International Journal of Diabetes and Clinical Research
DKA in an Adolescent with Established Diagnosis of Type 1 Diabetes
Yael Levy Shraga1,2*, Uri Hamiel2,3 and Orit Pinhas Hamiel1,2,4
1Pediatric Endocrine and Diabetes Unit, Edmond and Lily Safra Children’s Hospital, Sheba Medical Center, Israel
2Sackler School of Medicine, Tel Aviv University, Israel
3Department of Pediatrics, Assaf Harofeh Medical Centre, Israel
4Juvenile Diabetes Center, Maccabi Health Care Services, Israel
*Corresponding author:
Yael Levy Shraga, MD, Pediatric Endocrine and Diabetes Unit, Edmond and Lily Safra Children's Hospital, Sheba Medical Center, Tel Hashomer, Ramat-Gan, 52621, Israel, E-mail: yael.levy.shraga@gmail.com
Int J Diabetes Clin Res, IJDCR-4-067, (Volume 4, Issue 1), Case Report; ISSN: 2377-3634
Received: June 08, 2016 | Accepted: March 06, 2017 | Published: March 08, 2017
Citation: Shraga YL, Hamiel U, Hamiel OP (2017) DKA in an Adolescent with Established Diagnosis of Type 1 Diabetes. Int J Diabetes Clin Res 4:067. 10.23937/2377-3634/1410067
Copyright: © 2017 Shraga YL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Diabetic ketoacidosis (DKA) and its complications are the most common cause of hospitalization, morbidity and mortality in children and adolescents with established diagnosis of type 1 diabetes mellitus (T1DM). The overall incidence of recurrent DKA episodes is estimated as 8 per 100 patient years. Interestingly, almost 60% of recurrent DKA episodes occur in 5% of patients. Precipitating causes of DKA are infectious diseases, non-infectious diseases, and poor adherence to insulin therapy. Even mild infections, such as a cold or sore throat, can lead to DKA in patients with long standing poor glycaemic control. Poor glycaemic control is common during adolescence, secondary to poor compliance, fear of hypoglycemia or intentional insulin omission as a weight loss strategy. We present a case of DKA in a teenage girl with established T1DM and a step-by-step investigation process leading to diagnosis of intentional insulin omission. The identification of intentional insulin omission and the establishment of a comprehensive treatment are extremely important since it may reduce the rate of recurrent DKA as well as the long term complications associated with this disorder.
Introduction
Diabetic ketoacidosis (DKA) and its complications are the most common cause of hospitalization, morbidity and mortality in children with established diagnosis of type 1 diabetes mellitus (T1DM) [1]. The overall incidence of DKA in patients with established T1DM is estimated as 8 per 100 patient years with almost 60% of recurrent DKA episodes occur in 5% of patients [2]. Interestingly, adolescent girls are at highest risk for DKA, with an incidence rate of 12 per 100 patient years [2]. A significant mortality risk is associated with recurrent DKA, with a four-fold increased risk of death in those with recurrent DKA admissions compared with those with a single episode [3].
The assessment of a patient with DKA is often challenging. The signs and symptoms of DKA and of the underlying disease that triggers the evolvement of DKA may cause diagnostic confusion [4]. Hyperpnoea in a patient with DKA suggests respiratory compensation for metabolic acidosis [5]; however, it could be secondary to respiratory infection. Abdominal tenderness is a frequent symptom of DKA, and is correlated with the severity of acidosis; but, it can also be due to an acute event such as appendicitis or pancreatitis that may have precipitated the development of DKA [6]. Similarly, fever can result from dehydration, or be secondary to infection [7]. In patients treated with an insulin pump, technical problems with the infusion set of the pump may lead to interruption of insulin delivery and deficiency of the short-acting insulin, and can rapidly lead to acute decompensation and DKA [8]. Finally, intentional insulin omission as a weight loss strategy may be a precipitating factor [9].
We present a case of DKA in a teenage girl with established T1DM and the step-by-step investigation process leading to diagnosis of the precipitating factor.
