International Journal of Cancer and Clinical Research
Rapid Fatal Progression of Cervical Cancer during Pregnancy Treated by Neoadjuvant Chemotherapy
Iptissem Naoura1*, Lise Selleret1, Frédéric Selle2 and Emile Daraï1
1Service de Gynécologie-obstétrique, Centre hospitalier universitaire Tenon, France
2Service d'oncologie médicale, Centre hospitalier universitaire Tenon, France
*Corresponding author:
Iptissem Naoura, Service de Gynécologie-obstétrique, Centre hospitalier universitaire Tenon, APHP, 4 rue de la Chine, 75020 Paris, France, Tel : +331-56-01-60-19, E-mail: iptissem.naoura@aphp.fr
Int J Cancer Clin Res, IJCCR-3-060, (Volume 3, Issue 3), Case Report; ISSN: 2378-3419
Received: April 27, 2016 | Accepted: June 27, 2016 | Published: June 29, 2016
Citation: Naoura I, Selleret L, Selle F, Daraï E (2016) Rapid Fatal Progression of Cervical Cancer during Pregnancy Treated by Neoadjuvant Chemotherapy. Int J Cancer Clin Res 3:060. 10.23937/2378-3419/3/3/1060
Copyright: © 2016 Naoura I, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Cervical cancer is the most commonly diagnosed gynaecological malignancy in pregnant women. The prognosis of this cancer does not seem to be influenced by the pregnancy. However, the cancer management comes into competition with maternal and foetal outcomes, especially during the 2nd trimester of pregnancy. Challenging issues lie in the cervical screening during pregnancy, the evaluation of lymph node status and the indication of neoadjuvant chemotherapy to avoid foetal prematurity without comprising maternal prognosis.
Case: We report a case of locally advanced cervical cancer diagnosed during the second trimester of pregnancy with rapid progression and fatal maternal outcome despite neoadjuvant chemotherapy.
Conclusion: Chemotherapy for cervical cancer during pregnancy is not always effective and maternal outcome could potentially be compromised.
Keywords
Cervical cancer, Pregnancy, Neoadjuvant chemotherapy
Teaching Points
- To evaluate lymph node status for patient with cervical cancer diagnosed during pregnancy is a main prognostic issue.
- The efficiency of neoadjuvant chemotherapy during pregnancy for patient diagnosed with cervical cancer remains debated.
- Identifying predictive factors of chemo-sensitivity of cervical cancer is necessary to perform a better selection of candidate for neoadjuvant therapy.
Introduction
Cervical cancer is the first gynaecological cancer diagnosed during pregnancy [1] but most cases (70%) are diagnosed at an early stage thanks to routine cervical screening in prenatal care.
Abnormal cervical cytology is encountered in around 5% of pregnant women whereas invasive cervical cancer affects only 1 to 2 women among 10 000 pregnancies [2,3]. Some authors report that maternal outcome is not worsened by pregnancy in particular for squamous cell types [2,4].
As in other cancers that occur during pregnancy, mother and child outcomes are weighed against each other. In this context, the management of patients with locally advanced cervical cancer remains controversial especially in the second trimester of pregnancy. Treatment is mainly influenced by the risk of lymphatic spread and distance metastases, gestational age, previous pregnancies and the parents' decision [1]. Termination is often recommended due to the poor prognosis of the disease. However, a few cases of neoadjuvant chemotherapy with preservation of the pregnancy have recently been reported suggesting that this option might be an alternative to systematic pregnancy termination.
We report a case of squamous cell carcinoma diagnosed during the second trimester of pregnancy with a rapid progression and a fatal maternal fatal outcome despite neoadjuvant therapy.
Case
A 30-year-old woman, gravida 1 para 0, was referred to the expert centre of pregnancy associated cancers (CALG) at Tenon Hospital for a cervical cancer diagnosed at 19 weeks of pregnancy. The Papanicolaou (Pap) smear test carried out at the first prenatal visit because of vaginal bleeding was negative although only endocervical cells were detected. Hence, vaginal bleeding during the first trimester of pregnancy was attributed to cervical ectropion.
At the visit corresponding to the 19th week of gestation, the physician diagnosed an exophytic cervical lesion measuring at least 4 cm without invasion of parametrium corresponding to IB2 FIGO stage. A cervical biopsy was performed and histology revealed an undifferentiated squamous cell carcinoma. Immunohistochemistry showed that cervical lesion was positive for p16 and p63 protein as well as for KL1 and CD 5/6 cytokeratin confirming the diagnosis of squamous cell carcinoma. No chromogranin, CD 56 neither synaptophysin expression was observed ruling out the neuroendocrine origin.
