Journal of Dermatology Research and Therapy
Adjuvant Radiation Therapy is Feasible in Epidermolysis bullosa: A Case Report
Theodora A Koulis1,2*, Colleen Herring2, Wendy Smith2 and Jon-Paul Voroney1,2
1Division of Radiation Oncology, University of Calgary, Calgary, Alberta, Canada
2Tom Baker Cancer Centre, Calgary, Alberta, Canada
*Corresponding author:
Theodora A Koulis, MD, Tom Baker Cancer Centre, 1331 29 Street NW, Calgary, AB, Canada, Tel: 1-403-465-0217, Fax: 1-403-283-1651, E-mail: Theodora.koulis@albertahealthservices.ca
J Dermatol Res Ther, JDRT-1-006, (Volume 1, Issue 1), Case Report; ISSN: 2469-5750
Received: May 27, 2015 | Accepted: August 31, 2015 | Published: September 02, 2015
Citation: Koulis TA, Herring C, Smith W, Jon-Paul V (2015) Adjuvant Radiation Therapy is Feasible in Epidermolysis bullosa: A Case Report. J Dermatol Res Ther 1:006. 10.23937/2469-5750/1510006
Copyright: © 2015 Koulis TA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A patient with a severe form of Epidermolysis Bullosa (EB) presented after an above knee amputation and groin dissection for a 57 cm squamous cell carcinoma (SCC) of the skin and underlying soft tissue of the right leg. Five of 19 nodes were involved with SCC metastases. Standard care would involve adjuvant radiation therapy (RT) but RT has historically been avoided in patients with EB who can have severe, non-healing cutaneous and mucosal morbidity from acute RT reactions. A radiation treatment plan was developed to include the nodal drainage while minimizing dose to the basement membranes of the skin and anal mucosa, where the EB mutation affects structural and cell adhesion proteins. Four non-opposing, conformal beams with mixed energies were used to deliver 48 Gray in 25 daily fractions over 5 weeks. The treatment was well tolerated with minimal acute or late side effects. The patient was recurrence free at last follow-up, 41 months after RT. By understanding the pathophysiology of EB it was possible to deliver adjuvant RT with minimal acute or late morbidity.
Keywords
Epidermolysis Bullosa, Radiation Therapy, Skin cancer, Squamous cell carcinoma
Introduction
Epidermolysis Bullosa (EB) is an inherited disorder characterized by mechanically fragile skin and mucous membranes. It is caused by a range of genetic mutations that affect the anchoring and stability of epithelial cells in the basement membrane zone [1]. This manifests clinically as skin and mucosal blistering and non-healing wounds following minor trauma. Blister formation occurs at different levels in the epithelium depending on the phenotype. EB has been classified into 3 broad categories: Simplex, Junctional, and Dystrophic [1]. EB lesions can be debilitating due to pseudosyndactyly (fusion of digits due to scarring) and strictures in the gastrointestinal and genitourinary tracts, and the upper airway [1-3]. Life-threatening complications include renal failure, sepsis and skin cancers. Patients with severe Dystrophic EB (DEB) phenotype have a median lifespan of 55 years with early death due to skin cancer [1,3]. DEB is caused by mutations in collagen VII which is integral to the attachment of the epithelium to the basement membrane [1].
There is limited data to guide the use of radiation therapy (RT) in patients with EB, especially the severe form of the disease. Primary adjuvant RT in severe EB has not been described. Previous reports or case series have used RT for regional recurrences or for palliation [2,4-7]. They reported no long lasting clinical benefit [2].Other reports have suggested moist desquamation occurs with doses greater than 45 Gray (Gy) with poor healing that takes longer than six weeks. In two case studies, post-operative RT was omitted due to the perceived poor tolerance and healing of tissues [8,9]. Three commonly used Radiation Oncology reference text books [10-12] do not mention EB as a point to consider during RT planning for skin cancer.
This is a report of a patient with the severe form of Recessive Dystrophic Epidermolysis Bullosa (RDEB) and an extensive cutaneous squamous cell carcinoma (SCC) treated with adjuvant regional nodal RT.
