International Journal of Cancer and Clinical Research
Magnetic Resonance Imaging Targeted Biopsies alone are not Sufficient for Detection of Clinically Significant Prostate Cancer - A Systematic Review and Meta-Analysis
Cayce B Nawaf1, Nathanial T Ondeck2, Laura A Skrip3, Rollin K Say1, John M Gunselman1,4, Jamil Syed1, Peter G Schulam1 and Preston C Sprenkle1*
1Department of Urology, Yale School of Medicine, New Haven, USA
2Yale School of Medicine, New Haven, USA
3Yale School of Public Health, New Haven, USA
4The Center for Men's and Women's Urology, Portland, USA
*Corresponding author:
Preston C Sprenkle, MD, Assistant Professor of Urology, Yale School of Medicine, Mail: PO Box 208058, New Haven, CT 06520-8058, USA, Ph: +1-203-785-7671, Fax: +1-203-737-8035, E-mail: preston.sprenkle@yale.edu
Int J Cancer Clin Res, IJCCR-3-067, (Volume 3, Issue 5), Original Research; ISSN: 2378-3419
Received: May 16, 2016 | Accepted: September 20, 2016 | Published: September 22, 2016
Citation: Nawaf CB, Ondeck NT, Skrip LA, Say RK, Gunselman JM, et al. (2016) Magnetic Resonance Imaging Targeted Biopsies alone are not Sufficient for Detection of Clinically Significant Prostate Cancer - A Systematic Review and Meta-Analysis. Int J Cancer Clin Res 3:067. 10.23937/2378-3419/3/5/1067
Copyright: © 2016 Nawaf CB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Context: Magnetic resonance imaging targeted biopsies (MRI-Tbx) has the potential to detect significant prostate cancer with a small number of cores; however, there are concerns that some clinically significant (CS) cancers may not be visible on MRI or identified with targeted biopsies alone.
Objective: We performed a systematic review and meta-analysis analyzing cancer detection rates of transrectal ultrasonography standard biopsies (TRUS-bx), MRI-Tbx and fusion biopsy (Fbx) (targeted plus standard).
Evidence acquisition: The authors conducted a systematic review, searching MEDLINE (OvidSP 1946 to Nov 7, 2014) and Embase (OvidSP 1974 to Nov 7, 2014). The search strategy was limited to the English language and 1990 to the present. Only studies that reported cancer detection rates on a per patient basis for TRUS-bx and MRI-Tbx were included.
Evidence synthesis: CS cancer was defined in each study. For the meta-analysis, effect sizes were calculated as single proportions or risk ratios. When no significant between-study heterogeneity was found (I2 > 50%), the fixed effects model was used to derive the pooled estimate. Otherwise, a random effects model was used. For the studies (n = 18) reporting fusion results, MRI-Tbx alone detected 89% of the significant cancers detected with Fbx (95% CI: 0.84 to 0.92). MRI-Tbx had a 22% higher detection rate of CS cancer than the TRUS-bx (RR: 1.22; 95% CI: 1.13 to 1.31, p < 0.001).
Conclusions: MRI-TBx alone missed 11% of significant cancers that were captured through fusion biopsy, strongly suggesting that targeted biopsy alone, without a 12-core standard biopsy, is inadequate to detect all CS prostate cancer.
Patient summary: In this report we compared the detection rates of two different types of prostate biopsies in a large population. We found that detection rates varied across the two methods. We conclude that the optimal approach to prostate biopsy includes using both the standard and targeted method.
Keywords
Biopsy, Image guided biopsy, Magnetic resonance imaging, Magnetic resonance imaging-guided targeted biopsy, Meta analysis, Prostate neoplasms, Software, Systematic review, Targeted biopsy
Introduction
Multiparametric-MRI (MP-MRI) has recently emerged as the most accurate imaging modality for the evaluation of localized prostate cancer. The addition of multi parametric examinations has improved detection such that MRI has evolved from intermittent use for staging purposes, to intra-prostatic lesion detection and tumor localization. Targeted biopsy of lesions identified by MP-MRI are associated with increased detection of clinically significant cancer compared to standard TRUS-guided biopsy (TRUS-Bx), while decreasing detection of indolent or clinically insignificant cancer [1,2]. Combining targeted biopsy with a standard biopsy, known as MRI Ultrasound Fusion Targeted Biopsy (MRI-Fbx), has a high negative predictive value for the exclusion of clinically significant disease, ranging from 63% to 98% [3]. An optimal biopsy technique would identify all clinically significant cancers while minimizing detection of indolent cancers, thereby avoiding the morbidity of repeat biopsies for missed significant disease, as well as minimizing the potential for overdetection and overtreatment of indolent cancers.
Although great strides have been made to increase the detection of clinically significant prostate cancer with the addition of MP-MRI, prostate biopsy remains a morbid and uncomfortable procedure [4-6]. Because of continued side effects and patient discomfort, efforts to decrease the morbidity of the procedure are always being pursued; one possibility is to reduce the number of biopsy cores removed. The improved accuracy of MRI-Fbx suggests that MRI-Targeted biopsy (MRI-Tbx) alone may be adequate to detect all clinically significant disease, thus allowing patients to avoid the 12 biopsy cores removed during a typical standard TRUS biopsy.
The advent of MRI-Tbx has prompted numerous studies comparing cancer detection rates of TRUS-bx, MRI-Tbx and Fbx [1,2,7-11]. As the technology is new and standardization is limited, sample sizes are small and can be difficult to compare. Several systematic reviews have attempted to collate the data and allow for a more centralized and powerful analysis [12-18].
Recently, Schoots, et al. published a meta-analysis indicating that MRI-Tbx has a higher rate of detection of clinically significant cancers and lower rate of detection of clinically insignificant cancer than standard TRUS-bx [15]. Their results support the improved diagnostic power of MRI-Tbx; however, it raises the question of the continuing necessity of standard TRUS-bx for the detection of prostate cancer. Therefore, this systematic review and meta-analysis was conducted to evaluate if targeted biopsy alone is adequate to detect clinically significant prostate cancer compared to a fusion prostate biopsy (Table 1).
![]()
Table 1: Final meta analysis.