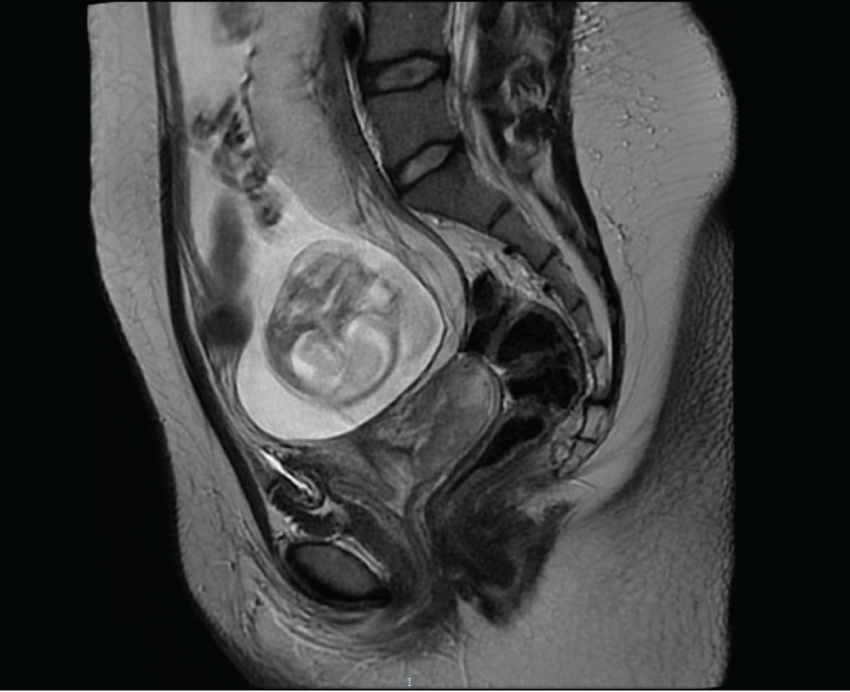
Magnetic Resonance Imaging (MRI) confirmed a 65 mm cervical lesion without nodal involvement (Figure 1).
.
Figure 1: At 19 weeks of pregnancy, magnetic resonance imaging detected a cervical cancer lesion of 65 mm exhibiting an extension to internal os of the cervix corresponding to IB2 FIGO stage.
View Figure 1
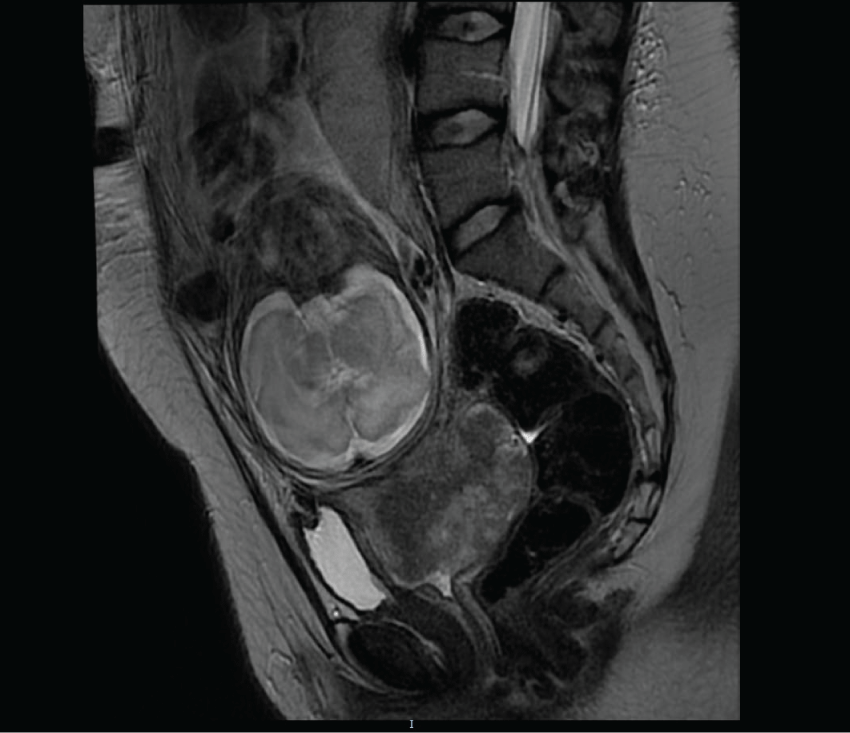
As the couple wished to conserve the pregnancy, neoadjuvant chemotherapy with carboplatin (5 AUC, 1160 mg/m2) and paclitaxel (175 mg/m2, 360 mg) was recommended. Fetal ultrasound during the first and second trimester showed no fetal malformation or intra-uterine growth restriction. After two cycles of chemotherapy, physical examination found an increase in tumour size confirmed by MRI showing a tumour of 8 cm of diameter with bilateral ureteral dilatation (Figure 2). In this context, preterm birth by caesarean section was organized at 29 weeks of gestation after foetal lung maturation (bethamethasone, 12 mg per day during 2 days). The infant girl weighed 1525 grams and had an Apgar score of 10 at 1 minute which decreased to 2 at 5 minutes justifying intubation for respiratory assistance. She remained in the hospital intensive care unit until she was 27 weeks old. Today the child is 4 months old and healthy. PET-FDG performed on the mother during the early postpartum period revealed a suspicious pelvic node but no uptake in the para-aortic areas. An extraperitoneal para-aortic lymphadenectomy was performed 2 weeks after the caesarean section and histology revealed metastases in 6 of the 16 lymph nodes. Exclusive concomitant radio-chemotherapy was consequently recommended (external beam radiotherapy: 45 Grays on the pelvis and a complement of 10 Grays on each iliac site associated with Cisplatin 30 mg/m2 weekly during 2 weeks then Topotecan, 4 mg/m2, one cycle).