Case Description
A 44 year old man with severe manifestations of RDEB presented with an extensive SCC arising in the skin. The tumor eroded into bone and caused a pathologic fracture of the right tibia. Initial treatment was an above knee amputation with inguinal and pelvic lymph node dissection. The pathology revealed a 57 cm; grade 1 SCC with five of 19 lymph nodes positive for metastatic deposits without extra-capsular extension. Resection margins were negative and no lymphovascularor perineural invasion was seen. Final pathologic TNM classification was pT4; pN2b; cM0 [13]. Over the three years prior to diagnosis multiple skin biopsies had been performed that showed severe inflammatory changes but no malignancy and he had received several dermatologic interventions for a non-healing patch over his right patella.
A literature review revealed scant evidence to guide practice. Following a discussion on the potential benefits and harms, the patient opted to have adjuvant regional nodal RT, consistent with institutional policy and international guidelines [14]. The right inguinal and pelvic nodal beds were contoured as the target volume. To account for anticipated toxicity due to the collagen VII gene mutation, the anal mucosa, and the external 5 mm of the skin were contoured as structures for which the dose should be minimized during the radiation planning process (avoidance structure; Figure 1). The 5 mm depth was chosen to generously encompass the basement membrane zone and allow for variation in set up due to movement the prescribed dose was 48 Gy in 25 daily fractions over five weeks. This dose, although slightly lower than recommended by NCCN [14], is higher than what is commonly used to for pelvic RT [10-12] and is appropriate to treat microscopic disease. It also allowed us to meet dose constraints to organs at risk.
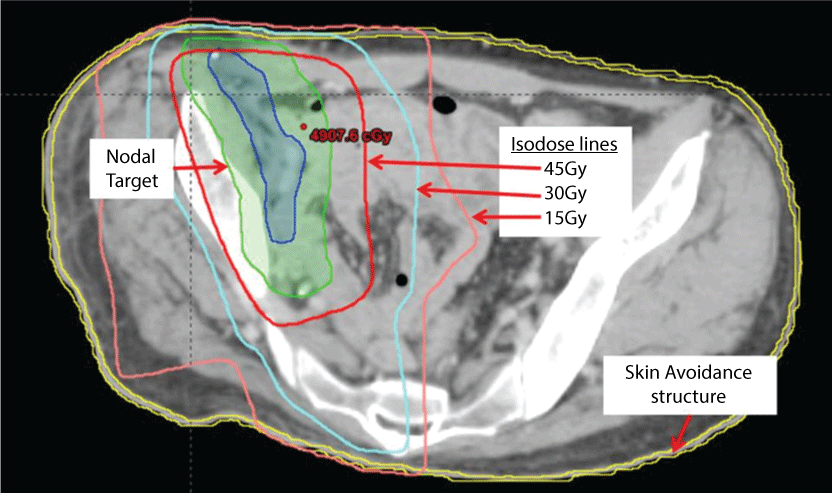
.
Figure 1: Axial CT image from the radiation therapy planning system showing the lymph node target for adjuvant RT (nodal target), the skin and basement membrane contoured as an avoidance structure, and the 15Gy, 30Gy, and 45Gy isodose lines.
View Figure 1
To minimize low dose scatter radiation and to reduce total body dose, a 3-dimensional (3D) conformal, 4 non-opposing fields rather than IMRT (intensity modulated radiation therapy) was used. Mixed 6 and 18 MV energy photons were used to improve skin sparing. Figure 1 shows an axial view of the dose distribution. No part of the skin avoidance structure received a dose greater than 40 Gy. Only 2.7 cm2 of skin received 30 Gy or more and 10.7 cm2 of skin received 15 Gy or more. Treatment commenced 11 weeks after the amputation and nodal dissection due to slow post-operative healing.
The treatment was well-tolerated with acute grade 1 skin toxicity [15] limited to mild in-field erythema and hair loss but no desquamation or new bullae. A small bulla present at the start of treatment did not worsen. The patient was followed every 3 months for the first year and then every 6 months thereafter. History and physical exam were performed at each visit. CT imaging was done at the first follow-up; however, due to the patient's EB, multiple abnormalities unrelated to his malignancy were detected. Imaging was not used for subsequent follow-up visits. At 41 months after RT, the patient remained clinically disease free with a small contracture at the site of the slow-healing bulla (grade 2 late subcutaneous toxicity [16]), a prominent scar, and sparse hair growth (grade 1 late skin toxicity [16]).