View Table 1
Evidence Acquisition
Objective
Our aim was to systematically evaluate the detection rates of clinically significant and clinically insignificant cancer for MRI-Fbx, which is the combination of targeted biopsies and standard biopsy compared to MRI-Tbx alone. Additional sub-analyses were performed comparing biopsy status (biopsy naïve versus repeat biopsy), and cancer status (cancer naïve versus active surveillance).
Protocol and registration
Methods of the analysis and inclusion criteria were specified in advance and documented in a PICO B [19] protocol shown in Appendix A.
Eligibility criteria
Prospective and retrospective cohort studies comparing standard, targeted and fusion prostate biopsies were included. Study eligibility was dependent on the use of the English language and a post-1990 publication date. Participants of any age, race, prostate specific antigen level, biopsy status (biopsy naïve versus repeat biopsy) and cancer status (cancer naïve versus active surveillance) were considered. Studies comparing all cancer and significant cancer detection rates of targeted and fusion biopsies were selected. This review was limited to patients that received both a standard and targeted biopsy (patients served as their own control). A standard biopsy included either a TRUS Bxora transperineal template biopsy. Targeted biopsies included MRI In-Bore biopsy, MRI Cognitive Fusion and MRI Computer Fusion. The primary outcome measure was total cancer detection for each biopsy method stratified into both clinically insignificant and clinically significant cancer. The definition of significant cancer was defined individually in each study and is stated in table 2.
![]()
Table 2: Final meta analysis.
View Table 2
Information Sources and Search
The authors conducted a systematic review, searching MEDLINE (OvidSP 1946 to November 7, 2014) and Embase (OvidSP 1974 to November 7, 2014). The search strategy was limited to the English language and limited to 1990 to the present. All searches were conducted on November 7, 2014. The search strategies used controlled vocabulary terms and synonymous free text words to capture the concepts of prostate neoplasm and biopsy methods. The MEDLINE search strategy was translated into the syntax appropriate for Embase. The full strategy is shown in the Appendix B.
Study selection
Two reviewers (CBN and NTO) completed the initial eligibility assessment (abstract review), with concerns arbitrated by a third reviewer (PCS). The same two reviewers independently undertook the second eligibility assessment (paper review). PCS arbitrated all disagreements.
Data collection process
A data extraction sheet was developed and refined according to availability of data. Data was extracted by one author and confirmed by a second author (NTO and CBN). If data abstraction consensus was not reached with discussion, a third reviewer (PCS) made the final decision. Due to the potential repetitive publication of data, author names (typically senior author), institutions and sample sizes were identified. If there was any concern for duplication, the most recent publication was chosen (which also corresponded with the largest patient populations).
Data items
Information was extracted from each study including 1) characteristics of trial participants (number of participants, average age, mean PSA, number of participants who were undergoing their first biopsy, number who had previous negative biopsies and number who had previous positive biopsies), 2) standard biopsy characteristics (TRUS-Bx vs. transperineal, average number of cores, patients diagnosed with cancer, patients diagnosed with significant cancer, patients diagnosed with cancer detected by standard biopsy only), 3) MRI targeted biopsy characteristics (MRI In-Bore vs. MRI Cognitive Fusion vs. MRI Computer Fusion including fusion system, targeted cores per lesion, patients diagnosed with cancer, patients diagnosed with significant cancer, patients diagnosed with cancer detected by targeted biopsy only), 4) Fusion Biopsy (patients diagnosed with cancer, patients diagnosed with significant cancer), 5) significant cancer definition, 6) MRI characteristics (T1/T2, Diffusion Weighted Image/Apparent Fusion Coefficient, Dynamic Contrast Enhanced, Magnetic Resonance Spectroscopy, 1.5T/3.0T, Pelvic Array Coil/Endorectal Coil, Imaging Grading), 7) study information (title, year, senior author institution, clinical study design).
Statistical analysis (Risk of bias in individual studies, summary measures, synthesis of results)
For each included study, an estimate was calculated for the raw proportion of clinically significant cancers detected by fusion biopsy that were also detected by targeted biopsy. Likewise, a raw proportion of insignificant cancers detected by fusion biopsy and targeted biopsy was found. Pooled estimates were determined using a random effects model or fixed effects model depending on whether or not inter-study heterogeneity was significant (Cochran's I2 > 50%, Q Test P < 0.10), respectively.
Subgroup analyses were conducted to consider if the results varied with biopsy classification (i.e. biopsy naïve versus repeat biopsy) and cancer detection (i.e. cancer naïve versus active surveillance). A leave-one-out sensitivity analysis was also performed to determine whether any singular study was driving the findings such that the conclusion changed substantively with its removal from the overall analysis [20].
Potential publication bias was assessed using Egger's linear regression test [21]. All analyses were conducted using R Version 3.0.1 (The R Foundation for Statistical Computing). For any test or model, a P-value less than 0.05 were considered to be statistically significant.
Significant heterogeneity was found when analyzing detection of significant (Cochran's I2 = 72.97%, Q test P < 0.001) and insignificant (Cochran's I2 = 94.10%, Q test P < 0.001) cancer, such that the pooled estimates were calculated using random effects models. No significant publication bias was observed in the insignificant cancer analysis (P = 0.934). However, the random/mixed-effects Egger's test suggested the presence of publication bias in the significant cancer analysis (P = 0.002). As a result, the pooled estimates reflect the result of the trim and fill method of Duvall and Tweedie [22-25].
Results
Study selection
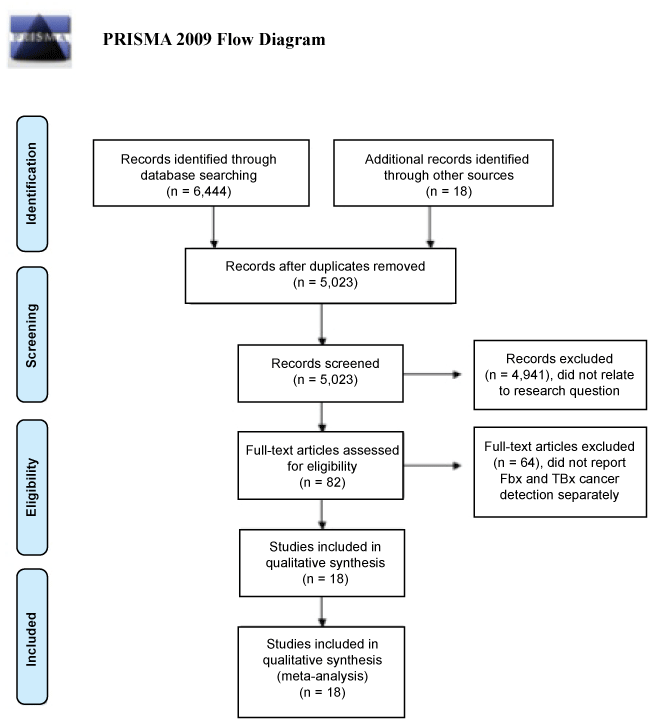
A search of MEDLINE and Embase yielded a total of 6,444 articles (Figure 1). After adjusting for duplicates, 5,023 articles remained. Title and abstract review screening yielded 82 potential studies with 4,941 studies clearly not meeting the inclusion criteria. The 82 papers selected for further review underwent a full text analysis, which returned 18 studies for inclusion.