.
Figure 2: Magnetic resonance imaging: cancer progression after neoadjuvant chemotherapy at 29 weeks of pregnancy with a cervical lesion of 8 cm.
View Figure 2
However, despite treatment, the patient developed intraperitoneal carcinomatosis with ascites, hydrodronephrosis and pulmonary embolism 11 weeks after the birth. The patient died of tumor progression 5 months after the initial diagnosis and 11 weeks after giving birth.
Discussion
Several studies have reported that pregnancy does not affect prognosis or survival of patients with early stages of cervical cancer justifying postponing treatment until after delivery [5-7]. However, this case report suggests that neoadjuvant chemotherapy administered during pregnancy is not always effective and can affect thus the prognosis compared to non pregnant patient.
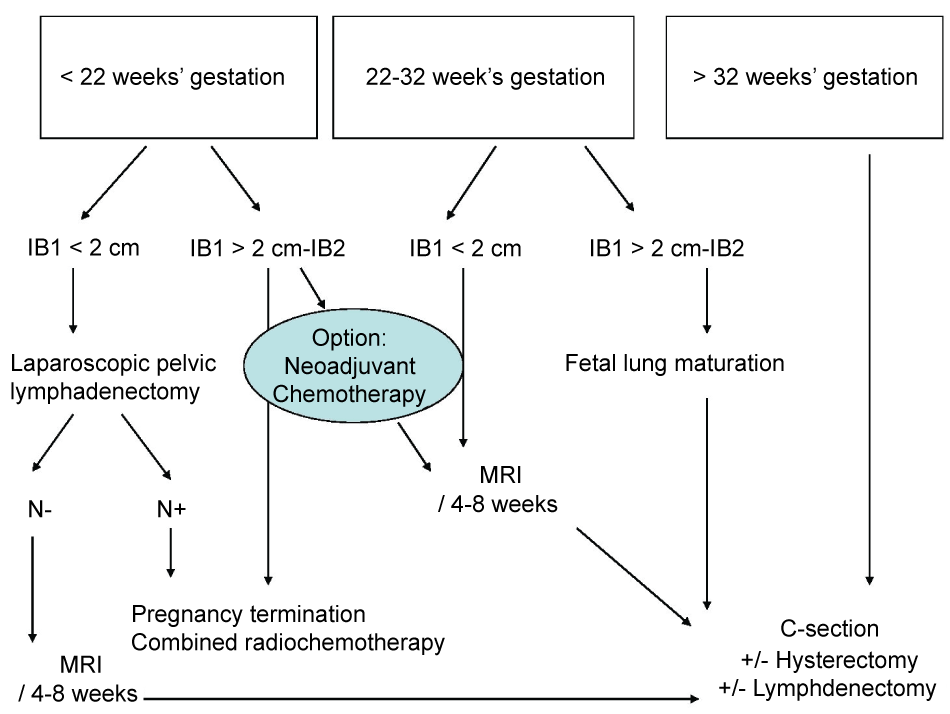
For pregnant patients with a tumour stage greater than IB1, guidelines for treatment differ from country to country [1,8] (Figure 3). The trimester during which the cancer is diagnosed and lymph node status are essential to determine therapeutic strategy. In the case reported here, two major arguments justified the decision to delay surgical lymph node staging: MRI did not show any suspicious lymph nodes; the pregnancy was in the 2nd trimester and the uterus was enlarged with numerous leiomyomas which may have compromised the feasibility of laparoscopic lymphadenectomy and increased maternal and foetal morbidities.
