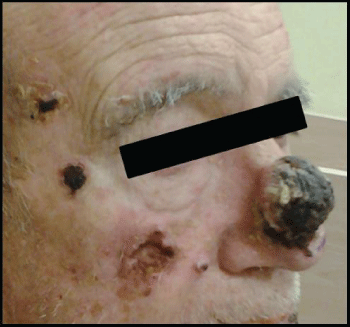
Figure 2: Tumor lesion of 4.5 cm and shaped crater, with large central hyperkeratotic horn fills the nasal dorsum.
View Figure 2
In May 2014 also an exophytic tumor lesion on the dorsum of the nasal pyramid was distinguished (Figure 2). It was only then that his gastroenterologist became involved again and the immunosuppressive therapy (Azathioprine) was stopped immediately. After the withdrawal of Azathioprine the patient presented a mild outbreak which improved with Prednisone. Further surgical resection of the most prominent facial lesion was carried out. The final pathological diagnosis was of squamous cell carcinoma. The patient died five months later due to cardiopulmonary complications.
Discussion
The chronic use of Immunomodulators in IBD is justified to try to keep the patient in remission thus avoiding the use of corticosteroids [1]. However, chronic use of Immunomodulators have been associated with the risk of developing lymphoma and nonmelanoma skin cancer (Table 1) [2-4]. Thus Peyrin et al. [5] have described a higher risk of non-melanoma skin tumors such as squamous cell carcinoma in patients taking these drugs, as indeed in our case.
![]()
Table 1: Risk of cancer with immunomodulator treatment.
View Table 1
In this case several risk factors for developing malignancies were observed, such as: IBD, smoking, continuous sun exposure and chronic use of immunomodulators. In this patient the treatment of skin lesions was removing the involved drug, cryotherapy and surgical resection with good response. Unfortunately, the patient died months later due to underlying conditions.
Therefore, it is important to monitor patients with IBD receiving immunomodulators therapy for skin lesions, especially if they also have additional risk factors like in this case, with a history of smoking and sun exposure. So that, it is of utmost importance to advise patients on Immunomodulator therapy to examine themselves and use sunscreen, avoiding unnecessary UV exposures.
Finally, it is also necessary to share this knowledge to other professionals upon detecting skin lesions associated with the use of Immunomodulators in IBD patients. In this regard an article has recently been published by Okafor et al. [6] concluding that all patients with inflammatory bowel disease with or without immunosuppressants must undergo annual skin cancer screening as a cost-effective intervention.
References
-
Dignass A, Lindsay JO, Sturm A, Windsor A, Colombel JF, et al. (2012) Second European evidence-based consensus on the diagnosis and management of ulcerative colitis part 2: current management. J Crohns Colitis 6: 991-1030.
-
Perrett CM, Walker SL, O'Donovan P, Warwick J, Harwood CA, et al. (2008) Azathioprine treatment photosensitizes human skin to ultraviolet A radiation. Br J Dermatol 159: 198-204.
-
Peyrin-Biroulet L, Khosrotehrani K, Carrat F, Bouvier AM, Chevaux JB, et al. (2011) Increased risk for nonmelanoma skin cancers in patients who receive thiopurines for inflammatory bowel disease. Gastroenterology 141: 1621-1628.
-
Beaugerie L (2013) Use of immunosuppressants and biologicals in patients with previous cancer. Dig Dis 31: 254-259.
-
Peyrin-Biroulet L, Chevaux JB, Bouvier AM, Carrat F, Beaugerie L (2012) Risk of melanoma in patients who receive thiopurines for inflammatory bowel disease is not increased. Am J Gastroenterol 107: 1443-1444.
-
Okafor PN, Stallwood CG, Nguyen L, Sahni D, Wasan SK, et al. (2013) Cost-effectiveness of nonmelanoma skin cancer screening in Crohn's disease patients. Inflamm Bowel Dis 19: 2787-2795.