Primary results
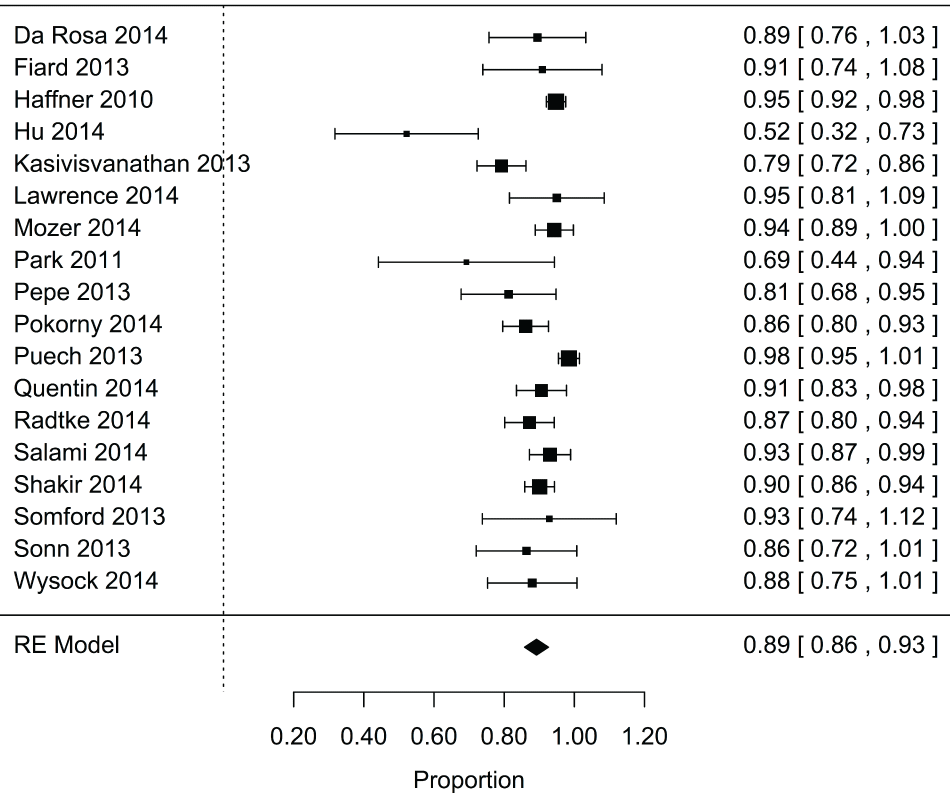
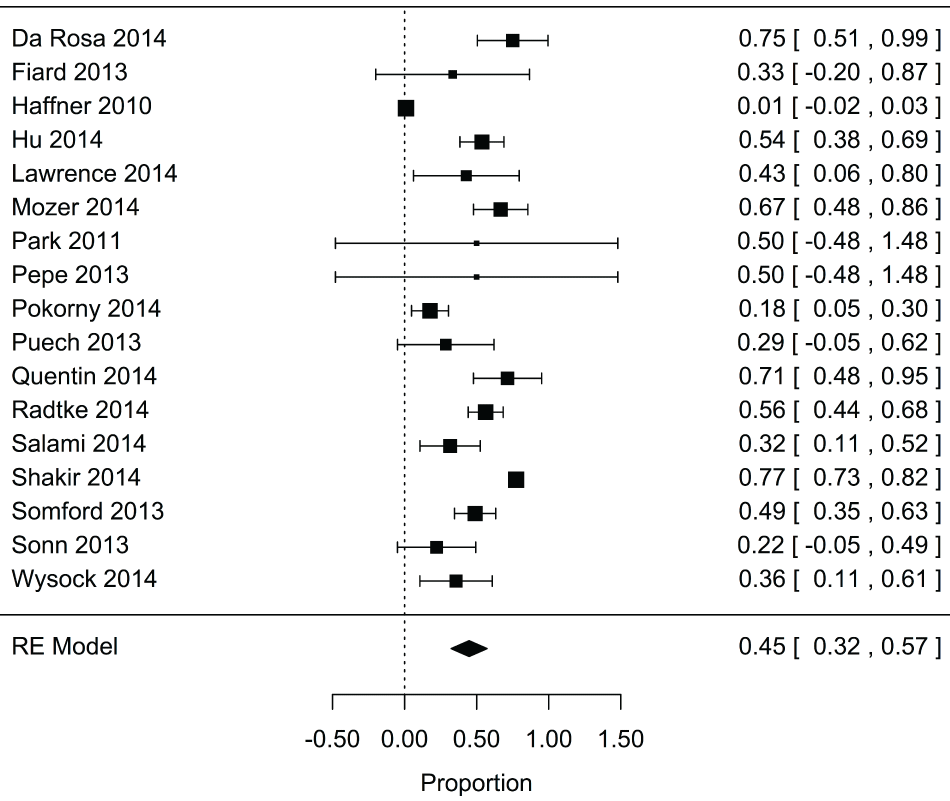
Based on the pooled results from 18 studies with 3,242 total patients, targeted biopsy detected 89% of significant cancers detected by fusion biopsy across the entire cohort (95% CI: 0.86, 0.93; P < 0.001) (Figure 2). In addition, an overall estimate calculated from 17 studies with 3,060 total patients suggested that only 45% of the insignificant cancers detected by fusion biopsy were also detected by targeted biopsy (95% CI: 0.32, 0.57; P < 0.001) (Figure 3).
.
Figure 2: Forest plot for proportion of significant cancers detected by fbx that were found using targeted biopsy.