Discussion
This report showed that by using 3D conformal RT planning and mixed photon energies, we were able to deliver an effective adjuvant radiation treatment to the target and to reduce dose to the skin avoidance structure. There is little data to guide the use of RT in patients with severe EB. Most of the published reports did not contain a description of the radiation therapy technique or the year of treatment [4-8]. This made it challenging to interpret the reports or the general conclusions that RT was either poorly tolerated or ineffective. The dose employed in this case was within the range of common adjuvant doses used in the pelvis, for example following hysterectomy for high-risk endometrial cancer [10-12]. The maximum dose to the skin avoidance structure was 40 Gy and is less than the previously reported maximum tolerated dose of 45 Gy [7] and likely contributes to the low acute and late skin toxicities.
Conclusion
A man with a severe variant of Epidermolysis Bullosa and a cutaneous SCC with a high risk of regional recurrence received well tolerated adjuvant groin and pelvic nodal RT and achieved long-term local control. Dose was minimized to tissues that require functional collagen VII by using them as avoidance structures. This substantially reduced acute and late toxicity compared to RT adverse effects previously described in the literature.
References
-
Bergman R (1999) Immunohistopathologic diagnosis of epidermolysis bullosa. Am J Dermatopathol 21: 185-192.
-
Fine JD, Johnson LB, Weiner M, Li KP, Suchindran C (2009) Epidermolysis bullosa and the risk of life-threatening cancers: the National EB Registry experience, 1986-2006. J Am Acad Dermatol 60: 203-211.
-
Fine JD (2010) Inherited epidermolysis bullosa: recent basic and clinical advances. Curr Opin Pediatr 22: 453-458.
-
Venugopal SS, Murrell DF (2010) Treatment of skin cancers in epidermolysis bullosa. Dermatol Clin 28: 283-287, ix-x.
-
McGrath JA, Schofield OM, Mayou BJ, McKee PH, Eady RA (1992) Epidermolysis bullosa complicated by squamous cell carcinoma: report of 10 cases. J Cutan Pathol 19: 116-123.
-
Bastin KT, Steeves RA, Richards MJ (1997) Radiation therapy for squamous cell carcinoma in dystrophic epidermolysis bullosa: case reports and literature review. Am J Clin Oncol 20: 55-58.
-
Edland RW (1969) Dystrophic epidermolysis bullosa. Tolerance of the bed and response of multifocal squamous cell carcinomas to ionizing radiation: report of a case. Am J Roentgenol Radium Ther Nucl Med 105: 644-647.
-
Yamada M, Hatta N, Sogo K, Komura K, Hamaguchi Y, et al. (2004) Management of squamous cell carcinoma in a patient with recessive-type epidermolysis bullosa dystrophica. Dermatol Surg 30: 1424-1429.
-
Lataifeh I, Barahmeh S, Amarin Z, Jaradat I (2010) Stage III squamous cell carcinoma of the vulva with groin nodes metastasis in a patient with epidermolysis bullosa. J Obstet Gynaecol 30: 750-752.
-
Halperin EC, Wazer DE, Perez CA, Brady CW (2013) Perez and Brady Principles and Practice of Radiation Oncology. (6th edn), Lippincott Williams & Wilkins, Philadelphia.
-
Gunderson LL, Tepper JE (2012) Gunderson and Tepper Clinical Radiation Oncology. (3rd edn), Elsiver Inc, Philadelphia.
-
Hoppe RT, Phillips TL, Roach, MIII (2010) Leibel and Phillips Text book of Radiation Oncology. (3rd edn), Elsevier Inc, Philadelphia.
-
(2010) Cutaneous Squamous Cell Carncinoma and Other cutaneous Carcinomas. In: Edge SP, Byrd DR, Compton CC, Fritz AG, Green FL, et al. AJCC Cancer Staging Handbook. (7th edn) III American Joint Committee on Cancer, NY: Springer; New York, 359-376.
-
http://www.nccn.org/professionals/physician_gls/f_guidelines.asp#squamous
-
https://www.rtog.org/researchassociates/adverseeventreporting/acuteradiationmorbidityscoringcriteria.aspx
-
https://www.rtog.org/researchassociates/adverseeventreporting/rtogeortclateradiationmorbidityscoringschema.aspx