Case Presentation
A 16-year-old girl diagnosed with T1DM at age 8 years and currently treated with an insulin pump, presented to the emergency room with nausea, vomiting, malaise and heavy breathing. The patient lost about 6 kg over the last few months and other than some fatigue has no complaints. She lives with her parents and two younger siblings. She is an excellent student and participates in dance class. Initial assessment revealed a somnolent patient with no focal neurological deficit. Body temperature was 100.8 °F, heart rate 133/minute with weak distal pulses, blood pressure 70/56 mmHg, and respiratory rate 30/minute with a Kussmaul pattern. Oxygen saturation was 96% in room air. The patient weighed 56.5 kg, and her height was 168 cm. Skin turgor was decreased, mucous membranes were dry, and the odor of acetone was noted on her breath. The thyroid gland was enlarged. The abdomen was soft, with decreased bowel sounds and diffuse tenderness but without guarding, rebound, or organomegaly.
Laboratory evaluation revealed a serum glucose concentration of 397 mg/dl, arterial pH 6.99, bicarbonate 3.4 mmol/l, arterial pCO2 14 mmHg, sodium 138 mmol/l (143 mmol/l when corrected for hyperglycemia), potassium 3.2 mmol/l, urea 17 mg/dl and lactate 10 mg/dl. The white cell count was 15,000/ml3 with 43% neutrophils, 44% lymphocytes, and 7% eosinophils. The hemoglobin level was 11.3 g/dl. High sensitive C-reactive protein (CRP) was 43 mg/l (normal range 0.08-5.00). Amylase was 42 U/l (normal range 20-90). Urinalysis revealed glucose > 1000 mg/dl and a test for ketones was strongly positive. The HbA1c level was 13.2%. In the presence of leukocytosis and high CRP, a source of infection was investigated. Blood and urine cultures returned negative and chest radiograph showed no evidence of parenchymal, interstitial, or pleural disease.
Since the results were diagnostic of severe DKA, she was admitted to the intensive care unit and treatment was initiated according to DKA protocol with satisfying improvement. Further work up revealed normal thyroid function tests and negative celiac screen. An ACTH test revealed a baseline cortisol level of 238 nmol/l (normal range 138-690), and a 60 minute cortisol level of 580 nmol/l.
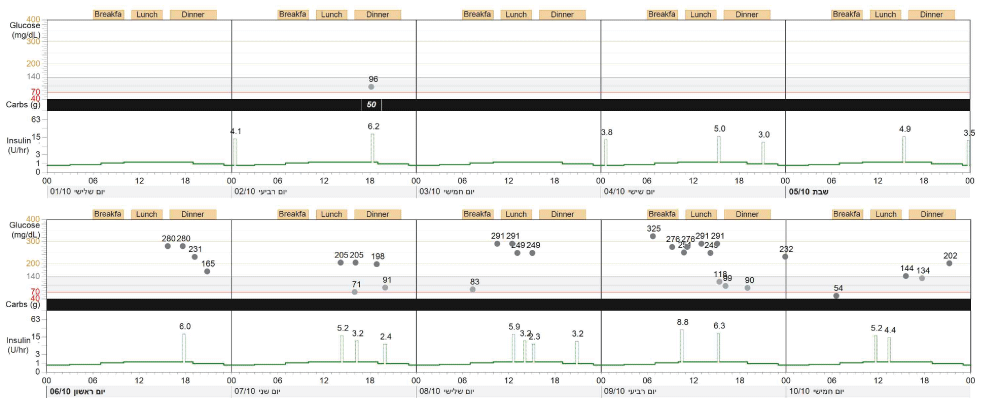
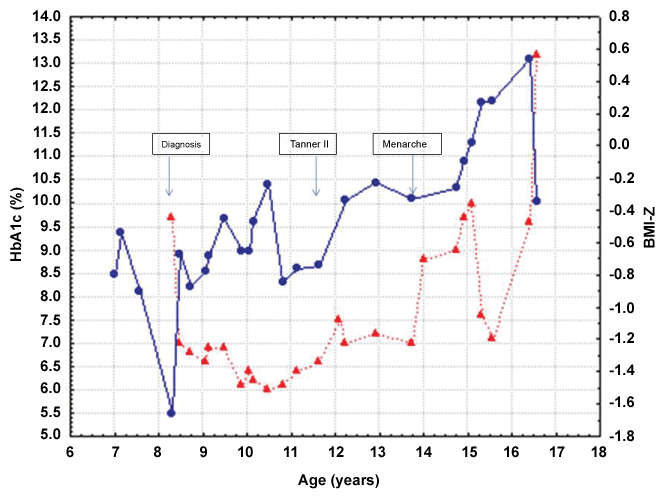
Reviewing the history of daily insulin dose by the pump during the weeks prior to admission revealed that the total daily dose of insulin was 37 units/day (0.65 units/kg/day). There were several days with no boluses prior to meals. Furthermore, there were numerous elevated glucose measurements that clearly were ignored, and no insulin was given for correction (Figure 1). Review of her HbA1c levels revealed an excellent glycaemic control between age 8 and 14 years, and then a gradual deterioration. The marked weight loss prior to the current admission was associated with an increase in HbA1c level as depicted in figure 2. All those raised the suspicion of intentional insulin omission for weight loss. She was referred for psychological evaluation. Low self-esteem related to body dissatisfaction was perceived. Diagnosis was confirmed after she admitted intentional insulin omission.