View Figure 2
.
Figure 3: Forest plot for proportion of insignificant cancers detected by Fbx that were found using targeted biopsy alone.
View Figure 3
Subgroup analysis: (Accompanying figures available in supplement)
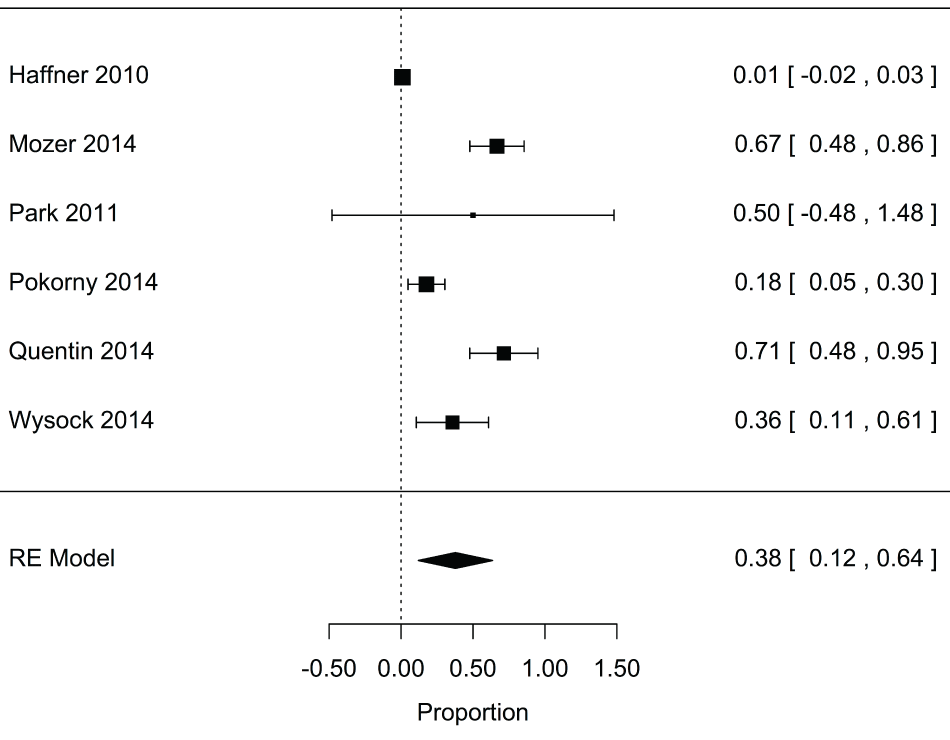
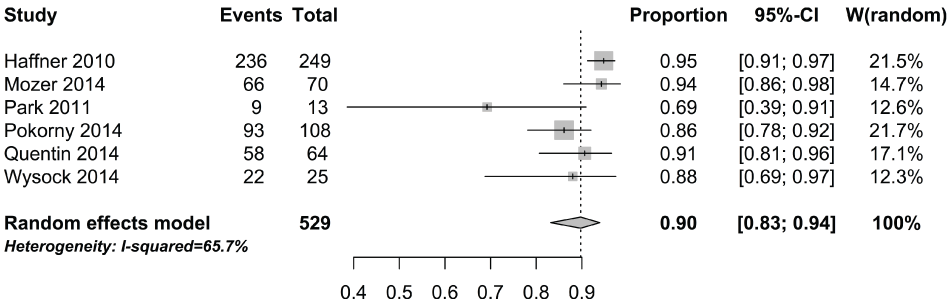
Studies were stratified according to whether patients were biopsy naïve (no previous biopsy) at the time of data collection or whether they had previously had a prostate biopsy (i.e. repeat biopsy). Only studies assessing these two cohorts were included in this subgroup analysis. For the six studies enrolling biopsy naïve patients (n = 1169 total patients) the pooled estimate for the proportion of insignificant cancers detected by fusion biopsy that were also detected by targeted biopsy was 0.36 (95% CI: 0.14, 0.59; P = 0.004) (Supplementary Figure 1). Overall, targeted biopsy detected 91% of significant cancers detected by fusion biopsy for biopsy naïve patients (95% CI: 0.85, 0.95; P < 0.001) (Supplementary Figure 2).
.
Supplementry Figure 1: Forest plot for proportion of insignificant cancers detected by fusion biopsy that were found using targeted biopsy alone for biopsy naive.
View Supplementry Figure 1
.
Supplementry Figure 2: Forest plot for proportion of significant cancers detected by fusion biopsy that were found using targeted biopsy alone for biopsy naive.