While a couple is more likely to accept termination of pregnancy when the cancer is diagnosed during the 1st trimester, physicians accept to delay treatment if the cancer is diagnosed during the last trimester [9]. However, when the cancer is diagnosed in the 2nd trimester, as in our case, the situation is less clear-cut and several ethical issues arise. Lymph node status assessment may help to make a decision but PET-FDG and MRI with gadolinium are not recommended during pregnancy. Although some authors have suggested that the 6mSv dose exposure delivered during PET-FDG exam at the 2nd trimester is acceptable for foetus [8], animal studies have demonstrated that foetal elimination of fluorine-18-labelled fluorodeoxyglucose is long [1,10]. MRI could be useful in evaluating tumour response to chemotherapy but it lacks accuracy in detecting lymph node metastasis [11]. Finally, the main challenge is evaluating the feasibility of para-aortic lymphadenectomy when the couple is not sure about preserving or terminating the pregnancy. In this specific setting, Alouini et al. reported good pregnancy outcome after para-aortic lymphadenectomy [11]. For patients with positive lymph nodes, termination of the pregnancy is mandatory as the optimal therapeutic option is concomitant radio-chemotherapy that is lethal for the foetus [12]. However, when the lymph nodes are negative, neoadjuvant chemotherapy is an option to reach foetal maturity. French recommendations accept to postpone optimal treatment for 6 to 8 weeks after diagnosis when the pregnancy term is over 22 weeks of gestation [13]. However, in the present case report, the couple's decision was clear in opting for conservative treatment which explains why no paraaortic lymphadenectomy was proposed.
Another major issue is how to evaluate whether the cervical tumour is chemosensitive.
Classic squamous cell type, especially undifferentiated as in the current case report, is more likely to respond to chemotherapy. Several cases of neoadjuvant chemotherapy for advanced stage cervical cancer during pregnancy with good outcome have been published [1,14-16]. However, there might be a publication bias leading to an overestimation of the safety and efficacy of chemotherapy for cervical cancer during pregnancy. In a review of 13 cases of stage IB2 cervical cancer diagnosed during pregnancy and treated with neoadjuvant chemotherapy, Morice et al. reported a recurrence rate of 54% [16].
It is impossible to know if the outcome of our patient would have been better if the pregnancy had been terminated at diagnosis. This underlines the need to identify additional predictive factors such as Ki-67, p53, p16, CK7 expression [17]. However, the fact that the tumour failed to respond to radio-chemotherapy after the birth and that there was rapid progression with development of peritoneal carcinomatosis, would suggest chemoresistance of the tumour.
Finally, the present case report underlines the risk of misinterpretation of Pap smear during pregnancy due to anatomic cervical transformation. In the present case report, the absence of squamous cell should have motived the physician to repeat cervical screening either by performing a Pap test ensuring that junction cells were collected or performing a colposcopy.
Pap smear currently has a sensitivity for detecting high-grade cervical neoplasia in non-pregnant patients in 70% to 80% [18]. In a study including 1593 pregnant compared to 214 375 non-pregnant patients, accuracy of Pap smear was not different between the population (27.6% for non-pregnant vs. 45% for pregnant patient) even there were more an overestimation of abnormal test during pregnancy [19]. This could be explained by large ectropion, frequent inflammation, presence of confusing decidual cells during pregnancy that could mimicked atypia. In the current case, the patient had effectively an ectropion and Pap smear did not note the presence of glandular cells. This underlines quality criteria of Pap smear imposing detecting both the presence of glandular and squamous cells.
The current case report highlights that, although neoadjuvant chemotherapy is an option to treat cervical cancer during pregnancy, chemotherapy is not always effective and maternal outcome could potentially be compromised. There is hence a need to identify predictive factors of chemo-sensitivity of cervical cancer to better select patients that could benefit from this therapeutic strategy.