.
Figure 1: The x-axis shows the time during each day. The grey circles are glucose levels, at the time they were measured. The grey block is the target for glucose levels. The majority of glucose levels are above the target. The green line denotes basal insulin levels during the day. The sharp block indicates the amount of insulin given as a bolus either prior to a meal or for correction of a high glucose level. During some of the days, glucose was not measured, and no bolus insulin was given.
View Figure 1
.
Figure 2: Body mass index Z score (BMI-Z), blue line, and HbA1c levels, red line, throughout the years.
View Figure 2
Discussion
DKA is a medical emergency that requires prompt recognition and appropriate treatment. Careful attention should be given to identification of the precipitating factor, especially in a patient with established diabetes and recurrent episodes of DKA. Precipitating causes of DKA are infectious disease, non-infectious disease, and poor adherence to insulin therapy [10]. Since infection is the leading trigger of DKA [7], a source of infection was investigated. Although there is no proof that individuals with diabetes are more susceptible to infection, they seem to have more difficulty handling infection once it occurs [11]. In patients with long standing poor glycaemic control, even mild infections, such as a cold or sore throat, can lead to DKA. Second, non-infectious diseases should be considered. Patients with T1DM are at increased risk for other autoimmune comorbidities such as celiac, autoimmune thyroid disease and although relatively rare-adrenal insufficiency [12]. Weight loss and abdominal pain can be presenting signs of celiac disease. Thyrotoxicosis in the set-up of an enlarged thyroid gland can be the reason for weight loss and can trigger DKA. Weight loss with fatigue, anaemia, and mild eosinophilia can be attributed to adrenal insufficiency [13]. The evaluation for celiac, thyroid disease and adrenal insufficiency revealed normal results.
The HbA1c level was 13.2% which reflects long standing poor glycaemic control. The BMI-Z score shows a decline prior to the diagnosis of diabetes, and an increase with the onset of insulin treatment. HbA1c levels show a mirror image, an elevation at disease onset followed by an improvement with the initiation of insulin treatment, an excellent glycaemic control between age 8 and 14 years, and then a gradual deterioration. The marked weight loss prior to the current admission was associated with an increase in HbA1c level. These fluctuations of HbA1c levels and the weight together with the history of missed insulin doses by the pump history during the weeks prior to admission raised the suspicion of intentional insulin omission.
Identification of intentional insulin omission for weight loss as the precipitating cause of recurrent DKA is challenging since this behavior is often well hidden and denied. According to the American Psychiatric Association's Diagnostic and Statistical Manual of Mental Disorders fifth edition (DSM-5) [14], intentional insulin omission or reduction for the purpose of preventing weight gain is recognized as either an inappropriate compensatory feature of bulimia nervosa, or as a purging disorder, a component of other specified feeding or eating disorder (OSFED). The onset of insulin omission is typical in adolescence or early adulthood with prevalence that increases with age from 2% among girls aged 9-13 years [15] up to 40% of 18-30 year old [16]. In a study of 770 children and adolescents (age 11-19 years), one-third reported skipping their insulin dose entirely, at least occasionally after overeating [17].