View Supplementry Figure 2
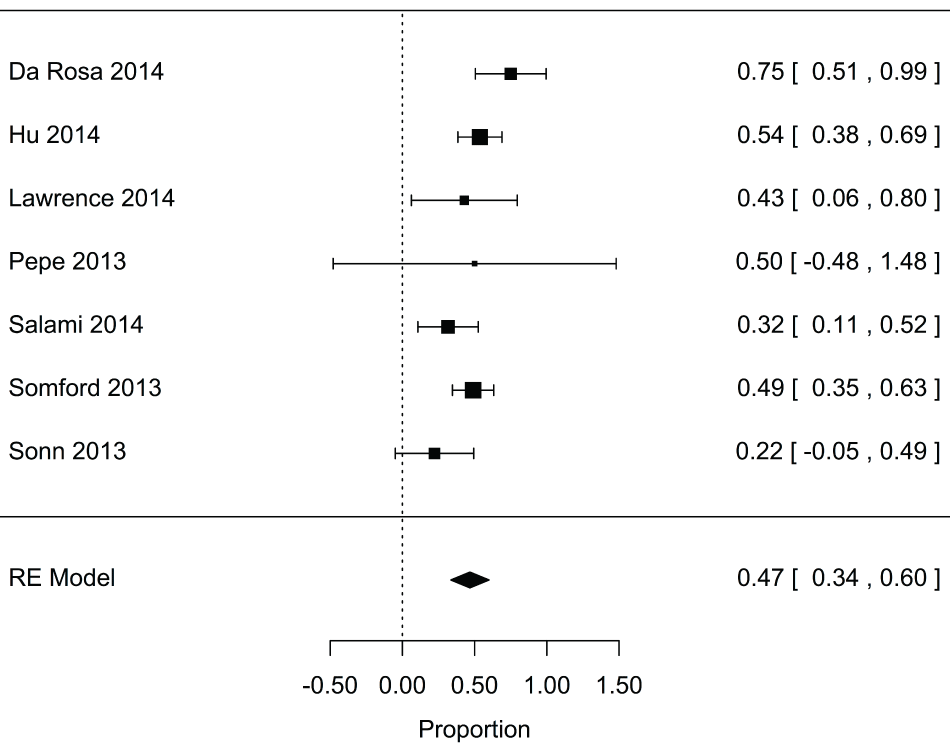
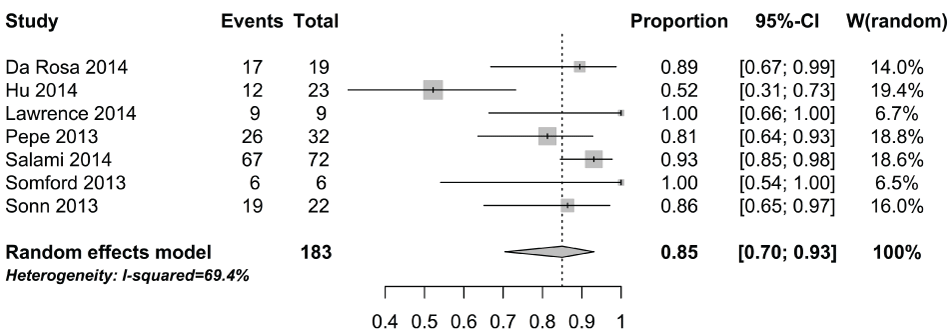
Seven studies with a total of 567 patients included repeat biopsy patients. The pooled estimate for the proportion of insignificant cancers detected by fusion biopsy and by targeted biopsy was 0.45 (95% CI: 0.33, 0.57; P < 0.001) (Supplementary Figure 3) for this subgroup of studies. Furthermore, among repeat biopsy patients, targeted biopsy detected 88% of significant cancers detected by fusion biopsy (95% CI: 0.75, 0.95; P < 0.001) (Supplementary Figure 4). Between-study heterogeneity was observed to decrease for both the insignificant and clinically significant cancer analyses upon sub grouping by biopsy classification. However, the random effects model was still used based on the results of the Cochran's I2 and Q Test.
.
Supplementry Figure 3: Forest plot for proportion of insignificant cancers detected by fusion biopsy that were found using targeted biopsy alone for repeat biopsy.
View Supplementry Figure 3
.
Supplementry Figure 4: Forest plot for proportion of significant cancers detected by fusion biopsy that were found using targeted biopsy alone for repeat biopsy.
View Supplementry Figure 4
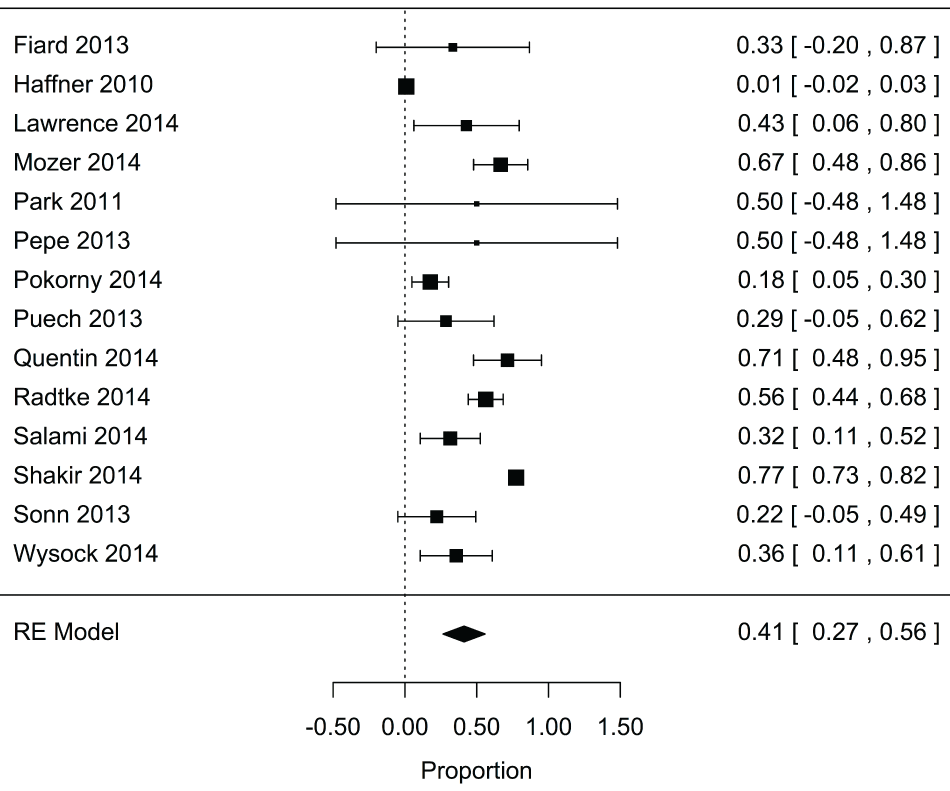
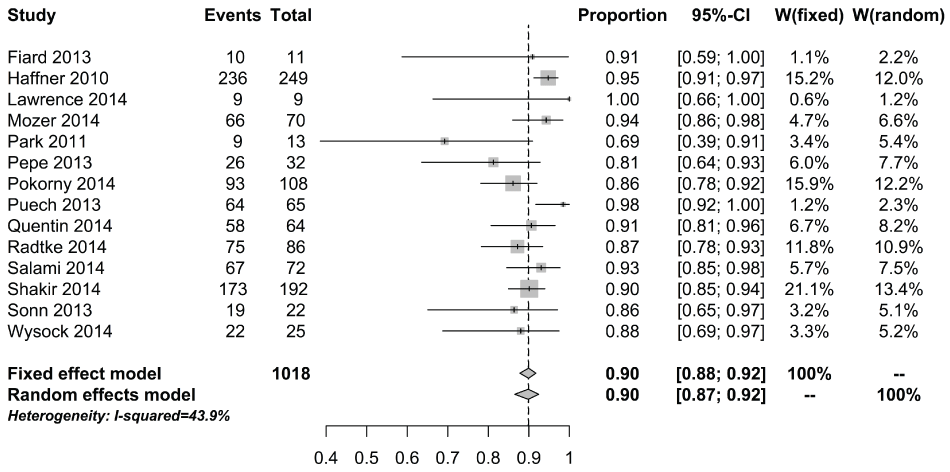
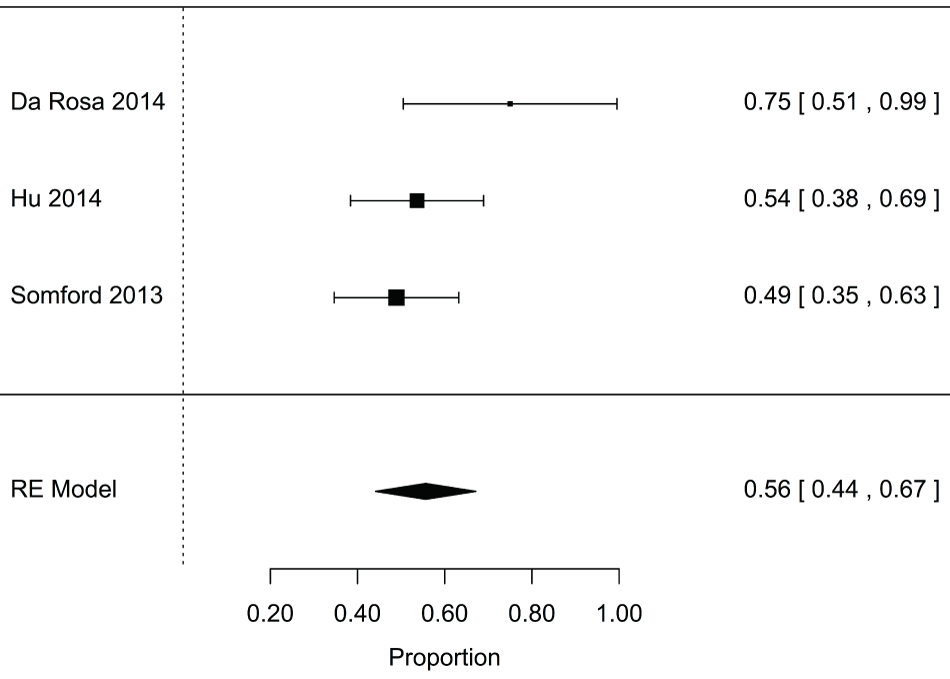
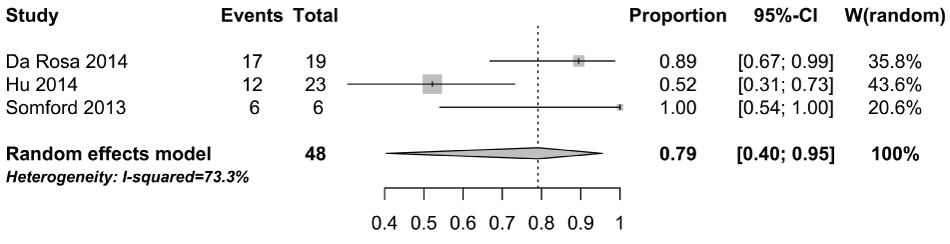
A second subgroup analysis was conducted according to the cancer detection history of patients. Fourteen studies included only patients without a prior cancer diagnosis (n = 2,844 total patients). The pooled estimate for the number of insignificant cancers detected by fusion biopsy and targeted biopsy was 0.41 (95% CI: 0.27, 0.56; P < 0.001) (Supplementary Figure 5). Among patients without a prior cancer diagnosis, 90% of clinically significant cancers detected by fusion biopsy were detected by targeted biopsy (95% CI: 0.87, 0.92; P < 0.001) (Supplementary Figure 6). Only three studies consisted of patients (n = 216 total patients) with previously diagnosed cancer-likely patients on active surveillance. Among these active surveillance patients, 56% of insignificant cancers detected by fusion biopsy were also detected by targeted biopsy (95% CI: 0.44, 0.67; P < 0.001) (Supplementary Figure 7). The proportion of clinically significant cancers detected by fusion biopsy also detected by targeted biopsy was 0.79 (95% CI: 0.40, 0.95; P < 0.001) (Supplementary Figure 8).