References
-
Morice P, Uzan C, Gouy S, Verschraegen C, Haie-Meder C (2012) Gynaecological cancers in pregnancy. Lancet 379: 558‑569.
-
Al-Halal H, Kezouh A, Abenhaim HA (2013) Incidence and obstetrical outcomes of cervical intraepithelial neoplasia and cervical cancer in pregnancy: a population-based study on 8.8 million births. Arch Gynecol Obstet 287: 245‑250.
-
Creasman WT (2001) Cancer and pregnancy. Ann N Y Acad Sci 943: 281‑286.
-
Stensheim H, Moller B, van Dijk T, Fossa SD (2009) Cause-specific survival for women diagnosed with cancer during pregnancy or lactation: a registry-based cohort study. J Clin Oncol 27: 45‑51.
-
van Vliet W, van Loon AJ, ten Hoor KA, Boonstra H (1998) Cervical carcinoma during pregnancy: outcome of planned delay in treatment. Eur J Obstet Gynecol Reprod Biol 79: 153‑157.
-
Sood AK, Sorosky JI, Krogman S, Anderson B, Benda J, et al. (1996) Surgical management of cervical cancer complicating pregnancy: a case-control study. Gynecol Oncol 63: 294‑298.
-
Germann N, Haie-Meder C, Morice P, Lhomme C, Duvillard P, et al. (2005) Management and clinical outcomes of pregnant patients with invasive cervical cancer. Ann 16: 397‑402.
-
Amant F, Brepoels L, Halaska MJ, Gziri MM, Calsteren KV (2010) Gynaecologic cancer complicating pregnancy: an overview. Best Pract Res Clin Obstet Gynaecol 24: 61‑79.
-
Han SN, Kesic VI, Van Calsteren K, Petkovic S, Amant F (2013) Cancer in pregnancy: a survey of current clinical practice. Eur J Obstet Gynecol Reprod Biol 167: 18‑23.
-
Bartlett RM, Nickles RJ, Barnhart TE, Christian BT, Holden JE (2010) Fetal dose estimates for (18)F-fluoro-L-thymidine using a pregnant monkey model. J Nucl Med 51: 288‑292.
-
Alouini S, Rida K, Mathevet P (2008) Cervical cancer complicating pregnancy: implications of laparoscopic lymphadenectomy. Gynecol Oncol 108: 472‑477.
-
Stovall M, Blackwell CR, Cundiff J, Novack DH, Palta JR, et al. (1995) Fetal dose from radiotherapy with photon beams: report of AAPM Radiation Therapy Committee Task Group No. 36. Med Phys 22: 63‑82.
-
Morice P, Narducci F, Mathevet P, Marret H, Darai E, et al. (2009) French recommendations on the management of invasive cervical cancer during pregnancy. Int J Gynecol Cancer 19: 1638‑1641.
-
Benhaim Y, Pautier P, Bensaid C, Lhommé C, Haie-Meder C, et al. (2008) Neoadjuvant chemotherapy for advanced stage cervical cancer in a pregnant patient: report of one case with rapid tumor progression. Eur J Obstet Gynecol Reprod Biol 136: 267‑268.
-
Fruscio R, Villa A, Chiari S, Vergani P, Ceppi L, et al. (2012) Delivery delay with neoadjuvant chemotherapy for cervical cancer patients during pregnancy: a series of nine cases and literature review. Gynecol Oncol 126: 192‑197.
-
Morice P, Uzan C, Leary A (2012) Are the outcomes of neoadjuvant chemotherapy for stage IB2 cervical cancer similar in pregnant and nonpregnant patient? Gynecol Oncol 127: 257-259.
-
Iida M, Banno K, Yanokura M, Nakamura K, Adachi M, et al. (2014) Candidate biomarkers for cervical cancer treatment: Potential for clinical practice (Review). Mol Clin Oncol 2: 647‑655.
-
Sherman ME, Schiffman M, Herrero R, Kelly D, Bratti C, et al. (1998) Performance of a semiautomated Papanicolaou smear screening system: results of a population-based study conducted in Guanacaste, Costa Rica. Cancer 84: 273‑280.
-
Morimura Y, Fujimori K, Soeda S, Hashimoto T, Takano Y, et al. (2002) Cervical cytology during pregnancy--comparison with non-pregnant women and management of pregnant women with abnormal cytology. Fukushima J Med Sci 48: 27‑37.