We presented a three level model to describe the development of eating disorder (ED) in adolescents with T1DM [18]. The first level involves the premorbid status, i.e. overweight, low self-esteem, personality characteristics and family dynamics. The second level describes weight loss at disease onset and subsequent weight gain with the initiation of insulin treatment. The third level includes factors associated with the ongoing management of T1DM such as intensive insulin treatment, dietary restraint, hypoglycemic episodes and depression [18]. Clinical signs that should alert health care providers to the possibility of an ED in T1DM patients include preoccupation with appearance, frequently missed medical appointments, poor glycaemic control, and recurrent episodes of DKA [19,20]. Specific surveys designed for patients with diabetes may be used to identify at risk patients [21]. Using data mining methods we developed a clinical prediction model for the detection of intentional insulin omission for weight loss in adolescent girls with T1DM [22]. According to this model adolescent girls with intentional insulin omission were discriminated by HbA1c > 9.2% and more than 20% of HbA1c measurements above the 90th percentile [22].
In addition to recurrent episodes of DKA, insulin omission increases the risk of long-term complications such as retinopathy and nephropathy [23]. In a 4 year follow-up study, 86% of girls with T1DM and severe ED developed retinopathy, 3.6 fold more than girls without an ED [24]. In another study, 25% of women aged 13-60 years, who reported insulin omission had nephropathy, compared with 10% of those who did not [25]. The mortality rate in girls with both diabetes and an ED was assessed as 34.6 per 1,000 person years compared to 2.2 per 1,000 person years in girls with only diabetes [26].
Awareness of the different aspects of ED in T1DM patients may be helpful in prevention and treatment. Treatment involves a complex interplay of psychological and biological aspects and requires a multidisciplinary team. Psychological interventions should be implemented, as appropriate, to increase self-esteem and body acceptance, provide treatment if depression or anxiety exist, and to improve family communication [27]. Intensive insulin treatment, dietary restraint and hypoglycemic episodes should be addressed by the diabetes management team [18]. As good metabolic control is associated with weight gain, changes in target blood glucose level should be gradual. T1DM education should emphasize realistic targets of glucose levels with flexible non-depriving approaches to eating [28]. Setting higher than standard target blood glucose ranges (preprandial 120 to 150 mg/dL and postprandial < 200 mg/dL) may yield more benefit in the long run. In contrast, achieving excellent control may result in marked weight gain. Moreover, since low glucose target levels are associated with an increased risk of recurrent hypoglycemic episodes, which may result in additional increased calorie intake, setting higher target levels may be a better initial objective. Failure of outpatient treatment, the presence of a severe psychopathological state and poor glycaemic control are key elements in the decision for hospitalization [18]. Our patient was treated by a multidisciplinary team to address the medical, dietary and psychological aspects with gradual improvement.
In conclusion, this case demonstrates the importance of searching the underlying cause of DKA. The identification of ED among T1DM patients and the establishment of a comprehensive treatment may reduce the rate of DKA as well as the long term complications associated with these disorders.
Acknowledgement
We thank Ms. Cindy Cohen for her excellent editorial help.
References
-
Edge JA, Hawkins MM, Winter DL, Dunger DB (2001) The risk and outcome of cerebral oedema developing during diabetic ketoacidosis. Arch Dis Child 85: 16-22.
-
Rewers A, Chase HP, Mackenzie T, Walravens P, Roback M, et al. (2002) Predictors of acute complications in children with type 1 diabetes. JAMA 287: 2511-2518.
-
Gibb FW, Teoh WL, Graham J, Lockman KA (2016) Risk of death following admission to a UK hospital with diabetic ketoacidosis. Diabetologia 59: 2082-2087.
-
Olivieri L, Chasm R (2013) Diabetic ketoacidosis in the pediatric emergency department. Emerg Med Clin North Am 31: 755-773.
-
Maletkovic J, Drexler A (2013) Diabetic ketoacidosis and hyperglycemic hyperosmolar state. Endocrinol Metab Clin North Am 42: 677-695.
-
Umpierrez G, Freire AX (2002) Abdominal pain in patients with hyperglycemic crises. J Crit Care 17: 63-67.
-
Azoulay E, Chevret S, Didier J, Barboteu M, Bornstain C, et al. (2001) Infection as a trigger of diabetic ketoacidosis in intensive care-unit patients. Clin Infect Dis 32: 30-35.
-
Hanas R, Lindgren F, Lindblad B (2009) A 2-yr national population study of pediatric ketoacidosis in Sweden: predisposing conditions and insulin pump use. Pediatr Diabetes 10: 33-37.