.
Supplementry Figure 5: Forest plot for proportion of insignificant cancers detected by fusion biopsy that were found using targeted biopsy alone for cancer naive.
View Supplementry Figure 5
.
Supplementry Figure 6: Forest plot for proportion of significant cancers detected by fusion biopsy that were found using targeted biopsy alone for cancer naive.
View Supplementry Figure 6
Biopsy technique information, including in-bore vs. cognitive fusion vs. computer fusion, was collected but these results did not reach statistical significance.
.
Supplementry Figure 7: Forest plot for proportion of insignificant cancers detected by fusion biopsy that were found using targeted biopsy alone for active surveillance.
View Supplementry Figure 7
.
Supplementry Figure 8: Forest plot for proportion of significant cancers detected by fusion biopsy that were found using targeted biopsy alone for active surveillance.
View Supplementry Figure 8
Sensitivity analysis
A leave-one-out sensitivity analysis was conducted for the primary outcomes. Leaving out any single study resulted in a range of pooled estimates between 0.42 (95% CI: 0.30, 0.54; P < 0.001) with the removal of Salami 2014 and 0.49 (95% CI: 0.38, 0.60; P < 0.001) with the removal of Haffner 2010. Similarly, the sensitivity analysis resulted in almost no change in the result for detection of clinically significant cancer, with the proportion of cancers detected by fusion biopsy and targeted biopsy ranging from slightly under 0.89 (95% CI: 0.85, 0.92; P < 0.001) with the removal of Puech 2013 to 0.90 (95% CI: 0.87, 0.93; P < 0.001) with the removal of Kasivisvanathan 2013.
Discussion
The ideal model for prostate cancer detection maximizes diagnostic accuracy while minimizing morbidity. In addition, distinguishing the clinically significant cancers from the insignificant cancers is essential and ultimately determines whether a patient proceeds to treatment or an active surveillance plan. MRI-Tbx strives to optimize detection of clinically significant lesions while obtaining the fewest biopsies necessary. In this review, however, targeted biopsy missed 11% of the clinically significant cancers that were detected by inclusion of a standard biopsy.
Detecting 11% more clinically significant cancer by the addition of standard biopsy to targeted biopsy is a relatively large improvement. These are typically all patients that would benefit from treatment. As a comparison, breast cancer diagnosis has been evolving throughout the decades, with core needle biopsies now increasingly favored over fine needle aspirations due to an increased sensitivity of over 10% of breast cancers, as shown in multiple studies [26-29].
MP-MRI is very good at detecting cancer and has been demonstrated in studies comparing preoperative MP-MRI to whole mount prostatectomy pathology to identify tumors greater than 0.5 ml, with a sensitivity and specificity of 86% and 94% respectively [30]. However, a recent manuscript by Le, et al. described the identification of clinically significant (Gl ≥ 7) cancer on final pathology that had not identified on MP-MRI in up to 28% of patients [31]. MRI-Fbx, which combines targeted and standard biopsy techniques, has been shown to have a high cancer detection rate of 85% and negative predictive value of 73% for the detection of clinically significant prostate cancer [2].
Registration of the MRI image to the real time ultrasound in the setting of fusion biopsy is a crucial step in adequate lesion sampling. Errors in registration can lead to inaccurate targeting and missed lesions. Wysock, et al. found improved cancer detection rate with a computer fusion system compared to cognitive fusion, but this was not statistically significant (p = 0.1374) [32].
In this study, missed clinically significant cancers were found across all patient populations with no systematic difference across targeted biopsy techniques. This suggests an error native to the imaging rather than an error in registration when performing targeted biopsy. While MP-MRI is a significant advance in prostate cancer detection, limitations of MRI technology and reader variability remain. More accurate identification and staging of intra-prostatic lesions are exciting areas for continued research and improvement in the field.
Detecting 11% fewer clinically significant cancers by avoiding a standard biopsy may not seem like a major decrease, but the benefits of avoiding additional biopsy cores is largely academic. Targeted biopsy requires the same patient preparation and clinical resources as a standard 12-core biopsy. The primary benefits of reducing the number of biopsy cores are a decreased procedure time and possibly a decrease in patient discomfort (if local anesthesia has not been fully effective). This gain in efficiency has not been quantified, and differences in morbidity have been demonstrated only when the number of samples reach high saturation levels, [33-35] whereas a combined targeted and TRUS guided biopsy rarely exceeds 30 samples. Despite this, some patients may prefer the fewest number of biopsies possible. This analysis provides a quantitative value of the risks associated with performing only MRI-Tbx and may be a valuable reference for counseling patients.
Considering the continued limitations of MRI technique and the evidence presented in this study, we strongly suggest that the current best strategy for diagnosis of prostatic cancer includes MRI-Tbx in addition to standard TRUS-Bx.
Strengths and limitations
Major strengths of our study are that it contains a larger sample size and number of studies than any other previously published meta-analysis, and uses strict inclusion criteria.
Several limitations may have influenced the findings, including the inconsistent definition of clinically significant cancer across studies, the non-standardized procedure of fusion targeted biopsy, as well as variability in MR characteristics, grading scale, threshold for biopsy, experience, and number of cores per target - this is, however, representative of the published literature.
Individual-level data were not available in all studies limiting the number of subgroup analyses that could be conducted, such as biopsy method (i.e. MRI in-bore, cognitive fusion, or computer fusion). There was also significant heterogeneity in the patient populations studied, but this was assessed in subgroup analyses.
Publication bias and small study effects can influence a meta-analysis because studies are less likely to publish negative results, which may overstate a beneficial effect. Selection bias may also influence the results: included studies evaluated patients with a positive MRI, which may have influenced the number of overall and significant prostate cancers detected. Although there have been studies which have demonstrated great negative predictive value of MRI, these results are not 100% and therefore lesions may have been missed, which can only be validated with radical prostatectomy and correlation with whole mounts.
Conclusions
Targeted biopsies alone would miss 11% of clinically significant cancer. The optimal approach to prostate biopsy includes combining both a standard biopsy, as well as targeted biopsies. This analysis provides a quantitative value of the risks associated with performing only MRI-Tbx, and may be a valuable reference for counseling patients. Large multicenter studies with standardized reporting are needed to further validate our findings.
Take Home Message
We compared the detection rates of two different types of prostate biopsies in a large population. The detection rates varied across the two methods. We conclude that the optimal approach to prostate biopsy includes both the standard and targeted method.
Disclosure
None of the authors have any disclosures to report.
References
-
Le JD, Stephenson S, Brugger M, Lu DY, Lieu P, et al. (2014) Magnetic resonance imaging-ultrasound fusion biopsy for prediction of final prostate pathology. J Urol 192: 1367-1373.
-
Siddiqui MM, Rais-Bahrami S, Turkbey B, George AK, Rothwax J, et al. (2015) Comparison of MR/ultrasound fusion-guided biopsy with ultrasound-guided biopsy for the diagnosis of prostate cancer. JAMA 313: 390-397.
-
Futterer JJ, Briganti A, De Visschere P, Emberton M, Giannarini G, et al. (2015) Can Clinically Significant Prostate Cancer Be Detected with Multiparametric Magnetic Resonance Imaging? A Systematic Review of the Literature. Eur Urol 68: 1045-1053.
-
Ubee SS, RR Marri, SJ Srirangam (2013) Pain related and overall morbidity with TRUS guided prostate biopsy--a prospective study. Int Braz J Urol 39: 671-674.
-
Pinsky PF, HL Parnes, G Andriole (2014) Mortality and complications after prostate biopsy in the Prostate, Lung, Colorectal and Ovarian Cancer Screening (PLCO) trial. BJU Int 113: 254-259.
-
Ehdaie B, Vertosick E, Spaliviero M, Giallo-Uvino A, Taur Y, et al. (2014) The impact of repeat biopsies on infectious complications in men with prostate cancer on active surveillance. J Urol 191: 660-664.
-
Da Rosa MR, Milot L, Sugar L, Vesprini D, Chung H, et al. (2015) A prospective comparison of MRI-US fused targeted biopsy versus systematic ultrasound-guided biopsy for detecting clinically significant prostate cancer in patients on active surveillance. J Magn Reson Imaging 41: 220-225.
-
Fiard G, Hohn N, Descotes JL, Rambeaud JJ, Troccaz J, et al. (2013) Targeted MRI-guided prostate biopsies for the detection of prostate cancer: initial clinical experience with real-time 3-dimensional transrectal ultrasound guidance and magnetic resonance/transrectal ultrasound image fusion. Urology 81: 1372-1378.