-
Lohiya S, Kreisberg R, Lohiya V (2013) Recurrent diabetic ketoacidosis in two community teaching hospitals. Endocr Pract 19: 829-833.
-
Randall L, Begovic J, Hudson M, Smiley D, Peng L, et al. (2011) Recurrent diabetic ketoacidosis in inner-city minority patients: behavioral, socioeconomic, and psychosocial factors. Diabetes Care 34: 1891-1896.
-
Joshi N, Caputo GM, Weitekamp MR, Karchmer AW (1999) Infections in patients with diabetes mellitus. N Engl J Med 341: 1906-1912.
-
Hughes JW, Riddlesworth TD, DiMeglio LA, Miller KM, Rickels MR, et al. (2016) Autoimmune Diseases in Children and Adults with Type 1 Diabetes from the T1D Exchange Clinic Registry. J Clin Endocrinol Metab 101: 4931-4937.
-
Barker JM (2006) Clinical review: Type 1 diabetes-associated autoimmunity: natural history, genetic associations, and screening. J Clin Endocrinol Metab 91: 1210-1217.
-
(2013) American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders. (5th edn), (DSM-5). Arlington, VA.
-
Colton P, Olmsted M, Daneman D, Rydall A, Rodin G (2004) Disturbed eating behavior and eating disorders in preteen and early teenage girls with type 1 diabetes: a case-controlled study. Diabetes care 27: 1654-1659.
-
Stancin T, Link DL, Reuter JM (1989) Binge eating and purging in young women with IDDM. Diabetes Care 12: 601-603.
-
Wisting L, Froisland DH, Skrivarhaug T, Dahl-Jorgensen K, Ro O (2013) Disturbed eating behavior and omission of insulin in adolescents receiving intensified insulin treatment: a nationwide population-based study. Diabetes care 36: 3382-3387.
-
Pinhas-Hamiel O, Hamiel U, Levy-Shraga Y (2015) Eating disorders in adolescents with type 1 diabetes: Challenges in diagnosis and treatment. World J Diabetes 6: 517-526.
-
Golden MP, Herrold AJ, Orr DP (1985) An approach to prevention of recurrent diabetic ketoacidosis in the pediatric population. J Pediatr 107: 195-200.
-
Goebel-Fabbri AE (2008) Diabetes and eating disorders. J Diabetes Sci Technol 2: 530-532.
-
Markowitz JT, Butler DA, Volkening LK, Antisdel JE, Anderson BJ, et al. (2010) Brief screening tool for disordered eating in diabetes: internal consistency and external validity in a contemporary sample of pediatric patients with type 1 diabetes. Diabetes Care 33: 495-500.
-
Pinhas-Hamiel O, Hamiel U, Greenfield Y, Boyko V, Graph-Barel C, et al. (2013) Detecting intentional insulin omission for weight loss in girls with type 1 diabetes mellitus. Int J Eat Disord 46: 819-825.
-
Takii M, Uchigata Y, Tokunaga S, Amemiya N, Kinukawa N, et al. (2008) The duration of severe insulin omission is the factor most closely associated with the microvascular complications of Type 1 diabetic females with clinical eating disorders. Int J Eat Disord 41: 259-264.
-
Rydall AC, Rodin GM, Olmsted MP, Devenyi RG, Daneman D (1997) Disordered eating behavior and microvascular complications in young women with insulin-dependent diabetes mellitus. N Engl J Med 336: 1849-1854.
-
Goebel-Fabbri AE, Fikkan J, Franko DL, Pearson K, Anderson BJ, et al. (2008) Insulin restriction and associated morbidity and mortality in women with type 1 diabetes. Diabetes Care 31: 415-419.
-
Nielsen S, Emborg C, Mølbak AG (2002) Mortality in concurrent type 1 diabetes and anorexia nervosa. Diabetes Care 25: 309-312.
-
Nansel TR, Anderson BJ, Laffel LM, Simons-Morton BG, Weissberg-Benchell J, et al. (2009) A multisite trial of a clinic-integrated intervention for promoting family management of pediatric type 1 diabetes: feasibility and design. Pediatr diabetes 10: 105-115.
-
Rodin G, Olmsted MP, Rydall AC, Maharaj SI, Colton PA, et al. (2002) Eating disorders in young women with type 1 diabetes mellitus. J Psychosom Res 53: 943-949.