-
Mozer P, Rouprêt M, Le Cossec C, Granger B, Comperat E, et al. (2015) First round of targeted biopsies using magnetic resonance imaging/ultrasonography fusion compared with conventional transrectal ultrasonography-guided biopsies for the diagnosis of localised prostate cancer. BJU Int 115: 50-57.
-
10. Puech P, Rouvière O, Renard-Penna R, Villers A, Devos P, et al. (2013) Prostate cancer diagnosis: multiparametric MR-targeted biopsy with cognitive and transrectal US-MR fusion guidance versus systematic biopsy--prospective multicenter study. Radiology 268: 461-469.
-
Salami SS, Ben-Levi E, Yaskiv O, Ryniker L, Turkbey B, et al. (2015) In patients with a previous negative prostate biopsy and a suspicious lesion on magnetic resonance imaging, is a 12-core biopsy still necessary in addition to a targeted biopsy? BJU Int 115: 562-570.
-
Blomqvist L, Carlsson S, Gjertsson P, Heintz E, Hultcrantz M, et al. (2014) Limited evidence for the use of imaging to detect prostate cancer: a systematic review. Eur J Radiol 83: 1601-1606.
-
de Rooij M, Hamoen EH, Fütterer JJ, Barentsz JO, Rovers MM (2014) Accuracy of multiparametric MRI for prostate cancer detection: a meta-analysis. AJR Am J Roentgenol 202: 343-351.
-
Moore CM, Robertson NL, Arsanious N, Middleton T, Villers A, et al. (2013) Image-guided prostate biopsy using magnetic resonance imaging-derived targets: a systematic review. Eur Urol 63: 125-140.
-
Schoots IG, Roobol MJ, Nieboer D, Bangma CH, Steyerberg EW, et al. (2015) Magnetic resonance imaging-targeted biopsy may enhance the diagnostic accuracy of significant prostate cancer detection compared to standard transrectal ultrasound-guided biopsy: a systematic review and meta-analysis. Eur Urol 68: 438-450.
-
Valerio M, Donaldson I, Emberton M, Ehdaie B, Hadaschik BA, et al. (2015) Detection of Clinically Significant Prostate Cancer Using Magnetic Resonance Imaging-Ultrasound Fusion Targeted Biopsy: A Systematic Review. Eur Urol 68: 8-19.
-
van Hove A, Savoie PH, Maurin C, Brunelle S, Gravis G, et al. (2014) Comparison of image-guided targeted biopsies versus systematic randomized biopsies in the detection of prostate cancer: a systematic literature review of well-designed studies. World J Urol 32: 847-858.
-
Zhang ZX, Yang J, Zhang CZ, Li KA, Quan QM, et al. (2014) The value of magnetic resonance imaging in the detection of prostate cancer in patients with previous negative biopsies and elevated prostate-specific antigen levels: a meta-analysis. Acad Radiol 21: 578-589.
-
Schardt C, Adams MB, Owens T, Keitz S, Fontelo P (2007) Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med Inform Decis Mak 7: 16.
-
Rothstein H, AJ Sutton, M Borenstein (2005) Publication bias in meta-analysis : prevention, assessment and adjustments. Wiley, England 356.
-
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315: 629-634.
-
Duval S, R Tweedie (2000) Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56: 455-463.
-
Sue Duval, Richard Tweedie (2000) A nonparametric ‘trim and fill' method of accounting for publication bias in meta-analysis. Journal of the American Statistical Association 95: 89-99.
-
Sue Duval, Richard Tweedie (1998) Practical estimates of the effect of publication bias in meta-analysis. Australasian Epidemiologist 5: 14-17.
-
Sue Duval, Richard Tweedie (2000) Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56: 455-463.
-
Lam WW, Chu WC, Tse GM, Ma TK, Tang AP (2006) Role of fine needle aspiration and tru cut biopsy in diagnosis of mucinous carcinoma of breast--from a radiologist's perspective. Clin Imaging 30: 6-10.
-
Leifland K, U Lagerstedt, G Svane (2003) Comparison of stereotactic fine needle aspiration cytology and core needle biopsy in 522 non-palpable breast lesions. Acta Radiol 44: 387-391.
-
Nagar S, Anthony Iacco, Thomas Riggs, William Kestenberg, Richard Keidan (2012) An analysis of fine needle aspiration versus core needle biopsy in clinically palpable breast lesions: a report on the predictive values and a cost comparison. Am J Surg 204: 193-198.
-
Ohashi R, Matsubara M, Watarai Y, Yanagihara K, Yamashita K, et al. (2016) Diagnostic value of fine needle aspiration and core needle biopsy in special types of breast cancer. Breast Cancer 23: 675-683.
-
Puech P, Potiron E, Lemaitre L, Leroy X, Haber GP, et al. (2009) Dynamic contrast-enhanced-magnetic resonance imaging evaluation of intraprostatic prostate cancer: correlation with radical prostatectomy specimens. Urology 74: 1094-1099.
-
Le JD, Tan N, Shkolyar E, Lu DY, Kwan L, et al. (2015) Multifocality and prostate cancer detection by multiparametric magnetic resonance imaging: correlation with whole-mount histopathology. Eur Urol 67: 569-576.
-
Wysock JS, Rosenkrantz AB, Huang WC, Stifelman MD, Lepor H, et al. (2014) A prospective, blinded comparison of magnetic resonance (MR) imaging-ultrasound fusion and visual estimation in the performance of MR-targeted prostate biopsy: the PROFUS trial. Eur Urol 66: 343-351.
-
Patel A (2007) Finding a balanced strategy in prostate cancer diagnosis for case detection by prostate needle biopsy at first presentation: "all for more cores and more cores for all" or individualised sampling regimens? Which way forward? Eur Urol 52: 313-315.
-
Patel AR, Jones JS, Zhou M, Schoenfield L, Magi-Galluzzi C (2007) Parasagittal biopsies are more important as part of an initial biopsy strategy than as part of a repeat biopsy strategy: observations from a unique population. Prostate Cancer Prostatic Dis 10: 352-355.
-
Simon J, Kuefer R, Bartsch G Jr, Volkmer BG, Hautmann RE, et al. (2008) Intensifying the saturation biopsy technique for detecting prostate cancer after previous negative biopsies: a step in the wrong direction. BJU Int 102: 459-462.